Scalable One-Pot Synthesis of Pivmecillinam for Global Pharmaceutical Supply Chains
Scalable One-Pot Synthesis of Pivmecillinam for Global Pharmaceutical Supply Chains
The pharmaceutical industry constantly seeks more efficient pathways for producing critical antibiotics, and the recent disclosure in patent CN114573603A presents a transformative approach to manufacturing Pivmecillinam. This oral broad-spectrum semi-synthetic penicillin is vital for treating urinary tract infections caused by sensitive bacteria such as Escherichia coli and Klebsiella. The patented method introduces a streamlined one-pot process that converts 6-aminopenicillanic acid (6-APA) directly into the final active pharmaceutical ingredient through sequential oxygen alkylation and nitrogen imidization. By integrating these steps into a single reactor vessel, the technology addresses longstanding inefficiencies in beta-lactam modification, offering a route that is not only chemically elegant but also commercially robust for high-volume production environments.
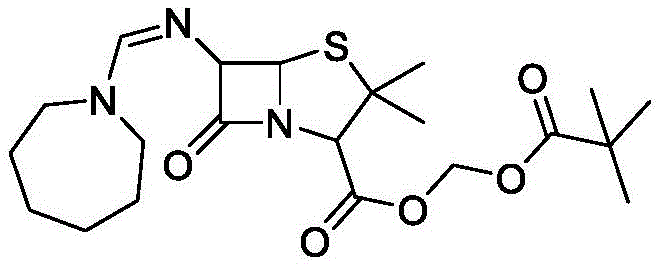
Pivmecillinam represents a significant evolution in penicillin therapy, designed to overcome the acid instability and narrow spectrum of natural penicillins. The molecular architecture, as depicted in the structural analysis, relies on specific modifications at the 6-position amino group and the 3-position carboxyl group to enhance oral bioavailability and gram-negative coverage. Traditional manufacturing routes often suffer from multiple isolation steps, which increase the risk of product degradation and elevate operational expenditures. The innovation described in the patent leverages a telescoped synthesis strategy that maintains the integrity of the sensitive beta-lactam ring while achieving high conversion rates. This technical advancement is particularly relevant for procurement and supply chain leaders looking to secure reliable sources of high-purity pharmaceutical intermediates without compromising on cost or delivery timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pivampicillin and related prodrugs involved discrete, multi-stage operations that required the isolation and purification of unstable intermediates. In conventional workflows, the alkylation of the 6-APA carboxyl group to form the pivaloyloxymethyl ester was typically performed as a standalone reaction, necessitating extensive downstream processing such as extraction, washing, and drying before the subsequent amidination step could commence. These intermediate isolation steps are not only time-consuming but also expose the reactive beta-lactam scaffold to potential hydrolytic degradation and thermal stress. Furthermore, the use of stoichiometric activating agents and the generation of substantial solvent waste during each isolation phase contribute to a higher environmental footprint and increased raw material costs. The cumulative yield losses across these disconnected stages often result in suboptimal overall efficiency, making the final API more expensive to produce and harder to source consistently in the global market.
The Novel Approach
The novel one-pot methodology described in the patent fundamentally reengineers this workflow by combining the alkylation and imidization reactions into a continuous sequence within the same reaction medium. As illustrated in the reaction scheme, the process initiates with the catalytic alkylation of 6-APA using chloromethyl pivalate, followed directly by the addition of the amidinating agent, N-formyl hexamethylene imine dimethyl sulfate, without isolating the ester intermediate. This telescoping of reactions eliminates the need for intermediate workups, drastically reducing the total processing time and solvent consumption. The use of mild reaction conditions, specifically maintaining temperatures between 20-30°C for the initial alkylation and 50-60°C for the subsequent imidization, ensures that the delicate penicillin nucleus remains intact throughout the transformation. This approach not only simplifies the operational complexity but also enhances the total yield, with reported examples achieving upwards of 86%, thereby offering a compelling value proposition for industrial manufacturers.
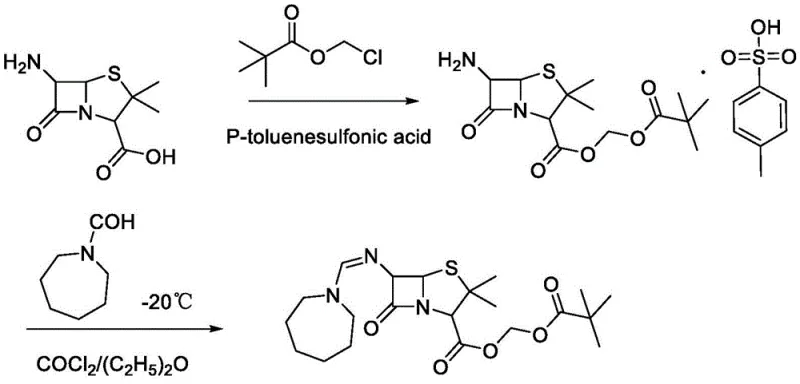
Mechanistic Insights into Catalytic Alkylation and Imidization
The core of this synthetic breakthrough lies in the precise orchestration of nucleophilic substitutions and condensation reactions under controlled catalytic conditions. The first stage involves the oxygen alkylation of the carboxylic acid moiety of 6-APA, where the presence of an iodide catalyst, such as sodium iodide or potassium iodide, plays a pivotal role in facilitating the displacement of the chloride in chloromethyl pivalate. This halogen exchange generates a more reactive iodomethyl species in situ, which is then rapidly attacked by the carboxylate anion of the 6-APA, formed through deprotonation by a base like triethylamine. The choice of polar aprotic solvents like DMF is critical here, as it stabilizes the ionic intermediates and ensures homogeneous mixing of the reagents, promoting high conversion rates without the need for excessive heating that could compromise the beta-lactam ring stability.
Following the formation of the ester intermediate, the process transitions seamlessly into the nitrogen imidization phase, where the 6-amino group of the penicillin core reacts with the N-formyl hexamethylene imine derivative. The use of the dimethyl sulfate salt of the imine serves as a potent electrophile, driving the formation of the amidine linkage essential for the drug's mechanism of action. The reaction temperature is carefully ramped to 50-60°C to provide sufficient activation energy for this condensation while remaining below the threshold where significant beta-lactam hydrolysis occurs. This mechanistic pathway effectively suppresses the formation of common impurities such as open-ring penicilloic acids or unreacted starting materials, resulting in a crude product profile that is amenable to simple recrystallization. The ability to control these competing reaction pathways through temperature and reagent stoichiometry is a key factor in achieving the high purity levels required for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Pivmecillinam Efficiently
The implementation of this one-pot synthesis route requires careful attention to reagent addition sequences and thermal profiles to maximize yield and minimize impurity formation. The process begins by charging the reactor with 6-APA, a catalytic amount of sodium iodide, and a tertiary amine base in a polar solvent, followed by the slow addition of chloromethyl pivalate to manage the exotherm. Once the alkylation is complete, the amidinating agent is introduced, and the mixture is heated to drive the imidization to completion. The detailed standardized synthesis steps, including specific mass ratios and stirring times, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Charge 6-APA, catalyst (NaI/KI), base (Triethylamine), solvent (DMF), and chloromethyl pivalate into a reactor and react at 20-30°C for 20-24 hours.
- Add N-formyl hexamethylene imine dimethyl sulfate to the mixture, heat to 50-60°C, and maintain reaction for 5-7 hours to complete imidization.
- Quench with water to precipitate crude product, filter, recrystallize from acetone, and dry at 60-70°C to obtain high-purity Pivmecillinam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis technology translates into tangible improvements in cost structure and supply reliability. By eliminating the isolation of the intermediate ester, the process significantly reduces the volume of solvents required for washing and crystallization, leading to lower waste disposal costs and a smaller environmental footprint. The simplification of the workflow also means fewer unit operations are needed, which reduces the demand on manufacturing equipment and labor hours, allowing facilities to increase their throughput capacity without capital expansion. These operational efficiencies create a buffer against raw material price volatility, enabling suppliers to offer more stable pricing models for long-term contracts. Furthermore, the robustness of the reaction conditions allows for flexible scheduling and faster turnaround times, which is critical for maintaining continuity of supply in the face of fluctuating market demands for antibiotics.
- Cost Reduction in Manufacturing: The consolidation of two reaction steps into a single vessel eliminates the need for intermediate filtration, drying, and re-dissolution, which are traditionally resource-intensive operations. This reduction in unit operations directly lowers the consumption of utilities such as steam for drying and electricity for agitation, while also minimizing the loss of product during transfer between vessels. The high overall yield reported in the patent examples indicates that less starting material is wasted, further driving down the cost of goods sold. Additionally, the use of commodity chemicals like DMF and acetone, which are readily available in the global chemical market, ensures that raw material sourcing remains cost-effective and不受 supply chain disruptions affecting exotic reagents.
- Enhanced Supply Chain Reliability: The simplicity of the workup procedure, which involves merely adding water to precipitate the crude product, reduces the complexity of the manufacturing process and the likelihood of operational errors or delays. This straightforward isolation method is less prone to bottlenecks compared to complex extraction or chromatography protocols, ensuring that production batches can be completed within predictable timeframes. The reliance on 6-APA, a widely produced fermentation product, as the sole starting scaffold means that the supply chain is anchored to a stable and abundant raw material base. Consequently, manufacturers utilizing this process can offer greater assurance of delivery consistency, mitigating the risks associated with drug shortages that often plague the antibiotic sector.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the use of hazardous reagents or extreme conditions that are difficult to manage in large reactors. The moderate temperatures and atmospheric pressure operations simplify the engineering requirements for scale-up, allowing for seamless transition from pilot plant to commercial production scales. From an environmental perspective, the reduction in solvent usage and the elimination of intermediate waste streams align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This sustainability advantage not only reduces regulatory risk but also enhances the corporate social responsibility profile of the supply chain, which is becoming a key criterion for procurement decisions in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Pivmecillinam using this advanced one-pot methodology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of the process for potential partners and stakeholders. Understanding these details is crucial for evaluating the strategic fit of this technology within your existing supply network.
Q: What are the key advantages of the one-pot synthesis method for Pivmecillinam?
A: The one-pot method eliminates the need for isolating the intermediate ester, significantly reducing solvent consumption, processing time, and overall production costs while maintaining high yields up to 86.6%.
Q: How does this process ensure the stability of the beta-lactam ring?
A: By strictly controlling the reaction temperatures between 20-30°C for alkylation and 50-60°C for imidization, the process minimizes thermal degradation and hydrolysis of the sensitive beta-lactam core.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like DMF and acetone, simple workup procedures involving water precipitation, and robust catalysts, making it highly scalable for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pivmecillinam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for essential antibiotics like Pivmecillinam. Our team of expert chemists has extensively evaluated the one-pot process described in patent CN114573603A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical companies. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Pivmecillinam meets the highest quality standards required for regulatory submission and patient safety.
We invite you to collaborate with us to leverage this innovative synthesis technology for your supply chain needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security for your antibiotic portfolio.
