Revolutionizing Acesulfame K Production: A Greener Copper-Catalyzed Route for Commercial Scale-up
Revolutionizing Acesulfame K Production: A Greener Copper-Catalyzed Route for Commercial Scale-up
The global demand for high-intensity sweeteners continues to surge, driven by the food and beverage industry's relentless pursuit of sugar reduction strategies without compromising taste profiles. In this competitive landscape, the manufacturing efficiency and environmental footprint of Acesulfame Potassium (Acesulfame K) production have become critical differentiators for suppliers. Patent CN112876427B introduces a transformative synthesis methodology that fundamentally restructures the traditional chemical pathway, replacing hazardous sulfur trioxide cyclization with a sophisticated copper-pyridine complex catalytic system. This innovation not only addresses the severe environmental liabilities associated with legacy processes but also delivers substantial improvements in reaction selectivity and overall yield. For R&D directors and procurement leaders seeking a reliable food additive supplier, this technology represents a pivotal shift towards sustainable, high-yield manufacturing that aligns with modern green chemistry principles while maintaining rigorous purity standards essential for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Acesulfame K has been dominated by the diketene-sulfur trioxide method, a process fraught with significant operational and environmental challenges that hinder scalability and cost-efficiency. As illustrated in the traditional reaction pathway below, this legacy route relies heavily on the use of stoichiometric amounts of triethylamine as a sulfonic acid protective agent, followed by a harsh cyclization step utilizing excess sulfur trioxide (SO3) at low temperatures. The reliance on SO3 creates an intensely acidic environment that promotes the unwanted polymerization of the intermediate's double bond, effectively capping the reaction yield at less than 82% and generating substantial polymeric impurities that complicate downstream purification. Furthermore, the hydrolysis of excess SO3 post-reaction generates approximately 20 tons of waste acid for every single ton of product produced, imposing a massive burden on wastewater treatment facilities and escalating environmental compliance costs. The necessity to recover and recycle the expensive triethylamine adds further complexity, requiring energy-intensive distillation columns and specialized equipment to manage the potent odor and volatility of the amine, thereby inflating both capital expenditure and operational overheads.
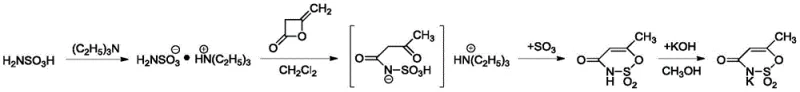
The Novel Approach
In stark contrast to the corrosive and wasteful legacy methods, the novel approach detailed in Patent CN112876427B utilizes methyl sulfamate as a strategic starting material, reacting it with diketene under the catalysis of a strong basic macroporous resin to form the key intermediate, acetoacetamido-N-sulfonyl methyl ester. This initial acylation step, depicted in the reaction scheme below, operates under mild conditions at -10°C in chlorinated hydrocarbon solvents, leveraging the heterogeneous nature of the resin catalyst to facilitate easy separation and reuse, thus eliminating the need for stoichiometric organic bases like triethylamine. By shifting the synthetic logic away from sulfamic acid and towards methyl sulfamate, the process inherently avoids the formation of difficult-to-treat fluoride wastes seen in alternative fluoride-based routes, while simultaneously securing a more stable and cost-effective raw material supply chain. The elimination of triethylamine not only reduces raw material costs but also removes the safety hazards and odor control issues associated with handling large volumes of volatile amines, creating a significantly cleaner and safer operating environment for plant personnel and surrounding communities.
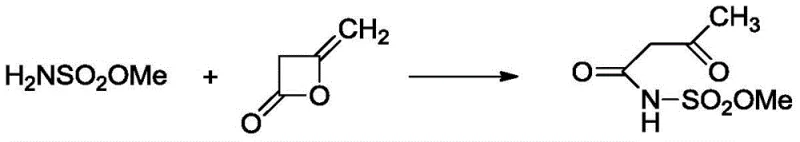
Mechanistic Insights into Copper-Pyridine Complex Catalyzed Cyclization
The core technological breakthrough of this patent lies in the second step: the cyclization of the acetoacetamido-N-sulfonyl methyl ester intermediate into the final oxathiazinone dioxide ring structure. Unlike the brute-force acid catalysis of the past, this method employs a finely tuned copper-pyridine complex catalyst system, comprising a copper salt such as CuCl2 or Cu(AcO)2 coordinated with pyridine or its derivatives like 2,2-bipyridine. Mechanistically, this transition metal complex acts as a dual-function catalyst that facilitates the critical keto-enol tautomerism of the beta-keto ester moiety, stabilizing the enol form which is the active species required for the intramolecular nucleophilic attack on the sulfonyl group. The coordination of the copper ion with the nitrogen atoms of the pyridine ligands creates a specific electronic environment that lowers the activation energy for cyclization while preventing the electrophilic attack on the double bond that leads to polymerization, a common failure mode in strong acid media. This precise control over the reaction trajectory ensures that the intermediate converts selectively into the desired cyclic structure rather than degrading into tars or polymers, directly contributing to the observed increase in cyclization selectivity to over 95%.
Furthermore, the integration of reactive distillation into this catalytic cycle serves as a powerful thermodynamic driver that pushes the equilibrium towards completion. As the cyclization proceeds, methanol is generated as a stoichiometric byproduct; if allowed to accumulate, methanol could potentially reverse the reaction or participate in transesterification side reactions. By continuously removing methanol through vacuum distillation at controlled temperatures between 60°C and 70°C, the system effectively traps the product in its lowest energy state, preventing retro-reactions and ensuring high conversion rates. This combination of homogeneous transition metal catalysis and process intensification via reactive distillation exemplifies a sophisticated understanding of physical organic chemistry, allowing for the production of high-purity Acesulfame K with minimal impurity profiles. The result is a robust process capable of consistently delivering chemical purity greater than 99.0%, meeting the stringent specifications required for high-purity food additives in regulated markets.
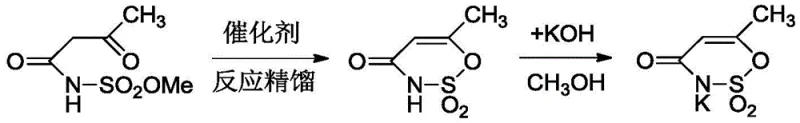
How to Synthesize Acesulfame Potassium Efficiently
The implementation of this advanced synthesis route requires careful attention to catalyst loading, temperature gradients, and vacuum control to maximize the benefits of the copper-pyridine system. The process begins with the preparation of the intermediate in a chlorinated solvent, followed by a seamless transfer to a reactive distillation setup where the cyclization occurs. Operators must maintain strict control over the reflux ratio and kettle temperature to ensure efficient methanol removal without thermal degradation of the sensitive oxathiazinone ring. The detailed standardized synthesis steps, including specific molar ratios of copper salts to pyridine ligands and precise vacuum parameters for different scales, are outlined in the guide below to ensure reproducible high-yield outcomes.
- Dissolve methyl sulfamate in a chlorinated hydrocarbon solvent and react with diketene at -10°C using a strong basic macroporous resin catalyst to form acetoacetamido-N-sulfonyl methyl ester.
- Transfer the intermediate solution to a rectification tower containing a copper-pyridine complex catalyst and heat to 60-70°C under vacuum to remove methanol byproduct via reactive distillation.
- Neutralize the resulting acetyl sulfanilic acid (ASH) with a methanol-KOH solution to pH 6.8-7.2, then cool, crystallize, and dry to obtain high-purity Acesulfame Potassium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers profound strategic advantages that extend far beyond simple yield metrics, fundamentally altering the cost structure and risk profile of Acesulfame K manufacturing. By eliminating the requirement for sulfur trioxide and triethylamine, the process removes two of the most volatile and hazardous cost centers from the bill of materials, replacing them with more stable and manageable reagents like methyl sulfamate and recoverable copper catalysts. This shift not only stabilizes raw material pricing against market fluctuations associated with specialty amines but also drastically simplifies the logistics of hazardous material transport and storage, reducing insurance premiums and regulatory compliance burdens. The qualitative reduction in waste generation translates directly into lower disposal costs and reduced strain on environmental infrastructure, allowing manufacturing sites to operate with greater flexibility and fewer permitting restrictions.
- Cost Reduction in Manufacturing: The elimination of stoichiometric triethylamine represents a direct and significant saving in raw material expenditure, as this reagent is both expensive and energy-intensive to recover and recycle in traditional processes. Additionally, the avoidance of sulfur trioxide removes the need for specialized corrosion-resistant equipment and the extensive neutralization chemicals required to treat the massive volumes of waste acid generated by the old method. The use of a macroporous resin in the first step allows for catalyst recovery and reuse, further driving down the variable cost per kilogram of product and improving the overall margin profile for large-scale production runs.
- Enhanced Supply Chain Reliability: Sourcing methyl sulfamate is generally more straightforward and less prone to geopolitical or logistical disruptions compared to specialized precursors like sulfamoyl fluoride or substituted phenyl sulfamates, which often have limited supplier bases. The robustness of the copper-pyridine catalyst system, which tolerates a range of operating conditions without significant loss of activity, ensures consistent production output even when facing minor variations in utility supply or raw material quality. This reliability is crucial for maintaining continuous supply to major food and beverage clients who demand just-in-time delivery and cannot tolerate interruptions in their sweetener supply chains.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of continuous reactive distillation, a unit operation that is well-understood and easily expanded from pilot plants to multi-ton commercial reactors without the nonlinear scale-up issues often seen in batch acid cyclizations. The drastic reduction in three-waste pollution, particularly the near-elimination of waste acid and fluoride-containing effluents, positions manufacturers to easily meet increasingly stringent global environmental regulations. This proactive environmental stewardship minimizes the risk of production shutdowns due to non-compliance and enhances the brand reputation of the supplier as a sustainable partner in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Acesulfame K synthesis route, drawing directly from the experimental data and comparative examples provided in the patent documentation. These insights are designed to clarify the operational benefits and technical feasibility for stakeholders evaluating the transition from legacy technologies to this advanced catalytic platform.
Q: Why is the copper-pyridine complex superior to traditional sulfur trioxide cyclization?
A: Traditional SO3 cyclization is highly acidic, causing polymerization of the intermediate double bond and limiting yields to below 82%. The copper-pyridine complex acts as a mild Lewis acid/base system that catalyzes the keto-enol tautomerism required for cyclization without inducing polymerization, boosting yields to over 94% while eliminating 20 tons of waste acid per ton of product.
Q: What are the specific advantages of using methyl sulfamate over sulfamoyl fluoride?
A: Sulfamoyl fluoride routes avoid triethylamine but introduce expensive, difficult-to-source raw materials and generate hard-to-treat fluoride wastewater. Methyl sulfamate is cost-effective, readily available, and the byproduct methanol is easily removed via distillation, simplifying the supply chain and waste treatment protocols significantly.
Q: How does the reactive distillation step improve process efficiency?
A: The cyclization reaction produces methanol as a byproduct. By integrating reactive distillation, methanol is continuously removed from the reaction equilibrium as it forms. This drives the reaction forward according to Le Chatelier's principle, ensuring high conversion rates and selectivity without requiring excessive reagent loading or prolonged reaction times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acesulfame Potassium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient chemical processes is not just an environmental imperative but a commercial necessity for maintaining competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the copper-pyridine catalyzed route are fully realized in practical, large-scale manufacturing environments. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify the absence of polymeric impurities and residual solvents, guaranteeing a safe and high-quality sweetener for your end applications.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this superior process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your product portfolio with a sustainably produced, high-performance sweetener.
