Advanced Decarboxylative Selenylation for High-Purity Aryl Methyl Selenide Manufacturing
Advanced Decarboxylative Selenylation for High-Purity Aryl Methyl Selenide Manufacturing
The synthesis of organoselenium compounds represents a critical frontier in modern medicinal chemistry and agrochemical development, driven by the unique biological activities of the selenium atom. From the anti-inflammatory properties of Ebselen to the antioxidant capabilities found in glutathione peroxidase mimics, aryl methyl selenides serve as indispensable building blocks for next-generation therapeutics. However, traditional synthetic routes have long been plagued by the use of malodorous, toxic reagents and harsh reaction conditions that complicate scale-up. A groundbreaking solution to these challenges is detailed in Chinese Patent CN111620800B, which discloses a novel method for preparing aryl methyl selenide compounds via the decarboxylative selenylation of aromatic carboxylic acids. This technology leverages a synergistic catalytic system involving transition metal copper, silver salts, and specific ligands to transform cheap, readily available carboxylic acids into high-value selenium intermediates. By utilizing odorless and stable selenomethyl bunte salts as the selenium source, this process not only mitigates severe safety hazards associated with volatile selenium reagents but also opens new avenues for the efficient construction of C-Se bonds under oxidative conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl methyl selenide motifs has relied on methodologies that are increasingly incompatible with modern green chemistry standards and industrial safety protocols. Early approaches, such as those described by Edward S. Lewis et al. in 1985, utilized nucleophilic substitution reactions involving pre-functionalized selenide species. As illustrated in the reaction scheme below, these methods often required the preparation of unstable intermediates and suffered from limited substrate scope due to the sensitivity of the reacting species.
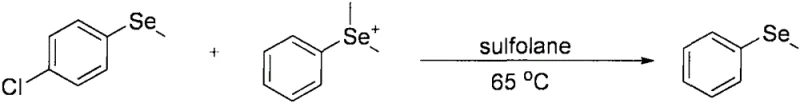
Furthermore, alternative routes reported by researchers like Tadao Yoshida involved the use of nickel complexes and highly toxic benzene as a solvent, creating significant environmental and occupational health liabilities. Another common strategy involved the reaction of diaryldiselenides with iodomethane under strongly basic conditions using sodium hydride, as shown in the following equation. While effective for simple substrates, this approach is notoriously intolerant of base-sensitive functional groups and generates stoichiometric amounts of waste salts, making it economically unviable for large-scale production.
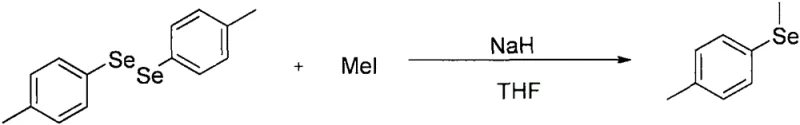
The Novel Approach
In stark contrast to these legacy methods, the technology protected under Patent CN111620800B introduces a paradigm shift by employing aromatic carboxylic acids as the aryl source. This strategy capitalizes on the vast availability and low cost of benzoic acid derivatives, bypassing the need for pre-halogenated arenes or organometallic precursors. The core innovation lies in the use of selenomethyl bunte salt, a crystalline, odorless solid that serves as a safe and stable methylselenol equivalent. Under the cooperative catalysis of a copper-silver dual metal system, this reagent undergoes a smooth decarboxylative coupling with the carboxylic acid substrate. The general reaction pathway, depicted below, highlights the elegance of this transformation, where oxygen acts as the terminal oxidant to regenerate the active catalytic species, ensuring high atom economy and minimal waste generation.

Mechanistic Insights into Cu/Ag Synergistic Catalysis
The success of this decarboxylative selenylation hinges on a finely tuned catalytic cycle involving Copper(I) Iodide (CuI) and Silver Carbonate (Ag2CO3). Mechanistic studies suggest that the copper catalyst initially coordinates with the selenomethyl bunte salt, facilitating the transfer of the methylseleno group. Simultaneously, the silver salt plays a dual role: it acts as an oxidant to maintain the copper in its active oxidation state and potentially assists in the decarboxylation step by interacting with the carboxylate anion. The choice of ligand is equally critical; 1,10-phenanthroline has been identified as the optimal ligand, likely due to its ability to stabilize the copper center and modulate its redox potential, thereby preventing catalyst deactivation. Without this specific nitrogen-based ligand, the reaction fails to proceed, underscoring the necessity of precise electronic tuning around the metal center.
Impurity control in this system is achieved through the selective nature of the radical or organometallic intermediates generated during the cycle. The use of Cesium Carbonate (Cs2CO3) as the base is particularly advantageous for maintaining a clean reaction profile. Unlike weaker bases that might lead to incomplete conversion or side reactions such as esterification, Cs2CO3 ensures rapid and complete deprotonation of the carboxylic acid in polar aprotic solvents like DMF. This rapid activation minimizes the lifetime of reactive intermediates that could otherwise dimerize or decompose, resulting in a product stream with high purity that requires minimal downstream purification. The tolerance for diverse functional groups, including nitro, trifluoromethyl, and ketone moieties, further demonstrates the robustness of the catalytic manifold against competing reduction or oxidation pathways.
How to Synthesize Aryl Methyl Selenide Efficiently
Implementing this synthesis route requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and reproducibility. The process involves charging a reaction vessel with the aromatic carboxylic acid, four equivalents of selenomethyl bunte salt, and the specific catalytic cocktail of CuI, 1,10-phenanthroline, Ag2CO3, and Cs2CO3 in DMF. The mixture is then subjected to an oxygen atmosphere and heated to temperatures between 120°C and 140°C. Detailed standard operating procedures regarding reagent addition order, oxygen flow rates, and workup protocols are essential for scaling this chemistry from milligram to metric ton quantities.
- Combine aromatic carboxylic acid, selenomethyl bunte salt, CuI catalyst, 1,10-phenanthroline ligand, silver carbonate, and cesium carbonate in DMF solvent.
- Purge the reaction vessel with oxygen and heat the mixture to 120-140°C for approximately 24 hours under stirring.
- Upon completion, cool the mixture, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography to isolate the target selenide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this decarboxylative selenylation technology offers transformative benefits that extend far beyond simple yield improvements. The primary value driver is the substitution of hazardous, volatile selenium reagents with stable, solid selenomethyl bunte salts. This change drastically reduces the complexity of raw material handling, storage, and transportation, eliminating the need for specialized containment systems required for malodorous liquids like dimethyl diselenide. Consequently, this leads to significant cost reductions in facility maintenance and safety compliance, allowing for more flexible manufacturing site selection and lower insurance premiums associated with hazardous chemical processing.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the use of aromatic carboxylic acids, which are commodity chemicals available at a fraction of the cost of aryl halides or organolithium reagents typically used in selenium chemistry. Furthermore, the catalytic nature of the copper system means that expensive transition metals are used in sub-stoichiometric amounts, reducing the heavy metal load in the final product and minimizing the cost of metal scavenging steps. The elimination of cryogenic conditions or inert gas blankets (other than oxygen) further simplifies the energy profile of the reaction, contributing to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: Relying on bench-stable solids for both the selenium source and the base components creates a more resilient supply chain. Unlike air-sensitive reagents that degrade upon exposure and require cold-chain logistics, the reagents in this protocol can be stored under ambient conditions for extended periods without loss of potency. This stability reduces the risk of production delays caused by spoiled raw materials and allows manufacturers to maintain leaner inventory levels while ensuring continuous production capability, a critical factor for meeting the just-in-time delivery demands of the pharmaceutical industry.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles by using oxygen as a benign oxidant and generating water and carbon dioxide as the primary byproducts of the decarboxylation. The absence of stoichiometric heavy metal waste and toxic organic solvents like benzene simplifies wastewater treatment and waste disposal protocols. This streamlined waste profile not only lowers disposal costs but also facilitates regulatory approval for commercial scale-up, enabling faster time-to-market for new drug candidates requiring these selenium-containing intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on catalyst selection, substrate compatibility, and process robustness for potential licensees and manufacturing partners.
Q: What are the advantages of using Selenomethyl Bunte Salt over traditional selenium reagents?
A: Selenomethyl Bunte Salt is odorless and stable at room temperature, unlike volatile and toxic dimethyl diselenide or malodorous selenols. This significantly improves workplace safety and simplifies handling protocols during large-scale manufacturing.
Q: Why is Cesium Carbonate preferred as the base in this reaction system?
A: Experimental data indicates that Cesium Carbonate provides superior solubility and dissociation in polar aprotic solvents like DMF compared to potassium or sodium carbonates. This enhances the deprotonation efficiency required for the decarboxylation step, leading to higher conversion rates.
Q: Can this method tolerate sensitive functional groups like nitro or ketone moieties?
A: Yes, the mild oxidative conditions and the specific copper catalytic system demonstrate excellent functional group tolerance. Substrates containing nitro groups, trifluoromethyl groups, and ketones have been successfully converted without significant side reactions or reduction of the sensitive groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Methyl Selenide Supplier
The technological advancements detailed in Patent CN111620800B represent a significant leap forward in the synthesis of organoselenium compounds, offering a safer, cleaner, and more cost-effective pathway to high-value intermediates. At NINGBO INNO PHARMCHEM, we recognize the strategic importance of such innovations for our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of selenium chemistry, ensuring stringent purity specifications and rigorous QC labs are in place to deliver materials that meet the exacting standards of the pharmaceutical and agrochemical sectors.
We invite R&D directors and procurement specialists to explore how this decarboxylative selenylation technology can optimize your supply chain and reduce overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next breakthrough in drug discovery and development.
