Advanced Asymmetric Hydrogenation Technology for High-Purity N-benzyl Phenylephrine Intermediates
Introduction to Next-Generation Chiral Synthesis
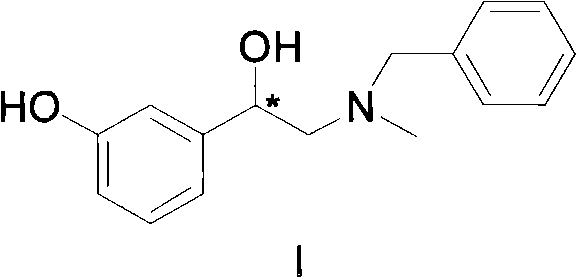
The pharmaceutical industry continuously demands more efficient and cost-effective routes for producing optically active intermediates, particularly for adrenomimetic drugs like phenylephrine derivatives. Patent CN102381990A introduces a groundbreaking preparation method for optically active N-benzyl phenylephrine, a critical intermediate in the synthesis of neophryn and related therapeutic agents. This technology leverages a sophisticated chiral diphosphine-chiral diamine transition metal asymmetric hydrogenation catalyst to transform a ketone precursor into the desired chiral alcohol with exceptional precision. By shifting away from expensive noble metals and complex purification protocols, this innovation addresses the dual challenges of economic viability and environmental sustainability in fine chemical manufacturing. The method ensures high product yield, superior chemical purity, and outstanding optical purity, making it an ideal candidate for large-scale industrial adoption by forward-thinking pharmaceutical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, specifically those documented in EP 1147075, relied heavily on rhodium-based catalysts to effect the asymmetric reduction of N-benzyl-N-methyl-3-hydroxyl-α-methyl phenyl ketone amine. While functional, these legacy processes suffer from significant economic and operational drawbacks that hinder their scalability. The primary issue lies in the prohibitive cost of rhodium catalysts, which places a heavy burden on the raw material budget for bulk production. Furthermore, the optical purity achieved through these conventional rhodium-catalyzed reactions often falls short of stringent pharmaceutical requirements, typically yielding an enantiomeric excess (ee) value of only around 88%. To bridge this gap and reach the necessary 96% ee threshold, manufacturers were forced to implement tedious and yield-destructive purification steps. This additional processing not only complicates the workflow but also results in a substantial loss of product, estimated at approximately 30%, which is both economically wasteful and environmentally burdensome due to increased solvent and energy consumption.
The Novel Approach
In stark contrast, the methodology disclosed in CN102381990A utilizes a ruthenium-based catalytic system that fundamentally redefines the efficiency profile of this synthesis. By employing a chiral diphosphine-chiral diamine ruthenium complex, the new process achieves asymmetric hydrogenation with remarkable selectivity and activity under relatively mild conditions. This approach eliminates the dependency on costly rhodium, immediately lowering the catalyst cost component of the bill of materials. More importantly, the reaction delivers high conversion rates exceeding 99% alongside optical purity values ranging from 95% to 99.9% directly from the reactor. This high initial purity obviates the need for the yield-sacrificing purification steps required by older methods, thereby preserving the theoretical yield and significantly enhancing the overall process mass intensity. The simplicity of the aftertreatment, involving merely neutralization, filtration, and concentration, further streamlines the operation, reducing cycle times and labor costs while minimizing waste generation.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise design of the transition metal catalyst, which orchestrates the stereoselective addition of hydrogen to the carbonyl group. The catalyst system comprises a central ruthenium atom coordinated with a chiral diphosphine ligand and a chiral diamine ligand, forming a highly active and selective complex. The chiral environment created by the bulky phosphine ligands, such as BINAP, SEGPHOS, or their derivatives, effectively shields one face of the ketone substrate, directing the hydride attack to the opposite face to generate the desired R or S configuration. This steric control is paramount for achieving the high enantiomeric excess values observed in the examples, ensuring that the final product meets the rigorous stereochemical standards required for biological activity. The synergy between the diphosphine and diamine components facilitates a concerted mechanism that lowers the activation energy for hydrogenation, allowing the reaction to proceed efficiently even at ambient temperatures and moderate hydrogen pressures.
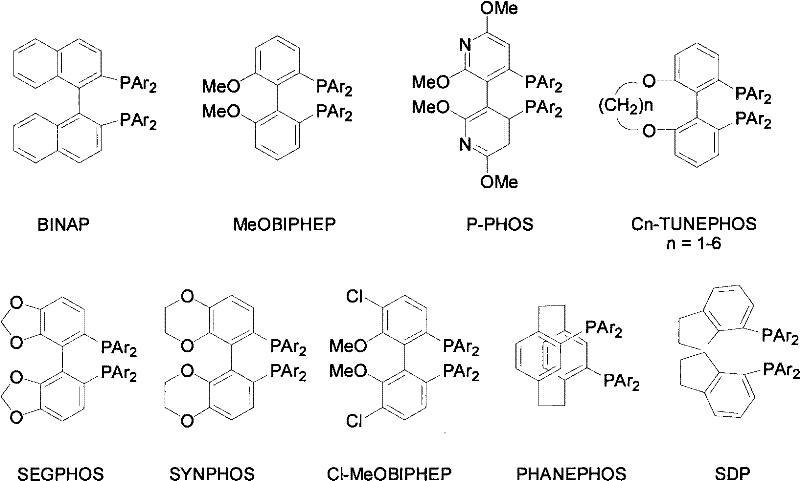
Beyond stereocontrol, the choice of ligands plays a critical role in impurity management and catalyst longevity. The robust nature of the ruthenium-phosphine-diamine complex ensures stability throughout the reaction course, minimizing the formation of side products that could arise from catalyst decomposition or non-selective background reactions. The patent highlights that by selecting specific configurations of the phosphine part, manufacturers can precisely dictate the absolute configuration of the hydrogenated product, offering flexibility to produce either enantiomer as needed for different drug candidates. This level of control is essential for maintaining a clean impurity profile, as it reduces the burden on downstream purification units to separate closely related diastereomers or regioisomers. Consequently, the mechanistic elegance of this catalytic system translates directly into a cleaner crude product, simplifying the path to high-purity final APIs and reducing the risk of genotoxic impurities often associated with heavy metal residues.
How to Synthesize N-benzyl Phenylephrine Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction kinetics with operational safety. The process begins with the suspension of the ketone precursor, typically in its hydrochloride salt form, within a polar protic solvent such as isopropanol, which serves both as a reaction medium and a proton source. An alkaline assistant, preferably potassium tert-butoxide, is added to activate the catalyst and facilitate the deprotonation steps essential for the catalytic cycle. Once the catalyst, such as Xyl-BINAP RuCl2DAIPEN, is introduced under an inert atmosphere, the mixture is subjected to hydrogen pressure in a standard autoclave setup. The detailed standardized synthetic steps see the guide below.
- Prepare the reaction mixture by suspending the ketone precursor (Compound II hydrochloride) in a suitable alcohol solvent such as isopropanol.
- Add the alkaline assistant (e.g., potassium tert-butoxide) and the chiral ruthenium catalyst (e.g., Xyl-BINAP RuCl2DAIPEN) under a nitrogen atmosphere.
- Transfer the mixture to an autoclave, pressurize with hydrogen (150-500 psi), and stir at room temperature until conversion exceeds 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ruthenium-catalyzed process presents a compelling value proposition centered on cost stability and operational efficiency. By replacing expensive rhodium catalysts with more abundant and affordable ruthenium alternatives, the direct material costs associated with catalysis are drastically reduced, providing a buffer against volatile precious metal markets. Additionally, the elimination of complex purification stages means that fewer unit operations are required, leading to reduced utility consumption, lower labor hours, and decreased solvent usage per kilogram of product. These factors collectively contribute to a leaner manufacturing process that enhances margin potential while maintaining high quality standards.
- Cost Reduction in Manufacturing: The transition to a ruthenium-based catalyst system inherently lowers the cost of goods sold by removing the reliance on premium-priced rhodium. Furthermore, the high conversion efficiency and superior optical purity achieved in the primary reaction step mean that yield losses associated with recrystallization or chromatographic purification are virtually eliminated. This maximizes the output from every batch of raw materials, effectively spreading fixed costs over a larger volume of saleable product and driving down the unit cost significantly without compromising on quality specifications.
- Enhanced Supply Chain Reliability: The simplicity of the reaction workup, which involves basic neutralization and filtration rather than complex separations, shortens the overall production cycle time. Faster cycle times translate to increased throughput capacity within existing facilities, allowing suppliers to respond more agilely to fluctuating market demands. Moreover, the use of commercially available and stable reagents ensures that the supply chain is less vulnerable to disruptions caused by the scarcity of specialized or exotic chemicals, thereby securing a continuous and reliable flow of intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: The mild reaction conditions, often operable at room temperature and moderate pressures, make this process highly scalable from pilot plant to commercial production scales with minimal engineering hurdles. The reduction in solvent usage and the avoidance of yield-destroying purification steps also align with green chemistry principles, resulting in a smaller environmental footprint. This ease of scale-up combined with improved waste profiles facilitates smoother regulatory approvals and supports corporate sustainability goals, making it a preferred choice for environmentally conscious manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and technical disclosures found within the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What represents the primary advantage of the Ruthenium catalyst over the traditional Rhodium catalyst for this synthesis?
A: The Ruthenium-based catalyst system described in CN102381990A offers a significant cost advantage due to the lower price of Ruthenium compared to Rhodium. Furthermore, it achieves high optical purity (ee value >95%) directly without the need for extensive purification steps that typically result in substantial yield loss.
Q: How does the new process improve the overall yield compared to prior art methods?
A: Prior art methods often required purification to boost optical purity, leading to a yield loss of approximately 30%. The novel asymmetric hydrogenation process achieves high conversion (>99%) and high optical purity simultaneously, eliminating the need for yield-reducing purification steps and thereby maximizing the final product output.
Q: What are the typical reaction conditions for this asymmetric hydrogenation?
A: The reaction is typically conducted in an alcoholic solvent like isopropanol, using an alkaline assistant such as potassium tert-butoxide. Hydrogen pressure ranges from 15 to 1500 psi, and temperatures can vary from -20°C to 200°C, with room temperature being highly effective for many catalyst systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-benzyl Phenylephrine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively evaluated the technology described in CN102381990A and possesses the capability to execute this asymmetric hydrogenation with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, to guarantee that every batch meets the highest international standards.
We invite you to collaborate with us to leverage this advanced technology for your specific project requirements. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this efficient route can optimize your budget. Please contact us today to obtain specific COA data and comprehensive route feasibility assessments, and let us partner with you to drive innovation and efficiency in your supply chain.
