Scalable Synthesis of Difluorodichlorobenzimide for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry's relentless pursuit of quality and safety places immense pressure on the availability of high-purity reference standards, particularly for complex antibiotic intermediates. Patent CN115819318A introduces a significant advancement in the synthesis of difluorodichlorobenzimide, a critical impurity associated with tetrafluorobenzoic acid production. This compound serves as an essential marker for quality control in the manufacturing of fluoroquinolone antibiotics, including the widely used levofloxacin. The disclosed method offers a streamlined pathway to generate this specific structural analog with exceptional purity, addressing a long-standing challenge in analytical chemistry where authentic impurity standards are often difficult to source or synthesize. By leveraging a direct halogen exchange strategy, this technology provides a reliable foundation for establishing rigorous impurity profiles, thereby supporting global regulatory submissions and ensuring the consistency of active pharmaceutical ingredients.
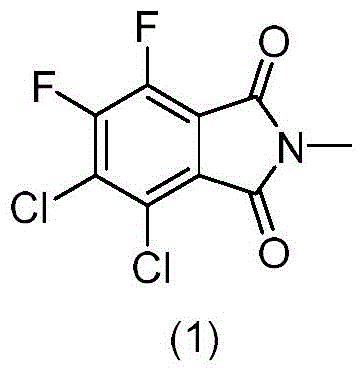
For R&D directors and process chemists, understanding the limitations of conventional impurity isolation versus this novel synthetic approach is vital for method validation. Traditionally, obtaining specific process impurities like difluorodichlorobenzimide involved laborious isolation from actual production batches of tetrafluorobenzoic acid, a process fraught with low yields and inconsistent purity levels. This conventional reliance on isolation often resulted in insufficient quantities for comprehensive toxicological studies or method calibration, creating bottlenecks in drug development timelines. Furthermore, the structural similarity of partial fluorination byproducts made separation via standard crystallization nearly impossible, often requiring repeated recrystallizations that degraded overall recovery rates and introduced solvent residues.
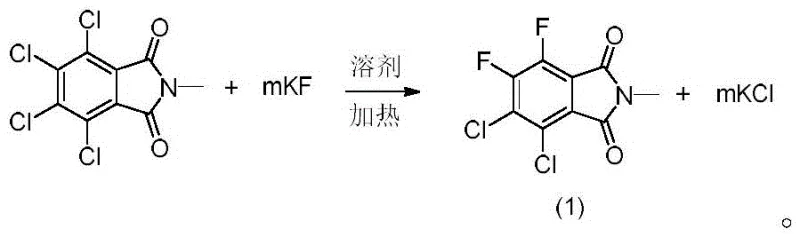
In stark contrast, the novel approach detailed in the patent utilizes a constructive synthetic route starting from N-methyltetrachlorophthalimide. This strategy bypasses the inefficiencies of isolation by building the target molecule directly through controlled nucleophilic substitution. By selecting specific polar aprotic solvents and optimizing fluoride sources, the process achieves selective fluorination, converting specific chloro-substituents while retaining others to match the target impurity structure. This shift from isolation to synthesis represents a paradigm change in reference standard manufacturing, offering predictable scalability and superior control over the final chemical identity. The ability to tune reaction parameters allows for the minimization of over-fluorinated side products, ensuring that the resulting material is fit for purpose as a high-grade analytical standard.
Mechanistic Insights into Fluoride-Mediated Halogen Exchange
The core chemical transformation driving this synthesis is a nucleophilic aromatic substitution (SnAr), where fluoride ions displace chlorine atoms on the electron-deficient phthalimide ring. The success of this reaction hinges on the activation of the fluoride salt within a high-boiling polar aprotic medium. Solvents such as sulfolane, dimethyl sulfoxide (DMSO), or N-methylpyrrolidone (NMP) play a dual role: they solvate the cation of the fluoride salt effectively, increasing the nucleophilicity of the naked fluoride anion, and they withstand the elevated temperatures required to overcome the activation energy of the C-Cl bond cleavage. The electron-withdrawing nature of the imide carbonyl groups further activates the aromatic ring towards nucleophilic attack, facilitating the substitution at the ortho and meta positions relative to the carbonyls.
Controlling the impurity profile during this exchange is a matter of precise stoichiometric and thermal management. The patent highlights that the ratio of fluoride salt to substrate is a critical variable; excessive fluoride can lead to over-fluorination, generating tetrafluoro-byproducts that are structurally similar and difficult to separate. Conversely, insufficient fluoride results in incomplete conversion, leaving mono-fluorinated intermediates. The mechanism suggests a stepwise replacement where the first fluorination activates the ring further for subsequent substitutions, necessitating careful monitoring via TLC or HPLC. Post-reaction, the purification strategy employs a combination of normal phase silica chromatography and reverse-phase preparative HPLC. This orthogonal purification approach exploits differences in polarity and hydrophobicity to separate the target difluoro-dichloro species from both the starting tetrachloro material and potential trifluoro or tetrafluoro contaminants, ensuring the final standard meets the stringent >95% purity requirement.
How to Synthesize Difluorodichlorobenzimide Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this valuable reference standard, emphasizing the importance of solvent selection and temperature control. The process begins with the suspension of N-methyltetrachlorophthalimide in a high-boiling solvent like sulfolane, followed by the addition of anhydrous potassium fluoride. The mixture is heated to temperatures ranging between 150°C and 200°C, a range identified as optimal for driving the reaction to completion without excessive degradation. Detailed standardized synthetic steps for scaling this reaction are provided in the guide below.
- React N-methyltetrachlorophthalimide with potassium fluoride in a polar aprotic solvent like sulfolane at elevated temperatures (150-200°C).
- Perform post-reaction workup involving extraction with organic solvents such as ethyl acetate or dichloromethane.
- Purify the crude product using silica gel column chromatography followed by preparative HPLC to achieve purity greater than 95%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology offers tangible benefits in terms of cost stability and sourcing reliability. Traditional methods of acquiring impurity standards often rely on custom synthesis houses charging premium prices for small-batch, low-yield isolations. By shifting to a direct synthesis model using commodity chemicals like potassium fluoride and bulk solvents, the cost of goods sold for this intermediate is significantly reduced. This reduction in raw material complexity translates to more competitive pricing for downstream pharmaceutical manufacturers who require these standards for routine QC testing, ultimately lowering the overall cost of quality assurance in antibiotic production.
- Cost Reduction in Manufacturing: The elimination of complex isolation procedures and the use of inexpensive, readily available fluoride salts drastically simplify the cost structure. Unlike specialized fluorinating reagents that are hazardous and costly, potassium fluoride is a bulk commodity, ensuring that raw material expenses remain low and predictable. Furthermore, the high conversion efficiency reduces waste disposal costs associated with unreacted starting materials, contributing to a leaner manufacturing process that maximizes resource utilization without compromising on the quality of the final reference standard.
- Enhanced Supply Chain Reliability: Sourcing risks are minimized because the synthesis relies on stable, non-proprietary starting materials that are widely available in the global chemical market. There is no dependency on rare catalysts or fragile reagents that might face supply disruptions. This robustness ensures consistent production schedules and reliable delivery timelines for clients who depend on a steady supply of reference materials for their regulatory filings. The scalability of the reaction from gram to kilogram scales means that supply can be rapidly ramped up to meet sudden increases in demand from the generic pharmaceutical sector.
- Scalability and Environmental Compliance: The process is designed with environmental sustainability in mind, utilizing solvents that can be recovered and recycled, thereby reducing the environmental footprint of the operation. The absence of heavy metal catalysts simplifies waste treatment protocols and ensures compliance with strict environmental regulations regarding effluent discharge. This green chemistry approach not only mitigates regulatory risk but also aligns with the corporate sustainability goals of major pharmaceutical partners, making the supply chain more resilient and future-proof against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of difluorodichlorobenzimide. These insights are derived directly from the patented methodology to ensure accuracy and relevance for industry professionals seeking to understand the capabilities and limitations of this synthesis route.
Q: Why is the synthesis of difluorodichlorobenzimide critical for pharmaceutical quality control?
A: Difluorodichlorobenzimide is a key process-related impurity in the production of tetrafluorobenzoic acid, a crucial intermediate for fluoroquinolone antibiotics like levofloxacin. Having a certified reference standard allows manufacturers to accurately quantify and control this impurity, ensuring regulatory compliance and patient safety.
Q: What are the optimal reaction conditions for this halogen exchange process?
A: The patent indicates that high-temperature conditions between 150°C and 200°C are necessary to drive the nucleophilic substitution. Polar aprotic solvents such as sulfolane, DMSO, or NMP are essential to solubilize the fluoride salt and facilitate the replacement of chlorine atoms with fluorine.
Q: How is high purity (>95%) achieved in the final product?
A: Achieving reference-grade purity requires a two-stage purification strategy. Initial cleanup is performed using silica gel column chromatography with gradient elution, followed by high-resolution separation using preparative HPLC (e.g., C18 columns) to isolate the target compound from isomers and byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluorodichlorobenzimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your analytical data depends on the quality of your reference standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our difluorodichlorobenzimide meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for critical antibiotic intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your R&D and quality control objectives efficiently and reliably.
