Advanced Catalytic Synthesis of 3-Phenoxy-Benzaldehyde for Scalable Agrochemical Manufacturing
Advanced Catalytic Synthesis of 3-Phenoxy-Benzaldehyde for Scalable Agrochemical Manufacturing
The global demand for high-performance pyrethroid pesticides continues to drive the need for efficient, cost-effective synthesis routes for key intermediates like 3-phenoxy-benzaldehyde. Patent CN101337868A introduces a robust methodology that addresses longstanding challenges in producing this critical building block. By utilizing a strategic protection-coupling-deprotection sequence, this technology enables the use of abundant copper catalysts to forge the essential carbon-oxygen bond. For R&D directors and procurement specialists in the agrochemical sector, this patent represents a significant opportunity to optimize manufacturing protocols. The process eliminates the reliance on expensive noble metals and reduces the environmental footprint associated with traditional halogenated solvent systems. As we analyze the technical specifics, it becomes clear that this approach offers a viable pathway for reliable agrochemical intermediate supplier networks seeking to enhance their production capabilities while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 3-phenoxy-benzaldehyde has been plagued by significant inefficiencies and economic burdens. Traditional methods, such as the direct oxidation of m-chlorotoluene or the use of m-cresol, often suffer from complex purification requirements and low overall yields. Specifically, the direct bromination and subsequent substitution routes can generate substantial amounts of hazardous waste and require harsh reaction conditions that degrade product quality. Furthermore, existing processes frequently struggle with selectivity issues, leading to difficult-to-remove impurities that compromise the efficacy of the final pesticide formulations. The reliance on stoichiometric reagents rather than catalytic systems in older methodologies results in inflated raw material costs and increased disposal expenses. These factors collectively hinder the ability of manufacturers to achieve cost reduction in agrochemical intermediates manufacturing, creating a pressing need for technological innovation that can streamline operations without sacrificing purity.
The Novel Approach
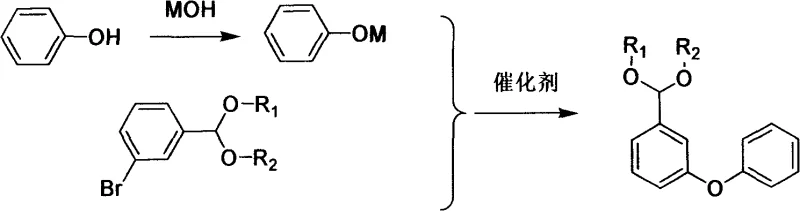
The methodology outlined in the patent data presents a transformative solution by introducing a protective group strategy prior to the critical coupling step. Instead of attempting to couple reactive aldehydes directly under harsh conditions, the process first converts 3-bromobenzaldehyde into a stable acetal derivative. This modification effectively shields the sensitive aldehyde functionality from oxidation or polymerization during the subsequent high-temperature etherification. The core of this innovation lies in the copper-catalyzed reaction between the protected bromide and phenol derivatives, which proceeds with remarkable efficiency. By decoupling the sensitivity of the aldehyde from the rigors of the C-O bond formation, the process achieves superior selectivity and yield. This strategic adjustment not only simplifies the downstream purification but also allows for the use of more economical catalysts, thereby fundamentally altering the cost structure of production. The result is a streamlined workflow that significantly mitigates the generation of three wastes, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Copper-Catalyzed Ullmann Etherification
At the heart of this synthesis lies a classic yet optimized Ullmann-type coupling mechanism, facilitated by earth-abundant copper species. The reaction initiates with the formation of a phenoxide nucleophile, generated in situ by treating phenol with a base such as sodium hydroxide or potassium hydroxide. This nucleophile then engages with the aryl bromide substrate in the presence of the copper catalyst. The catalytic cycle likely involves the oxidative addition of the aryl bromide to the copper center, followed by ligand exchange with the phenoxide and subsequent reductive elimination to form the diaryl ether bond. What distinguishes this specific patent implementation is the tolerance for various copper sources, including cuprous oxide, cuprous chloride, and even active copper powder. This flexibility suggests a robust mechanistic pathway that is not overly sensitive to the precise oxidation state of the initial catalyst precursor, provided the active species is generated under the reaction conditions. The use of polar aprotic solvents like DMF further enhances the solubility of the ionic intermediates, driving the equilibrium towards the desired coupled product.
Crucially, the inclusion of the acetal protecting group plays a pivotal role in impurity control and reaction fidelity. Without this protection, the free aldehyde group would be susceptible to nucleophilic attack by the phenoxide or base, leading to hemiacetal formation or Cannizzaro-type side reactions. Additionally, at the elevated temperatures required for copper catalysis (120-190°C), free aldehydes are prone to thermal degradation and polymerization. By masking the carbonyl as a diethyl acetal or cyclic ethylene acetal, the molecule remains inert to these competing pathways. This ensures that the only major transformation occurring is the desired C-O bond formation. Upon completion of the coupling, mild acid hydrolysis cleanly removes the protecting group, regenerating the aldehyde functionality with high fidelity. This mechanistic safeguard is essential for achieving the high-purity specifications required for pharmaceutical and agrochemical applications, minimizing the burden on downstream chromatographic or crystallization purification steps.
How to Synthesize 3-Phenoxy-Benzaldehyde Efficiently
The practical execution of this synthesis involves a logical three-stage sequence designed for operational simplicity and scalability. First, the condensation step protects the starting material, converting 3-bromobenzaldehyde into a stable acetal using glycols or alcohols under acidic catalysis with water removal. Second, the protected intermediate undergoes etherification with phenol in the presence of a base and copper catalyst at elevated temperatures, forming the core diphenyl ether skeleton. Finally, the protecting group is cleaved via acid hydrolysis to yield the target aldehyde. This modular approach allows for easy monitoring of reaction progress at each stage, ensuring that impurities are managed before they can propagate. For detailed operational parameters and specific molar ratios, please refer to the standardized guide below.
- Perform condensation of 3-bromobenzaldehyde with glycol or alcohol to form a protected acetal derivative, preventing side reactions.
- Execute copper-catalyzed etherification between the protected bromide and sodium phenolate at elevated temperatures (120-190°C).
- Conduct acid hydrolysis to remove the protecting group and isolate the final 3-phenoxy-benzaldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The shift towards a catalytic system using inexpensive copper salts drastically reduces the dependency on precious metal catalysts, which are subject to volatile market pricing and supply constraints. Moreover, the simplified workflow reduces the number of unit operations required, leading to lower energy consumption and reduced labor costs per kilogram of product. The ability to utilize standard industrial reactors without the need for specialized high-pressure equipment further enhances the feasibility of rapid scale-up. These factors combine to create a manufacturing profile that is both economically resilient and operationally agile, ensuring consistent supply continuity even in fluctuating market conditions.
- Cost Reduction in Manufacturing: The utilization of low-loading copper catalysts instead of expensive palladium or nickel systems results in substantial raw material savings. Since the catalyst loading is minimal (0.01 to 0.15 molar ratio), the cost contribution of the metal to the final product is negligible. Furthermore, the high selectivity of the protected route minimizes the loss of valuable starting materials to side products, effectively improving the overall mass balance. The elimination of complex purification steps required to remove heavy metal residues also translates into lower processing costs and reduced solvent usage. These cumulative efficiencies drive down the total cost of goods sold, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 3-bromobenzaldehyde, phenol, and common copper salts, are commodity chemicals with well-established global supply chains. This abundance mitigates the risk of supply disruptions that often plague processes relying on exotic or proprietary reagents. Additionally, the robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, ensuring consistent batch-to-batch performance. By reducing the lead time for high-purity agrochemical intermediates, manufacturers can respond more quickly to downstream demand spikes, thereby strengthening relationships with key clients in the pesticide formulation sector.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex agrochemical intermediates, utilizing standard heating and stirring equipment found in most fine chemical plants. The reduction in "three wastes" (wastewater, waste gas, and solid waste) is a significant compliance advantage, lowering the costs associated with environmental treatment and disposal. The use of recyclable solvents like toluene in the workup phases further supports sustainability goals. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these nuances is critical for process engineers aiming to adapt this technology to their specific production facilities. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance.
Q: What catalysts are suitable for this etherification process?
A: The patent specifies various copper-based catalysts including cuprous oxide, cuprous chloride, neutralized verdigris, or active copper powder, allowing for flexible sourcing.
Q: Why is the aldehyde group protected before coupling?
A: Protecting the aldehyde as an acetal prevents oxidation and unwanted side reactions during the high-temperature etherification step, ensuring higher purity.
Q: What are the typical reaction temperatures for the coupling step?
A: The etherification reaction is typically conducted between 120°C and 190°C, depending on the specific solvent and catalyst system employed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Phenoxy-Benzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global agrochemical value chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. By leveraging technologies such as the copper-catalyzed etherification described above, we can offer cost-effective solutions without compromising on quality or delivery timelines.
We invite potential partners to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate our material against your internal standards, we are ready to assist. Please contact us today to request route feasibility assessments and discover how our manufacturing expertise can drive value for your organization.
