Advanced Manufacturing of Rizatriptan Benzoate via Novel Indoline Dehydrogenation Strategy
Advanced Manufacturing of Rizatriptan Benzoate via Novel Indoline Dehydrogenation Strategy
The pharmaceutical landscape for anti-migraine treatments continues to evolve, driven by the demand for higher purity active pharmaceutical ingredients (APIs) and more efficient manufacturing processes. Patent CN103664901A introduces a groundbreaking methodology for the synthesis of Rizatriptan Benzoate, a potent 5-HT1B/1D receptor agonist. Unlike conventional pathways that rely on the problematic Fisher Indole Synthesis, this innovation leverages a strategic oxidative dehydrogenation of an indoline precursor. This approach not only simplifies the operational workflow but also drastically improves the impurity profile of the final product. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding this shift away from legacy chemistry is critical for securing supply chains that meet stringent regulatory standards.
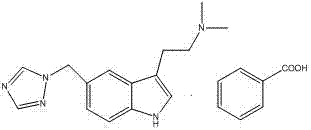
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Rizatriptan Benzoate has been hindered by the inherent flaws of the Fisher Indole Synthesis, as detailed in earlier patents like EP497512. This traditional route typically involves the condensation of hydrazine derivatives with ketones or aldehydes under acidic conditions to close the indole ring. However, this reaction is notorious for its lack of regioselectivity and the formation of complex polymeric by-products that are chemically similar to the target molecule. Consequently, manufacturers are forced to employ resource-intensive purification techniques, such as repeated column chromatography, to achieve acceptable purity levels. Furthermore, subsequent methylation steps often require expensive and toxic reducing agents like sodium cyanoborohydride, which complicates waste management and increases the overall cost of goods sold (COGS).
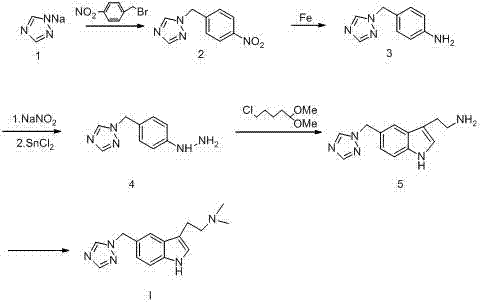
The Novel Approach
In stark contrast, the methodology disclosed in CN103664901A constructs the indole scaffold through a controlled dehydrogenation of a pre-functionalized indoline ring. By starting with indoline and performing a triazole methylation followed by a catalytic oxidation, the process achieves a highly selective formation of the 5-substituted indole core without generating the tarry residues typical of Fisher cyclizations. This strategic pivot allows for the use of standard, cost-effective reagents and eliminates the need for chromatographic purification in the critical ring-closing stage. The result is a streamlined five-step sequence that offers superior scalability and aligns perfectly with the industry's push for cost reduction in API manufacturing while maintaining exceptional product quality.
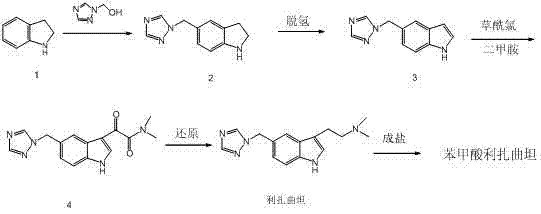
Mechanistic Insights into Oxidative Dehydrogenation of Indoline
The cornerstone of this novel synthesis is the oxidative dehydrogenation step, which transforms the saturated indoline ring into the aromatic indole system. This transformation is mediated by a specialized heterogeneous catalyst system, specifically 5% dicyanobenzene palladium dichloride supported on hydroxyapatite. Mechanistically, the palladium species facilitates the removal of hydrogen atoms from the 2 and 3 positions of the indoline ring, restoring aromaticity. The use of a supported catalyst is particularly advantageous for process chemistry, as it allows for the easy separation of the metal species from the reaction mixture via simple filtration. This not only ensures that the final product meets rigorous heavy metal specifications but also enables the recovery and reuse of the expensive palladium catalyst, thereby enhancing the economic viability of the process for commercial scale-up of complex pharmaceutical intermediates.
From an impurity control perspective, this mechanism offers a distinct advantage over acid-catalyzed cyclizations. The dehydrogenation pathway is clean and predictable, minimizing the formation of regioisomers or polymeric side products that often plague Fisher syntheses. The patent data indicates that this step can proceed with quantitative efficiency, achieving yields as high as 98.8% under optimized conditions (110°C in toluene). Such high conversion rates reduce the burden on downstream purification units and ensure a consistent supply of high-purity pharmaceutical intermediates. By avoiding the harsh acidic environments and high temperatures associated with traditional indole formation, the process also preserves the integrity of the sensitive triazole moiety, preventing degradation and ensuring a robust overall yield.
How to Synthesize Rizatriptan Benzoate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters, particularly during the acylation and reduction phases. The process begins with the methylation of indoline using triazole methanol in glacial acetic acid, followed by the critical dehydrogenation step described previously. Subsequent functionalization involves the introduction of the ethylamine side chain via an acylation-reduction sequence. Specifically, the indole intermediate is reacted with oxalyl chloride to form an acid chloride species, which is then treated with dimethylamine gas to generate the amide. This amide is subsequently reduced using lithium aluminum hydride to yield the free base rizatriptan. Finally, salt formation with benzoic acid in ethanol provides the stable crystalline API. For detailed operational parameters and safety protocols, please refer to the standardized guide below.
- React indoline with triazole methanol in glacial acetic acid at 80°C to form 5-((1H-1,2,4-triazol-1-yl)methyl)indoline.
- Perform oxidative dehydrogenation using 5% Pd(dcb)2/hydroxyapatite catalyst in toluene at 110°C to construct the indole ring.
- Acylate the indole intermediate with oxalyl chloride followed by dimethylamine gas to introduce the N,N-dimethylacetamide side chain.
- Reduce the amide intermediate using lithium aluminum hydride in anhydrous THF to yield free base rizatriptan.
- Form the final salt by reacting free base rizatriptan with benzoic acid in ethanol to obtain rizatriptan benzoate.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this novel synthetic route presents significant strategic benefits beyond mere technical elegance. By eliminating the reliance on the Fisher Indole Synthesis, manufacturers can bypass the bottlenecks associated with difficult purifications and low-yielding cyclization steps. This translates directly into a more resilient supply chain capable of meeting fluctuating market demands for anti-migraine medications. The use of reusable heterogeneous catalysts and common organic solvents further stabilizes the raw material supply, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risks associated with sourcing specialized or hazardous reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is substantial, primarily driven by the elimination of expensive reagents like sodium cyanoborohydride and the reduction of solvent consumption associated with chromatographic purifications. The ability to recover and reuse the palladium catalyst significantly lowers the variable cost per kilogram of production. Additionally, the high yields observed in the dehydrogenation and salt formation steps (exceeding 98% in pilot examples) minimize material waste, contributing to a leaner and more cost-effective manufacturing operation that maximizes return on investment.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway enhances supply security by relying on widely available starting materials such as indoline and benzoic acid. Unlike routes dependent on unstable diazonium salts or sensitive hydrazine intermediates, the indoline-based approach offers greater operational stability and shelf-life for intermediates. This reliability ensures consistent batch-to-batch quality and reduces the likelihood of production delays caused by raw material shortages or failed reaction batches, securing a steady flow of product to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this route is inherently greener and easier to scale. The avoidance of heavy metal contaminants in the final product simplifies the validation process for regulatory filings. Furthermore, the reduction in hazardous waste generation, particularly from chromatography columns and toxic reducing agents, aligns with modern sustainability goals. The process conditions are mild and manageable in standard stainless steel reactors, facilitating a smooth transition from laboratory scale to multi-ton commercial production without the need for exotic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Rizatriptan Benzoate using this advanced indoline-based methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process feasibility and quality assurance.
Q: Why is the Fisher Indole Synthesis avoided in this new Rizatriptan Benzoate process?
A: The traditional Fisher Indole Synthesis suffers from low regioselectivity, low yields, and the generation of significant polymeric by-products that are difficult to remove, often requiring extensive chromatographic purification. The novel indoline dehydrogenation route completely bypasses these issues, offering a cleaner reaction profile and higher product purity suitable for industrial scale-up.
Q: What is the key catalytic advantage in the indole ring construction step?
A: The process utilizes a heterogeneous catalyst system, specifically 5% dicyanobenzene palladium dichloride supported on hydroxyapatite. This catalyst enables quantitative conversion (up to 98.8% yield in examples) under mild conditions and, crucially, can be recovered by filtration and reused, significantly reducing catalyst costs and metal residue concerns in the final API.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability. It avoids expensive reagents like sodium cyanoborohydride used in older methods and eliminates the need for chromatographic purification in the critical ring-closing and side-chain addition steps. The use of common solvents like toluene, ethanol, and dichloromethane, along with robust reaction conditions, supports reliable commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rizatriptan Benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of the indoline dehydrogenation route are fully realized in practical manufacturing. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Rizatriptan Benzoate meets the highest international standards, providing our partners with the confidence needed to navigate complex regulatory landscapes.
We invite forward-thinking pharmaceutical companies to collaborate with us on optimizing their supply chains for anti-migraine therapeutics. By leveraging our expertise in process development and scale-up, we can help you achieve significant efficiencies and cost savings. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and comprehensive route feasibility assessments tailored to your project requirements.
