Advanced Recycling Strategies for Commercial Scale-up of Complex Oncology Intermediates
The pharmaceutical industry continuously seeks methodologies that enhance the economic viability of producing complex oncology agents, and the synthesis of Tipifarnib represents a prime example of such technological evolution. Patent CN112424149B introduces a groundbreaking approach to preparing the desired enantiomer of 6-[amino(4-chlorophenyl)(1-methyl-1H-imidazol-5-yl)methyl]-4-(3-chlorophenyl)-1-methyl-2(1H)-quinolinone, widely known as Tipifarnib or R115777. This potent farnesyltransferase inhibitor has shown significant promise in clinical development for treating solid tumors and hematological malignancies, making its efficient production a critical supply chain priority. The disclosed invention moves beyond traditional linear synthesis by implementing a cyclic recycling strategy that recovers and reprocesses the unwanted stereoisomer from resolution mother liquors. By transforming the discarded (S)-(-)-enantiomer back into a usable racemic intermediate, the process fundamentally alters the mass balance of the manufacturing workflow. This technical breakthrough not only addresses the inherent inefficiency of classical resolution but also establishes a new standard for resource utilization in the production of high-value chiral pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer drugs like Tipifarnib relied heavily on classical resolution techniques, which inherently suffer from a maximum theoretical yield of fifty percent. As illustrated in the prior art synthesis pathways, the initial formation of the racemic mixture followed by chiral separation results in the isolation of the desired (R)-(+)-enantiomer while the remaining (S)-(-)-enantiomer is typically discarded as waste. This disposal represents a massive economic loss, effectively doubling the raw material cost per unit of active pharmaceutical ingredient produced. Furthermore, the accumulation of chiral waste streams creates significant environmental burdens and complicates waste management protocols for manufacturing facilities. In the specific context of Tipifarnib, early synthetic schemes demonstrated that separating the enantiomers directly from the racemate yielded the target compound in merely 32.5% efficiency, highlighting the severe material attrition associated with non-recycling workflows. Such low yields are unsustainable for commercial-scale operations where cost competitiveness and supply security are paramount concerns for procurement teams.
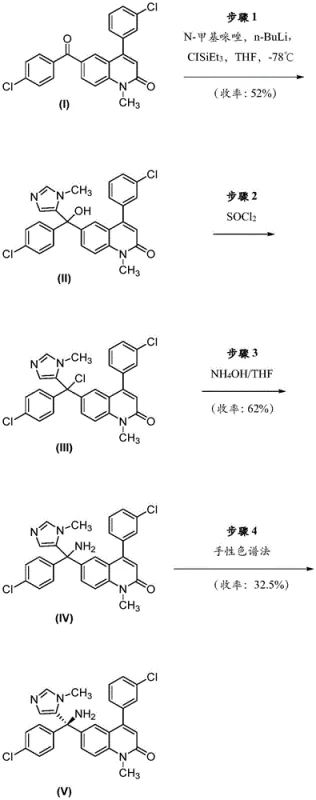
The Novel Approach
In stark contrast to these wasteful legacy methods, the novel approach detailed in the patent utilizes a sophisticated recycling loop that reintegrates the unwanted enantiomer into the production cycle. Instead of discarding the mother liquor containing the enriched (S)-enantiomer salt, the process subjects this stream to a specific chemical transformation that regenerates a racemic precursor. This allows the material to re-enter the synthesis at an earlier stage, effectively bypassing the 50% yield ceiling imposed by traditional resolution. The strategy involves converting the chiral amine salt back into a racemic alcohol intermediate, which can then be reconverted into the racemic amine for subsequent resolution. This closed-loop system ensures that nearly all carbon atoms introduced into the process eventually contribute to the final product, drastically improving the overall atom economy. By salvaging what was previously considered waste, manufacturers can achieve substantially higher throughput without proportionally increasing raw material consumption, thereby creating a more resilient and cost-effective supply chain for this critical oncology intermediate.
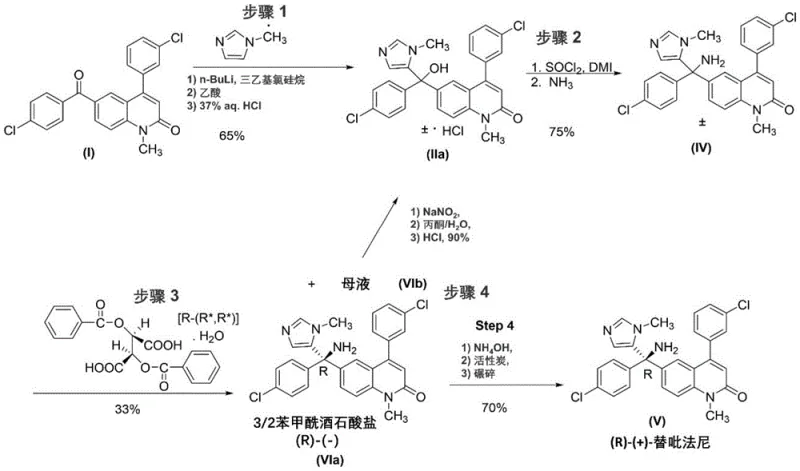
Mechanistic Insights into Sodium Nitrite Mediated Racemization
The chemical heart of this innovative process lies in the diazotization reaction that facilitates the conversion of the chiral amine back to the achiral alcohol. Specifically, the unwanted enantiomer salt, typically isolated as a dibenzoyl-L-tartaric acid salt from the mother liquor, is treated with sodium nitrite in a mixed solvent system comprising water and an organic co-solvent such as acetone or acetonitrile. Under controlled acidic conditions, often utilizing sulfuric or hydrochloric acid, the primary amino group of the Tipifarnib derivative undergoes diazotization to form an unstable diazonium intermediate. This highly reactive species subsequently decomposes, releasing nitrogen gas and generating a carbocation that is rapidly trapped by water to reform the hydroxyl group. Crucially, this substitution reaction proceeds with complete loss of stereochemical integrity at the benzylic center, resulting in the formation of the racemic alcohol intermediate. The patent data indicates that this transformation can be achieved at mild temperatures, such as 20°C, minimizing the formation of thermal degradation byproducts. The ability to execute this racemization efficiently in a homogeneous or biphasic system ensures high conversion rates, with experimental data showing near-quantitative recovery of the alcohol intermediate after extended stirring periods.
Controlling the impurity profile during this recycling step is essential for maintaining the quality of the final API, and the patent provides detailed guidance on managing side reactions. One potential pathway for impurity formation involves the generation of nitroso or other nitrogen-containing byproducts if the reaction conditions are not strictly monitored. However, the optimized protocol specifies precise stoichiometric ratios of sodium nitrite and acid to ensure complete consumption of the amine without excess reagent carryover. Furthermore, the choice of solvent plays a pivotal role in suppressing the formation of specific impurities, such as the nitro-substituted derivative identified as Impurity VII in the experimental data. By adjusting the water content in the solvent mixture, operators can balance the reaction rate against impurity generation, ensuring that the recovered racemic alcohol meets stringent purity specifications before re-entry into the main synthesis line. This level of process control demonstrates that the recycling loop is not merely a theoretical concept but a robust, manufacturable unit operation capable of delivering high-purity intermediates consistently.
How to Synthesize Tipifarnib Efficiently
The implementation of this recycling methodology requires a precise sequence of unit operations designed to maximize recovery while maintaining product quality. The process begins with the standard chiral resolution of the racemic amine using a resolving agent like (-)-dibenzoyl-L-tartaric acid to precipitate the desired enantiomer salt. Following the filtration of the product, the remaining mother liquor, which is enriched with the unwanted enantiomer, becomes the feedstock for the recycling campaign. Through careful pH adjustment and solvent manipulation, the unwanted salt is converted back to the free base and subsequently subjected to the diazotization conditions described previously. The resulting racemic alcohol is then isolated, purified, and chemically transformed back into the racemic amine, ready to be mixed with fresh feedstock for another round of resolution. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures required to execute this cycle, please refer to the standardized synthesis guide below.
- Synthesize racemic Tipifarnib amine and perform chiral resolution using dibenzoyl-L-tartaric acid to isolate the desired (R)-enantiomer salt.
- Collect the mother liquor containing the unwanted (S)-enantiomer salt and treat with sodium nitrite in an aqueous organic solvent system.
- Convert the recovered racemic alcohol intermediate back into the racemic amine mixture for re-entry into the resolution cycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this recycling technology translates directly into tangible strategic benefits that extend beyond simple chemistry metrics. The primary advantage is the drastic reduction in raw material costs, as the effective yield of the process is no longer capped by the stoichiometry of the resolution step. By recovering and reusing the unwanted enantiomer, the consumption of expensive starting materials and chiral resolving agents is significantly minimized, leading to a lower cost of goods sold. This efficiency gain is particularly valuable for high-volume commercial production, where even marginal improvements in yield can result in substantial financial savings over the lifecycle of the product. Additionally, the process reduces the volume of hazardous chemical waste generated, simplifying compliance with environmental regulations and lowering waste disposal expenses. These factors combined create a more competitive pricing structure for the final API intermediate, allowing suppliers to offer more attractive terms to their pharmaceutical partners.
- Cost Reduction in Manufacturing: The elimination of the 50% yield penalty associated with traditional resolution fundamentally changes the economics of Tipifarnib production. By converting waste streams into valuable feedstock, the process reduces the dependency on fresh raw materials, effectively lowering the variable cost per kilogram of product. This material efficiency means that manufacturers can absorb fluctuations in raw material pricing more effectively, providing greater price stability for long-term supply agreements. Furthermore, the reduced need for waste treatment and disposal lowers the operational overhead associated with environmental compliance, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Implementing a recycling loop increases the overall throughput capacity of a manufacturing facility without requiring proportional capital investment in new reactors or equipment. Since the same batch of material can be cycled through the resolution step multiple times, the effective output of the plant is amplified, ensuring a more secure and consistent supply of the intermediate. This resilience is critical for meeting the demanding delivery schedules of global pharmaceutical clients, particularly during periods of high market demand or raw material scarcity. The robustness of the chemistry, demonstrated at kilogram scales, further assures buyers that the supply source is technically mature and capable of sustaining commercial volumes.
- Scalability and Environmental Compliance: The process utilizes common, industrially available solvents such as acetone, acetonitrile, and isopropyl acetate, which facilitates easy scale-up from pilot plants to multi-ton production suites. The avoidance of exotic reagents or complex chromatographic separations simplifies the engineering requirements, making the technology accessible to a wide range of contract development and manufacturing organizations. Moreover, the significant reduction in chemical waste aligns with modern green chemistry principles, helping pharmaceutical companies meet their sustainability goals. This environmental stewardship enhances the brand reputation of both the supplier and the end-product manufacturer, appealing to increasingly eco-conscious stakeholders in the healthcare sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recycling technology. They are derived from the specific experimental data and process descriptions found within the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is crucial for technical teams evaluating the transferability of this process to their own manufacturing sites.
Q: How does the recycling process improve the overall yield of Tipifarnib?
A: Traditional resolution methods discard the unwanted (S)-enantiomer, limiting theoretical yield to 50%. This patented process converts the unwanted enantiomer back into a racemic intermediate, allowing it to be re-processed, thereby significantly boosting overall material efficiency and reducing raw material costs.
Q: What is the critical chemical transformation in the recycling loop?
A: The core innovation involves the diazotization of the chiral amine salt using sodium nitrite under controlled acidic conditions. This reaction effectively removes the chiral amino group and regenerates the achiral alcohol intermediate, erasing the stereochemical information and allowing for re-synthesis.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent data demonstrates successful execution at the kilogram scale (1.445 kg demo batch) with high purity (97.10%) and minimal impurity formation. The use of common solvents like acetone and acetonitrile ensures that the process is robust and easily transferable to multi-ton manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tipifarnib Supplier
The technical innovations described in patent CN112424149B underscore the complexity and sophistication required to produce high-quality oncology intermediates efficiently. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring such advanced processes to life. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications that ensure every batch of Tipifarnib intermediate meets the highest global regulatory standards. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and our team is dedicated to delivering products that facilitate your clinical and commercial success.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized recycling processes can reduce your overall procurement costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your supply chain requirements, ensuring a partnership built on transparency, quality, and mutual growth.
