Advanced One-Step Synthesis of 2-Amino-4-Chloro-5-Fluoropyrimidine for Commercial Pharmaceutical Manufacturing
Introduction to Patent CN110343074B Technology
The pharmaceutical industry constantly seeks more efficient pathways to access critical heterocyclic building blocks, and the technology disclosed in patent CN110343074B represents a significant stride in this direction. This patent details a robust synthetic method for 2-amino-4-chloro-5-fluoropyrimidine, a pivotal intermediate utilized extensively in the development of anti-cancer and anti-AIDS therapeutics. By leveraging a direct nucleophilic substitution strategy, the invention circumvents the traditional complexities associated with pyrimidine functionalization. The core innovation lies in the utilization of commercially abundant 2,4-dichloro-5-fluoropyrimidine as the foundational substrate, reacting it directly with aqueous ammonia under strictly controlled thermal conditions. This approach not only simplifies the operational workflow but also addresses critical pain points regarding raw material toxicity and environmental impact that have plagued earlier generations of synthesis protocols. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a stable supply of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated aminopyrimidines has been fraught with challenges that hinder efficient commercial production. Traditional routes often involve multi-step sequences that require harsh reagents, leading to significant safety hazards and the generation of toxic waste streams. Many legacy processes rely on expensive or difficult-to-handle nitrogen sources that necessitate rigorous anhydrous conditions, driving up both capital expenditure and operational costs. Furthermore, controlling regioselectivity in pyrimidine systems is notoriously difficult; without precise kinetic control, manufacturers often face a deluge of unwanted isomers that are chemically similar and energetically costly to separate. These inefficiencies result in prolonged lead times and inconsistent batch quality, creating bottlenecks in the supply chain for downstream API manufacturers who rely on just-in-time delivery models. The cumulative effect of these limitations is a fragile supply network vulnerable to raw material shortages and regulatory scrutiny regarding environmental compliance.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach outlined in the patent introduces a streamlined one-step substitution reaction that fundamentally alters the economic landscape of production. By selecting 2,4-dichloro-5-fluoropyrimidine as the starting material, the process capitalizes on the inherent reactivity differences between the chlorine atoms at the 2 and 4 positions. The method employs aqueous ammonia, a reagent that is not only inexpensive but also significantly safer to handle on a multi-ton scale compared to anhydrous ammonia gas or organic amines. The reaction is conducted in tetrahydrofuran, a solvent that offers excellent solubility for the substrates while allowing for precise temperature modulation. This strategic combination of readily available inputs and mild reaction conditions effectively bypasses the need for complex protection-deprotection strategies. 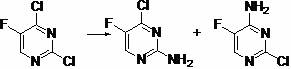
Mechanistic Insights into Nucleophilic Aromatic Substitution
The chemical transformation at the heart of this technology is a classic nucleophilic aromatic substitution (SNAr), yet its execution reveals sophisticated control over reaction kinetics. The mechanism initiates with the attack of the ammonia nucleophile on the electron-deficient pyrimidine ring. Due to the strong electron-withdrawing nature of the ring nitrogens and the fluorine substituent, the ring carbons become susceptible to nucleophilic attack. However, the critical challenge lies in differentiating between the C2 and C4 positions, both of which bear chlorine leaving groups. The patent specifies a low-temperature regime, typically between 0°C and -10°C, during the addition of ammonia. This thermal constraint is not arbitrary; it serves to kinetically favor the displacement of the chlorine at the C4 position or C2 position depending on the specific electronic activation, ultimately yielding a mixture where the desired 2-amino-4-chloro isomer is accessible. Following the initial low-temperature addition, the reaction mixture is allowed to naturally warm to room temperature. This gradual warming provides the necessary activation energy to drive the reaction to completion without triggering excessive side reactions or degradation of the sensitive fluorinated scaffold.
Impurity control is another cornerstone of this mechanistic design, particularly given the formation of a regioisomeric mixture. The process acknowledges that the reaction yields a mixture of 2-chloro-4-amino-5-fluoropyrimidine and the target 2-amino-4-chloro-5-fluoropyrimidine. While this might seem like a drawback, the patent outlines a robust purification protocol involving column chromatography to isolate the target compound with purity exceeding 98%. From a process chemistry perspective, this indicates that while the selectivity is not absolute, the physical properties of the isomers are sufficiently distinct to allow for effective separation. For industrial applications, this chromatographic step would likely be optimized into a crystallization or preparative HPLC process, but the fundamental principle remains: the chemical pathway generates a separable profile rather than an intractable tar. This level of purity is non-negotiable for pharmaceutical intermediates intended for oncology applications, where genotoxic impurities must be rigorously excluded.
How to Synthesize 2-Amino-4-Chloro-5-Fluoropyrimidine Efficiently
Implementing this synthesis requires adherence to specific operational parameters to ensure reproducibility and safety. The protocol begins with the dissolution of the dichloro precursor in anhydrous tetrahydrofuran, followed by a critical cooling phase. Operators must maintain the reaction temperature strictly below 0°C during the dropwise addition of aqueous ammonia to prevent runaway exotherms and minimize isomer formation. Once the addition is complete, the mixture is stirred at low temperature for 2-3 hours before being allowed to warm naturally. The workup involves solvent evaporation and extraction with ethyl acetate, followed by washing with saturated sodium bicarbonate and brine to remove residual acids and salts.
- Dissolve 2,4-dichloro-5-fluoropyrimidine in tetrahydrofuran (THF) and cool the solution to a low temperature range between 0°C and -10°C.
- Dropwise add aqueous ammonia while strictly maintaining the low temperature to control regioselectivity, followed by a reaction period of 2-3 hours.
- Allow the mixture to naturally warm to room temperature for 1-2 hours, then proceed with solvent evaporation, extraction, and chromatographic separation to isolate the target isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the value proposition of this patent extends far beyond the chemistry itself; it translates directly into tangible business advantages. The most immediate benefit is the drastic simplification of the raw material portfolio. By relying on 2,4-dichloro-5-fluoropyrimidine, a commodity chemical produced in vast quantities globally, manufacturers eliminate the risk associated with sourcing exotic or custom-synthesized precursors. This shift to bulk chemicals inherently stabilizes pricing and ensures continuity of supply, even during market fluctuations. Furthermore, the use of aqueous ammonia and recyclable solvents like THF and ethyl acetate aligns perfectly with modern green chemistry initiatives. The ability to recover and reuse solvents significantly reduces the volume of hazardous waste requiring disposal, thereby lowering environmental compliance costs and minimizing the facility's carbon footprint. These factors combine to create a manufacturing process that is not only economically superior but also resilient against regulatory changes.
- Cost Reduction in Manufacturing: The economic model of this synthesis is driven by the elimination of expensive catalysts and the reduction of unit operations. Unlike transition-metal catalyzed couplings that require costly ligands and extensive metal scavenging steps to meet ppm-level residuals, this metal-free ammonolysis avoids these expenses entirely. The simplified post-treatment process, which avoids complex distillations or hazardous quenches, reduces labor hours and energy consumption per kilogram of product. Additionally, the high atom economy of using ammonia as the nitrogen source means less waste is generated per mole of product formed. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, offering a competitive pricing structure for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents. Aqueous ammonia and tetrahydrofuran are standard inventory items for almost any fine chemical manufacturer, removing single-source dependencies. The robustness of the reaction conditions—tolerating a natural warm-up to room temperature—also implies a process that is forgiving of minor operational variances, leading to higher batch success rates. This reliability reduces the need for safety stock and allows for leaner inventory management. For global buyers, this means shorter lead times and a lower risk of shipment delays caused by production failures or raw material shortages.
- Scalability and Environmental Compliance: Scaling this process from the laboratory to multi-ton production is straightforward due to the absence of sensitive reagents. The exotherm is manageable through controlled dosing, and the solvent system is compatible with standard glass-lined or stainless steel reactors. From an environmental standpoint, the avoidance of heavy metals and chlorinated solvents (in the workup) simplifies wastewater treatment. The process generates primarily ammonium salts and organic solvents that can be treated or incinerated with standard protocols. This ease of compliance facilitates faster regulatory approvals and site audits, accelerating the time-to-market for new drug formulations relying on this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific constraints and benefits outlined in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these details is crucial for assessing the feasibility of integrating this intermediate into your existing manufacturing pipeline.
Q: What is the primary advantage of using 2,4-dichloro-5-fluoropyrimidine as a starting material?
A: The primary advantage lies in its commercial availability and cost-effectiveness. As a bulk chemical, it eliminates the need for complex multi-step precursor synthesis, significantly reducing raw material procurement costs and lead times for large-scale production.
Q: How does the process manage the formation of regioisomers during ammonolysis?
A: The process utilizes precise temperature control, maintaining the reaction between -10°C and 0°C during the addition of ammonia. This kinetic control favors the formation of the desired 2-amino-4-chloro isomer over the 4-amino-2-chloro isomer, although chromatographic separation is still employed to ensure high purity specifications.
Q: Is the solvent system suitable for industrial scale-up?
A: Yes, the use of tetrahydrofuran (THF) and ethyl acetate represents a standard, scalable solvent system. These solvents are widely available, easily recyclable through distillation, and compatible with standard stainless steel reactor infrastructure, facilitating a smooth transition from laboratory to commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Chloro-5-Fluoropyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics seen in the lab are faithfully reproduced at an industrial scale. We understand that for critical oncology intermediates, consistency is paramount; therefore, our facilities operate under stringent purity specifications and are supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation. We do not just supply chemicals; we deliver validated processes that guarantee the high-purity 2-amino-4-chloro-5-fluoropyrimidine your drug development programs demand.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this efficient route impacts your bottom line. We encourage potential partners to contact us directly to request specific COA data from our recent batches and to receive comprehensive route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your next-generation pharmaceutical products.
