Advanced Solid Acid Catalysis for Commercial Scale-up of High-Purity Hydroxybenzomorph
Advanced Solid Acid Catalysis for Commercial Scale-up of High-Purity Hydroxybenzomorph
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more sustainable catalytic processes, a transition vividly exemplified by the technological breakthroughs detailed in patent CN111170958A. This pivotal intellectual property introduces a novel preparation method for hydroxybenzomorph (also known as hydroxyphenylmorpholine), a critical heterocyclic building block extensively utilized in the formulation of hair dyes and as a versatile intermediate in pharmaceutical synthesis. Unlike traditional synthetic routes that rely on hazardous reagents and generate substantial toxic byproducts, this innovation leverages the power of fibrous solid superacids to achieve demethylation and cyclization in a purely aqueous medium. For R&D directors and procurement strategists alike, the implications are profound: a pathway that not only enhances product purity to levels exceeding 99% but also fundamentally alters the cost structure and environmental footprint of production. By replacing corrosive liquid acids and dangerous reducing agents with a recoverable solid catalyst, this technology addresses the twin challenges of regulatory compliance and economic efficiency that define modern chemical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxybenzomorph has been plagued by significant technical and safety hurdles that render many legacy processes unsuitable for modern, large-scale industrial application. One prominent prior art route involves the cyclization of 2-amino hydroquinone with 2-chloroacetyl chloride followed by a reduction step using lithium aluminum hydride (LiAlH4). As illustrated in the reaction scheme below, this pathway is fraught with peril; LiAlH4 is notoriously pyrophoric and moisture-sensitive, requiring stringent anhydrous conditions and specialized handling equipment that drive up capital expenditure and operational risk.
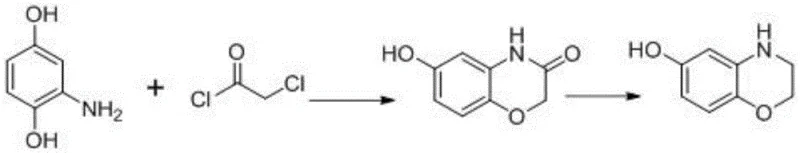
Furthermore, alternative methods utilizing 1,2-dibromoethane for cyclization suffer from poor regioselectivity and low yields due to the presence of multiple reactive sites on the aromatic ring, leading to complex impurity profiles that are difficult and costly to purge. Perhaps the most commercially prevalent yet environmentally damaging method employs 62% hydrobromic acid for demethylation. While chemically effective, this liquid-phase process generates approximately 2 tons of methyl bromide gas—a potent neurotoxin—for every ton of product manufactured. Additionally, the post-treatment requires neutralization with ammonia water, resulting in vast quantities of high-salt, high-ammonia nitrogen wastewater that imposes a heavy burden on effluent treatment facilities and violates increasingly strict environmental discharge standards.
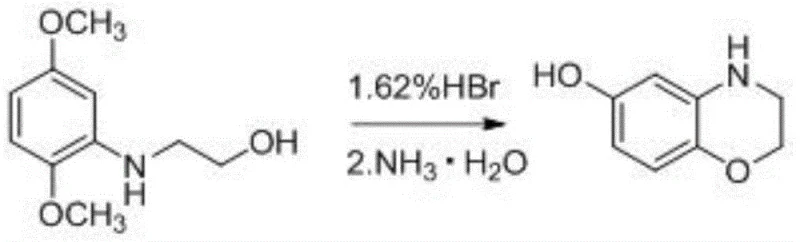
The Novel Approach
In stark contrast to these hazardous legacy techniques, the methodology disclosed in CN111170958A represents a leap forward in process chemistry by utilizing a heterogeneous solid superacid catalyst, specifically variants of sulfated zirconia supported on fibrous oxides (e.g., SO4 2-/ZrO2-MnO2). This approach allows the reaction to proceed in water, the greenest of all solvents, eliminating the need for volatile organic compounds (VOCs) and corrosive mineral acids. The solid acid acts as a robust promoter for both the cleavage of the methoxy groups and the subsequent intramolecular cyclization, achieving yields between 65% and 90% with exceptional purity. Crucially, the heterogeneous nature of the catalyst allows for its physical separation from the reaction mixture via simple hot filtration. This capability enables the catalyst to be recovered and recycled multiple times, transforming a consumable cost center into a reusable asset and dramatically lowering the variable cost per kilogram of the final API intermediate.
Mechanistic Insights into Solid Superacid-Catalyzed Cyclization
The efficacy of this novel synthesis lies in the unique electronic and structural properties of the fibrous solid superacid catalyst. These materials, typically prepared by loading metal nitrates onto collagen fibers followed by high-temperature calcination and sulfation, possess a high density of strong Lewis and Brønsted acid sites on their surface. In the aqueous reaction medium at elevated temperatures (around 110°C), these acid sites facilitate the protonation of the ether oxygen in the methoxy groups of the starting material, 2-((2,5-dimethoxyphenyl)amino)ethanol. This protonation weakens the carbon-oxygen bond, promoting nucleophilic attack by water molecules to effect demethylation, releasing methanol rather than toxic methyl bromide. Simultaneously, the acidic environment activates the phenolic hydroxyl groups and the amine functionality, driving the thermodynamic equilibrium towards the formation of the stable morpholine ring through an intramolecular nucleophilic substitution. The fibrous morphology of the catalyst support further enhances mass transfer, ensuring that the reactants have ample access to the active sites while preventing the pore blockage often seen in granular catalysts.
From an impurity control perspective, this mechanism offers superior selectivity compared to homogeneous acid catalysis. The rigid structure of the solid acid limits the formation of polymeric byproducts and over-alkylation species that frequently contaminate batches produced with liquid HBr. Moreover, because the reaction does not generate gaseous methyl bromide, there is no risk of re-alkylation side reactions that can occur when methylating agents are present in the headspace of the reactor. The result is a crude product stream that is significantly cleaner, reducing the load on downstream purification steps such as recrystallization. This inherent purity is critical for pharmaceutical applications where strict limits on genotoxic impurities and residual solvents must be met, thereby streamlining the regulatory filing process for drug developers relying on this intermediate.
How to Synthesize Hydroxybenzomorph Efficiently
Implementing this advanced synthetic route requires precise control over catalyst preparation and reaction parameters to maximize yield and catalyst longevity. The process begins with the meticulous synthesis of the fibrous solid acid, involving the coordination of zirconium and transition metal ions (such as Mn, Ni, or Fe) onto a collagen fiber matrix, followed by thermal treatment to create the active oxide phase. Once the catalyst is prepared, the core transformation involves suspending the catalyst in water with the dimethoxy precursor and heating the mixture under reflux conditions. The simplicity of the workup—filtering hot to recover the catalyst and cooling to crystallize the product—makes this method highly attractive for plant operators seeking to minimize unit operations. For a detailed breakdown of the specific temperatures, stirring rates, and molar ratios required to replicate this high-efficiency process, please refer to the standardized technical guide below.
- Prepare the fibrous solid acid catalyst (e.g., SO4 2-/ZrO2-MnO2) by loading metal nitrates onto collagen fibers followed by calcination and sulfation.
- Mix 2-((2,5-dimethoxyphenyl)amino)ethanol with the solid acid catalyst in water and heat to 110°C for 4 hours to effect demethylation and cyclization.
- Filter the mixture while hot to recover the catalyst, then cool the filtrate to crystallize and isolate the high-purity hydroxybenzomorph product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid-acid catalyzed process translates directly into tangible strategic advantages regarding cost stability and operational resilience. The elimination of hazardous reagents like LiAlH4 and concentrated HBr removes significant safety liabilities and the associated insurance and compliance costs that often inflate the price of specialty chemicals. Furthermore, the ability to recycle the solid acid catalyst creates a closed-loop system for one of the most expensive input materials, decoupling production costs from the volatility of the bulk acid market. This process intensification leads to a drastic simplification of the manufacturing workflow, removing the need for complex gas scrubbing systems required to handle methyl bromide emissions and reducing the volume of wastewater that requires expensive biological or chemical treatment before discharge.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the substitution of stoichiometric, single-use reagents with a catalytic, reusable system. By avoiding the purchase of large quantities of hydrobromic acid and the subsequent costs associated with neutralizing and disposing of the resulting saline waste, manufacturers can achieve substantial reductions in variable production costs. Additionally, the use of water as the sole solvent eliminates the procurement, storage, and recovery costs linked to organic solvents, further enhancing the overall margin profile of the hydroxybenzomorph supply chain.
- Enhanced Supply Chain Reliability: Relying on a process that utilizes robust, non-hazardous raw materials significantly de-risks the supply chain. Traditional routes dependent on regulated substances like methyl bromide precursors or pyrophoric reducing agents are susceptible to sudden regulatory bans or transportation restrictions that can halt production indefinitely. In contrast, the solid acid catalyst and benign starting materials used in this novel method are readily available and easy to transport, ensuring consistent production schedules and reliable delivery timelines for downstream pharmaceutical and cosmetic clients.
- Scalability and Environmental Compliance: The transition from batch processes involving toxic gas evolution to a contained, aqueous heterogeneous catalysis system greatly facilitates scale-up from pilot plant to multi-ton commercial production. The absence of high-pressure gas generation and the use of standard stainless steel reactors compatible with mild acidic conditions lower the barrier to entry for contract manufacturing organizations. Moreover, the dramatic reduction in "three wastes" (waste gas, wastewater, and solid residue) ensures that production facilities remain in full compliance with increasingly rigorous global environmental regulations, safeguarding the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply networks.
Q: What are the primary safety advantages of the solid acid method over traditional HBr demethylation?
A: The traditional method using 62% hydrobromic acid generates approximately 2 tons of methyl bromide gas per ton of product, which is a potent nerve agent and environmental hazard. The novel solid acid method operates in water without generating toxic methyl bromide gas, significantly improving workplace safety and reducing废气 (waste gas) treatment costs.
Q: Can the solid acid catalyst be reused in industrial production?
A: Yes, a key feature of this process is the recoverability of the catalyst. The solid acid can be filtered off while the solution is hot, washed, and reused in subsequent batches. This contrasts sharply with homogeneous acid catalysts which are consumed or require complex neutralization, thereby drastically lowering raw material consumption.
Q: How does this method impact wastewater treatment compared to prior art?
A: Conventional routes often generate high-salt and high-ammonia nitrogen wastewater due to the use of ammonia water for pH adjustment and neutralization. This new aqueous process avoids the generation of such difficult-to-treat wastewater streams, simplifying the effluent treatment process and ensuring better environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxybenzomorph Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just an environmental imperative but a commercial necessity for maintaining competitiveness in the global fine chemicals market. Our team of expert process chemists has extensively analyzed the potential of solid-acid catalyzed pathways, such as the one described in CN111170958A, and possesses the technical acumen to adapt and optimize these methodologies for your specific production needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity specifications and rigorous QC labs standards required by the pharmaceutical industry are consistently met. Our commitment to sustainability aligns perfectly with this technology, allowing us to offer a supply of hydroxybenzomorph that is both economically advantageous and environmentally responsible.
We invite you to engage with our technical procurement team to discuss how we can tailor this advanced synthesis route to your volume requirements. By leveraging our expertise in heterogeneous catalysis and green chemistry, we can provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your specific project. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative approach to hydroxybenzomorph manufacturing can become a cornerstone of your supply chain strategy.
