Advanced Synthetic Routes for Tadalafil Isomers: Enhancing Purity and Commercial Scalability
The pharmaceutical landscape for erectile dysfunction treatments continues to evolve, driven by the critical need for high-purity reference standards and impurity profiling to ensure patient safety. Patent CN107973796B, published in May 2020, introduces a robust and versatile preparation method for Tadalafil isomers, addressing significant bottlenecks in the synthesis of these complex heterocyclic compounds. This intellectual property outlines distinct pathways for generating Isomer I, Isomer II, and Isomer III, utilizing both partial racemization of the parent drug and a novel total synthesis route starting from L-Tryptophan. For R&D directors and procurement specialists, understanding these synthetic nuances is vital for securing a reliable pharmaceutical intermediates supplier capable of delivering materials that meet stringent regulatory specifications. The disclosed technology not only enhances the availability of critical reference substances but also offers a streamlined approach that mitigates the risks associated with complex chiral separations.
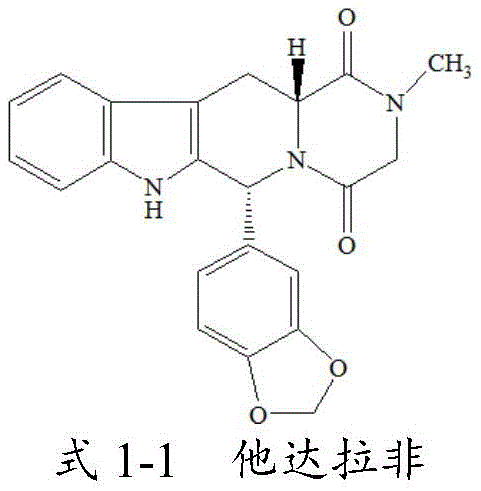
Tadalafil, chemically known as (6R,12aR)-6-(1,3-benzodioxol-5-yl)-2-methyl-2,3,6,7,12,12a-hexahydropyrazino[1',2':1,6]pyrido[3,4-b]indole-1,4-dione, possesses two chiral centers that give rise to multiple stereoisomers. The presence of these isomers necessitates rigorous control during the manufacturing of the active pharmaceutical ingredient (API) to prevent the accumulation of potentially toxic diastereomers or enantiomers. The patent highlights that while Tadalafil itself is the therapeutic agent, its isomers serve as indispensable tools for method validation, stability testing, and impurity identification. By providing a clear, reproducible method to access these specific molecular variants, the invention supports the broader ecosystem of drug development, ensuring that quality control laboratories have access to authentic standards for accurate quantification and risk assessment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of specific Tadalafil isomers has been plagued by inefficient processes that hinder both research and commercial scalability. Traditional methods often rely on non-selective synthesis followed by arduous chromatographic separations, which result in low overall yields and excessive solvent consumption. These conventional routes frequently suffer from poor stereocontrol, leading to complex mixtures that require multiple recrystallization steps to achieve the necessary optical purity. Furthermore, the reliance on expensive chiral catalysts or resolving agents in older methodologies drives up the cost of goods significantly, creating a barrier for generic manufacturers aiming to optimize their supply chains. The difficulty in purifying these closely related diastereomers often leads to extended lead times and inconsistent batch-to-batch quality, posing a substantial risk to the continuity of supply for critical drug development projects.
The Novel Approach
In stark contrast, the methodology disclosed in CN107973796B offers a paradigm shift by employing strategic partial racemization and chiral pool synthesis to bypass these traditional hurdles. For Isomer I and Isomer III, the patent describes a controlled epimerization process where the parent compound or a precursor is treated with an organic base in an aprotic polar solvent, allowing for the selective generation of the desired diastereomer with high efficiency. More notably, the total synthesis of Isomer II leverages the inherent chirality of L-Tryptophan, effectively transferring the stereochemical information from a cheap, abundant natural amino acid to the complex target molecule. This approach eliminates the need for external chiral auxiliaries and simplifies the downstream processing, as the intermediates can often be purified via simple crystallization rather than preparative HPLC.
Mechanistic Insights into Chiral Pool Synthesis and Base-Catalyzed Racemization
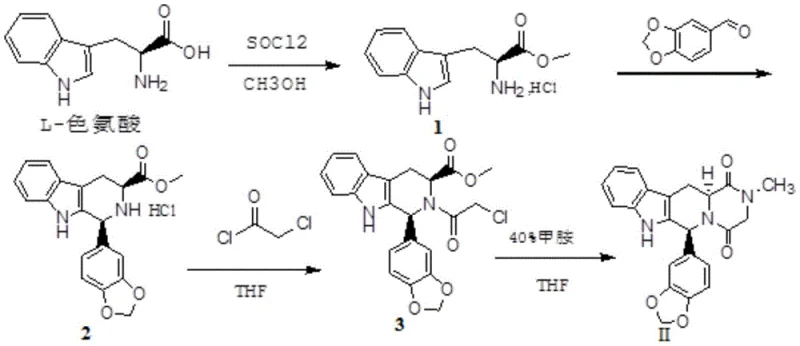
The core innovation lies in the mechanistic elegance of the four-step synthesis for Isomer II, which begins with the esterification of L-Tryptophan. In this initial stage, the carboxylic acid group is activated using thionyl chloride in methanol, forming the methyl ester hydrochloride salt while preserving the alpha-amino stereocenter. Subsequent condensation with 3,4-methylenedioxybenzaldehyde constructs the tetrahydro-beta-carboline core through a Pictet-Spengler-type cyclization, a reaction that is highly sensitive to reaction conditions but is optimized here to proceed in refluxing aprotic solvents like THF. The introduction of the chloroacetyl group in the third step sets the stage for the final ring closure, where the nucleophilic attack by the secondary amine on the chloroacetyl moiety is facilitated by the addition of aqueous methylamine. This cascade effectively builds the piperazinedione ring system characteristic of the Tadalafil scaffold without scrambling the original chirality derived from the tryptophan starting material.
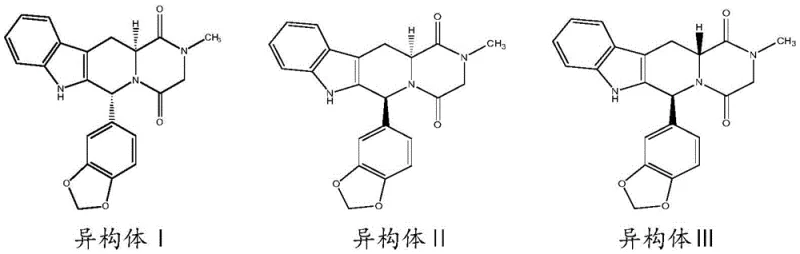
Regarding the racemization pathways for Isomer I and III, the mechanism relies on the acidity of the proton at the C-6 position adjacent to the electron-withdrawing carbonyl groups. Under the influence of strong organic bases such as DMAP, DBU, or triethylamine in solvents like DMF or DMSO, this proton is reversibly abstracted to form a planar enolate intermediate. Upon reprotonation, the stereochemistry at this center can invert, leading to an equilibrium mixture of diastereomers. By carefully controlling reaction time and temperature, the process can be tuned to favor the formation of the thermodynamically stable isomer or a specific kinetic product. This base-catalyzed epimerization is a powerful tool for interconverting stereoisomers, allowing manufacturers to convert less valuable or unwanted isomers back into the desired configuration, thereby maximizing atom economy and reducing waste generation in the overall production cycle.
How to Synthesize Tadalafil Isomer II Efficiently
The synthesis of Tadalafil Isomer II represents a masterclass in utilizing chiral pool strategy to construct complex pharmacophores with minimal stereochemical erosion. The process initiates with the conversion of L-Tryptophan into its methyl ester, followed by condensation with piperonal to establish the rigid indole-fused framework. Subsequent acylation with chloroacetyl chloride installs the necessary two-carbon linker, which then undergoes intramolecular cyclization upon exposure to methylamine to close the six-membered diketopiperazine ring. This linear sequence is designed for operational simplicity, avoiding cryogenic conditions or inert atmosphere requirements that often complicate scale-up. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures required to replicate this high-yielding pathway, please refer to the standardized synthesis guide below.
- Perform esterification of L-Tryptophan with thionyl chloride in methanol to obtain the methyl ester hydrochloride intermediate.
- Condense the tryptophan ester with 3,4-methylenedioxybenzaldehyde in an aprotic solvent like THF under reflux to form the tetrahydro-beta-carboline skeleton.
- React the condensed intermediate with chloroacetyl chloride in anhydrous tetrahydrofuran at low temperature to introduce the chloroacetyl group.
- Cyclize the chloroacetyl intermediate using aqueous methylamine solution in acetonitrile to finalize the piperazinedione ring structure of Isomer II.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the synthetic routes described in this patent offers profound advantages for procurement managers seeking cost reduction in API manufacturing. The shift towards using L-Tryptophan as a starting material capitalizes on the global availability and relatively low cost of this fermentation-derived amino acid, decoupling production from volatile markets for exotic chiral reagents. Furthermore, the reliance on common industrial solvents such as methanol, THF, and acetonitrile ensures that the process can be implemented in existing multipurpose reactors without requiring specialized equipment modifications. The simplification of purification steps, moving from complex chromatography to straightforward crystallization and filtration, drastically reduces solvent usage and processing time, which directly translates to lower operational expenditures and a smaller environmental footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the reduction in chromatographic purification steps significantly lower the variable costs associated with production. By achieving high yields in each step, particularly the final cyclization which reportedly reaches yields around 80%, the overall material throughput is maximized, reducing the cost per kilogram of the final isomer. Additionally, the ability to recycle solvents like DMF and THF further enhances the economic viability of the process, making it an attractive option for large-scale generic production where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals and widely available natural amino acids mitigates the risk of supply disruptions caused by the scarcity of specialized reagents. The robustness of the reaction conditions, which tolerate standard laboratory temperatures and pressures, ensures that the process can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs). This flexibility allows supply chain heads to diversify their vendor base and secure long-term contracts with confidence, knowing that the synthetic route is not dependent on a single source of proprietary technology or hard-to-source catalysts.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing waste generation through high atom economy and efficient solvent recovery systems. The avoidance of heavy metal catalysts simplifies the removal of trace impurities, ensuring that the final product meets strict residual solvent and heavy metal guidelines set by regulatory bodies like the FDA and EMA. This compliance readiness accelerates the regulatory filing process for new drug applications, allowing pharmaceutical companies to bring products to market faster while maintaining a sustainable manufacturing profile that aligns with modern corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of the Tadalafil isomer synthesis routes described in the patent literature. These insights are derived directly from the experimental data and process descriptions provided in CN107973796B, offering clarity on yield expectations, purity profiles, and scalability considerations for industrial application. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this technology into their existing production pipelines.
Q: What are the primary advantages of the racemization method for Isomer I described in CN107973796B?
A: The patent describes a partial racemization process using organic bases like DMAP in polar aprotic solvents, which simplifies post-treatment and achieves yields around 79.5%, significantly improving upon traditional low-yield purification methods.
Q: How does the total synthesis of Isomer II ensure stereochemical control?
A: By utilizing L-Tryptophan as the starting chiral pool material, the synthesis preserves the inherent stereochemistry of the amino acid throughout the four-step sequence, ensuring high optical purity without the need for complex chiral resolution steps.
Q: Is this preparation method suitable for large-scale commercial production?
A: Yes, the process utilizes commercially available reagents such as thionyl chloride and methylamine, and relies on standard unit operations like crystallization and filtration, making it highly adaptable for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tadalafil Isomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality isomers play in the development and validation of next-generation pharmaceuticals. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Tadalafil Isomer delivered meets the highest industry standards for identity, assay, and impurity content. Our commitment to technical excellence ensures that you receive materials that are not only chemically pure but also fully documented for regulatory submission.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods through our advanced synthetic capabilities. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can accelerate your development timelines and enhance the commercial viability of your pharmaceutical portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
