Advanced Catalytic Synthesis of 2-Hydroxy-N-(4'-chlorobiphenyl-2-yl)nicotinamide for High-Purity Agrochemical Standards
The global agrochemical sector continuously demands higher purity standards for fungicide active ingredients, particularly for succinate dehydrogenase inhibitors like Boscalid. A critical challenge in maintaining these standards is the availability of high-purity reference substances for impurity profiling. Patent CN111039860A introduces a breakthrough synthetic methodology for 2-hydroxy-N-(4'-chlorobiphenyl-2-yl)nicotinamide, a key impurity and degradation product associated with Boscalid manufacturing. This innovation addresses the longstanding inefficiencies in producing this specific nicotinamide derivative, which is essential for quality control laboratories and process development teams. By shifting from complex multi-step sequences to a direct catalytic condensation, the technology offers a robust pathway for generating analytical standards with improved economic and operational metrics.
For research and development directors overseeing agrochemical portfolios, the ability to rapidly synthesize precise impurity standards is vital for regulatory compliance and process optimization. The traditional reliance on low-yield extraction from reaction byproducts or inefficient total synthesis creates bottlenecks in analytical method validation. This patent provides a dedicated, scalable route that decouples the production of the standard from the main API synthesis line, ensuring a consistent supply of characterization material. The technical depth of this approach lies in its catalyst selection, which enables direct amide bond formation under relatively mild thermal conditions, bypassing the need for hazardous activating agents typically required in classical peptide coupling strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Boscalid and its associated impurities has relied on cumbersome pathways involving organometallic chemistry and harsh activation steps. As illustrated in the traditional reaction scheme, the conventional route to the biphenyl scaffold often necessitates a Grignard reaction followed by a Suzuki coupling, hydrogenation, and finally amidation using thionyl chloride.
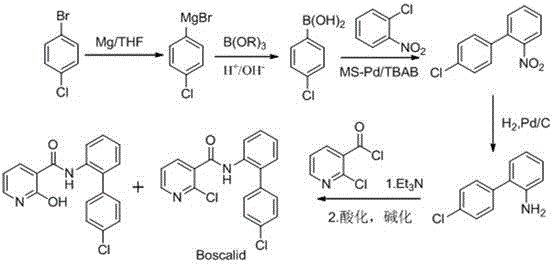
This multi-step sequence is not only capital intensive due to the requirement for expensive palladium catalysts and strictly anhydrous conditions but also poses significant safety risks associated with large-scale Grignard reagent handling. Furthermore, the final amidation step using thionyl chloride generates substantial amounts of hydrochloric acid and sulfur dioxide, necessitating complex scrubbing systems and leading to product instability. Alternative literature methods, such as the Smiles rearrangement approach, have been explored but suffer from critically low overall yields, often reported around 35%, making them commercially unviable for the production of bulk reference standards. These inefficiencies result in high costs and long lead times for obtaining the necessary impurity profiles required by regulatory bodies.
The Novel Approach
In stark contrast, the methodology disclosed in CN111039860A streamlines the synthesis into a direct, one-step condensation reaction. By reacting 2-chloronicotinic acid directly with 4'-chloro-2-aminobiphenyl in the presence of specific Lewis acid or boronic acid catalysts, the process eliminates the need for pre-activation of the carboxylic acid into an acid chloride.
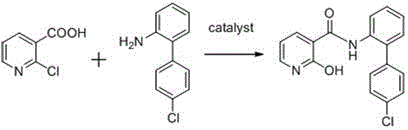
This novel approach utilizes catalysts such as 3-nitrophenylboronic acid or zirconium chloride to facilitate the nucleophilic attack of the amine on the carboxylic acid carbonyl. The reaction proceeds in common organic solvents like toluene or dioxane at temperatures ranging from 50°C to 200°C. The elimination of the acid chloride intermediate significantly reduces the formation of side products and avoids the generation of corrosive gaseous byproducts. Experimental data from the patent indicates that this direct route achieves yields exceeding 50.48%, with optimized examples reaching over 60% crude yield before recrystallization. This represents a substantial improvement in atom economy and process efficiency compared to the prior art, providing a reliable source of high-purity material for analytical applications.
Mechanistic Insights into Boronic Acid Catalyzed Direct Amidation
The core chemical innovation in this patent lies in the utilization of boronic acids and metal salts as catalysts for direct thermal amidation. Unlike traditional coupling reagents like DCC or EDC which form stoichiometric waste, these catalysts operate by coordinating with the carbonyl oxygen of the carboxylic acid, thereby increasing the electrophilicity of the carbonyl carbon. For instance, 3-nitrophenylboronic acid, with its electron-withdrawing nitro group, acts as a potent Lewis acid activator. This activation lowers the energy barrier for the nucleophilic attack by the aniline nitrogen of the 4'-chloro-2-aminobiphenyl. The reaction likely proceeds through a tetrahedral intermediate which subsequently eliminates water to form the stable amide bond. The choice of solvent, such as toluene, plays a crucial role in azeotropically removing the water generated during the reaction, driving the equilibrium towards product formation according to Le Chatelier's principle.
From an impurity control perspective, this mechanism is highly advantageous because it avoids the formation of reactive acid chlorides that can lead to over-chlorination or polymerization side reactions. The specificity of the catalyst ensures that the reaction occurs primarily at the carboxylic acid moiety of the nicotinic acid derivative, leaving the chloro-substituents on the aromatic rings intact. This chemoselectivity is paramount for producing the exact structural analogue required for HPLC calibration. The workup procedure described, involving washes with saturated sodium bicarbonate and brine, effectively removes unreacted acid and catalyst residues without exposing the sensitive amide bond to harsh acidic or basic hydrolysis conditions that could degrade the product. This gentle purification strategy contributes to the high recovery rates observed in the recrystallization steps.
How to Synthesize 2-Hydroxy-N-(4'-chlorobiphenyl-2-yl)nicotinamide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins with the dissolution of the acid and amine starting materials in a suitable solvent, followed by the addition of the catalyst and heating. Detailed standardized synthesis steps are provided below to guide technical teams in replicating this high-yield process.
- Dissolve 2-chloronicotinic acid in a solvent such as toluene, add 4'-chloro-2-aminobiphenyl and a catalyst like 3-nitrophenylboronic acid, then heat to 120°C for reaction.
- Wash the reaction mixture sequentially with saturated sodium bicarbonate solution and saturated sodium chloride solution to remove acidic impurities and residual salts.
- Concentrate the organic phase under reduced pressure and recrystallize the residue using a solvent system like ethyl acetate and petroleum ether to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible operational improvements and risk mitigation. The shift from a multi-step organometallic sequence to a single-pot condensation drastically simplifies the manufacturing workflow. This simplification reduces the number of unit operations, thereby lowering the consumption of utilities such as steam and cooling water, and minimizing the manpower required for reactor charging and discharging. The elimination of pyrophoric Grignard reagents and toxic thionyl chloride removes significant safety hazards, potentially lowering insurance premiums and reducing the need for specialized containment equipment. These factors collectively contribute to a more resilient and cost-effective supply chain for agrochemical intermediates.
- Cost Reduction in Manufacturing: The direct amidation strategy eliminates the need for expensive palladium catalysts and stoichiometric coupling reagents, which are major cost drivers in fine chemical synthesis. By utilizing inexpensive boronic acid derivatives or metal salts as catalysts, the raw material cost profile is significantly optimized. Furthermore, the higher yield means that less starting material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final reference standard. The simplified workup procedure also reduces solvent consumption and waste disposal costs, aligning with green chemistry principles and reducing the environmental compliance burden.
- Enhanced Supply Chain Reliability: The starting materials, 2-chloronicotinic acid and 4'-chloro-2-aminobiphenyl, are commercially available commodity chemicals with stable supply chains, unlike specialized organometallic intermediates which may have long lead times. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, ensures consistent production output even with minor variations in raw material quality. This reliability is crucial for maintaining continuous quality control operations in fungicide manufacturing plants, preventing delays in batch release due to a lack of analytical standards.
- Scalability and Environmental Compliance: The process is inherently scalable as it avoids exothermic hazards associated with Grignard formations and gas evolution from acid chloride generation. The use of common solvents like toluene allows for easy recovery and recycling via distillation, minimizing volatile organic compound (VOC) emissions. The aqueous workup generates saline wastewater that is easier to treat compared to the heavy metal-contaminated waste streams from palladium-catalyzed couplings. This environmental compatibility facilitates smoother regulatory approvals for manufacturing sites and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: What is the primary advantage of this new synthesis method over traditional routes?
A: The primary advantage is the significant improvement in yield, achieving over 50.48% compared to the 35% yield of previous two-step methods, while simplifying the process to a single catalytic step.
Q: Which catalysts are effective for this direct amidation reaction?
A: The patent identifies several effective catalysts including zirconium chloride, n-butylstannic acid, boric acid, 3,4,5-trifluorophenylboronic acid, and 3-nitrophenylboronic acid.
Q: How is this compound utilized in the Boscalid industry?
A: It serves as a critical reference standard for monitoring the production process of Boscalid and for detecting and quantifying this specific impurity in the final fungicide product to ensure quality compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-N-(4'-chlorobiphenyl-2-yl)nicotinamide Supplier
As the agrochemical industry evolves towards stricter quality controls, the demand for certified impurity standards like 2-hydroxy-N-(4'-chlorobiphenyl-2-yl)nicotinamide continues to grow. NINGBO INNO PHARMCHEM stands ready to support your quality assurance initiatives with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this synthesis, ensuring stringent purity specifications and rigorous QC labs verify every batch against international pharmacopoeia standards. We understand the critical nature of reference materials in regulatory filings and are committed to delivering consistency and reliability.
We invite you to collaborate with us to optimize your supply chain for Boscalid intermediates and standards. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance your operational efficiency and product quality.
