Revolutionizing Cefetamet Pivoxil Hydrochloride Production: Advanced Non-Aqueous Salt Formation for Global Supply Chains
The global demand for third-generation cephalosporin antibiotics continues to surge, driven by the persistent need for effective treatments against resistant bacterial strains in respiratory and urinary tract infections. At the forefront of meeting this demand is the efficient manufacturing of key intermediates like cefetamet pivoxil hydrochloride, a critical prodrug that enhances the oral bioavailability of the active antibiotic cefetamet. A significant technological breakthrough in this domain is documented in patent CN113801141B, which outlines a novel preparation method that fundamentally shifts the paradigm from complex, multi-step purification processes to a streamlined, direct salification technique. This innovation addresses long-standing industry pain points regarding yield loss, impurity profiles, and physical product characteristics, offering a robust pathway for pharmaceutical manufacturers seeking to optimize their supply chains. By leveraging a non-aqueous solvent system, this method ensures the structural integrity of the sensitive beta-lactam ring and the ester moieties, which are prone to degradation under traditional aqueous acidic conditions. For R&D directors and procurement strategists, understanding the nuances of this patented approach is essential for securing a reliable cefetamet pivoxil hydrochloride supplier capable of delivering consistent, high-quality material at a competitive cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of cefetamet pivoxil hydrochloride has been plagued by significant operational inefficiencies and quality control challenges that hinder large-scale commercial viability. Traditional methods often rely on the use of aqueous hydrochloric acid or hydrogen chloride gas in protic solvents like isopropanol, which frequently lead to undesirable side reactions such as the hydrolysis of the pivaloyloxymethyl ester group. This chemical instability not only reduces the overall molar yield but also generates a complex spectrum of related substances that require rigorous and costly removal. Furthermore, the physical properties of the product obtained through these legacy processes are often suboptimal, characterized by agglomeration, poor flowability, and a light yellow or off-white coloration that necessitates additional decolorization steps. To meet stringent pharmacopoeial standards, manufacturers are forced to implement repeated crystallization and purification cycles, which drastically increase solvent consumption, energy usage, and production time. These cumulative inefficiencies translate into higher manufacturing costs and extended lead times, creating bottlenecks for procurement managers tasked with maintaining continuous supply lines for essential antibiotic formulations in a volatile market environment.
The Novel Approach
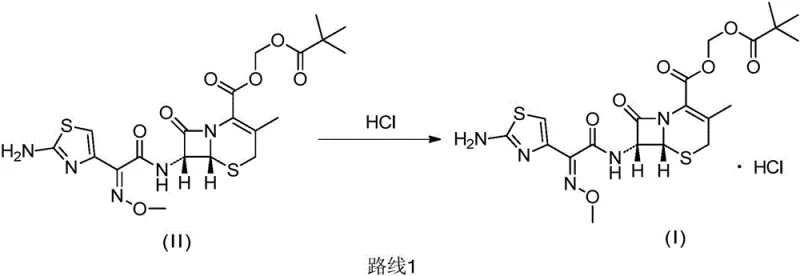
In stark contrast to these legacy limitations, the innovative method described in patent CN113801141B introduces a sophisticated solvent engineering strategy that utilizes saturated monobasic esters, particularly ethyl acetate, as the reaction medium. This approach fundamentally alters the reaction kinetics and thermodynamics, allowing for the direct conversion of cefetamet pivoxil to its hydrochloride salt with exceptional selectivity and efficiency. By employing a non-aqueous solution of hydrogen chloride, the process effectively eliminates the risk of water-induced hydrolysis, thereby preserving the delicate ester functionality required for the drug's prodrug activity. The result is a finished product that exhibits superior physical characteristics, appearing as a free-flowing white powder with excellent bulk density, which significantly simplifies downstream handling and tablet compression processes. Moreover, this method demonstrates remarkable tolerance to the purity of the starting material, meaning that expensive, high-grade intermediates are not a prerequisite for achieving a high-purity final product. This capability represents a major leap forward in cost reduction in antibiotic intermediates manufacturing, as it removes the need for resource-intensive intermediate purification stages while simultaneously boosting overall process throughput and reliability.
Mechanistic Insights into Non-Aqueous Acid Salification
The core chemical advantage of this novel synthesis lies in the precise control of the protonation environment surrounding the aminothiazole moiety of the cefetamet pivoxil molecule. In traditional aqueous systems, the presence of water molecules competes with the substrate for interaction with the acid catalyst, often leading to nucleophilic attack on the electrophilic carbonyl carbons of the ester groups. However, in the non-aqueous ester solvent system, the activity of the hydrogen chloride is modulated by the solvent's dielectric constant and solvation properties, favoring the selective protonation of the basic amino nitrogen without triggering ester cleavage. This selectivity is crucial for maintaining the high-purity cefetamet pivoxil profile required for regulatory approval, as it minimizes the formation of degradation products like cefetamet acid or delta-2 isomers. The reaction proceeds through a stable transition state where the hydrogen chloride is evenly distributed throughout the organic phase, ensuring uniform salt formation across the entire batch volume. This homogeneity is critical for preventing localized hotspots of acidity that could otherwise degrade the beta-lactam ring, a common failure mode in less controlled acidification processes.
Furthermore, the choice of ethyl acetate as the preferred solvent plays a dual role in both reaction facilitation and product isolation. Ethyl acetate provides an optimal solubility profile for the free base cefetamet pivoxil at elevated temperatures while allowing the hydrochloride salt to precipitate efficiently upon cooling. This temperature-dependent solubility differential drives the crystallization process, encouraging the formation of well-defined crystal lattices that exclude impurities from the growing solid phase. The mechanism effectively acts as a self-purifying step, where the thermodynamic drive to form the stable hydrochloride salt in the specific solvent matrix leaves most organic impurities in the mother liquor. Consequently, the final filtration yields a product with unknown single impurities consistently below 0.1% and known single impurities below 0.5%, meeting the most rigorous international quality standards without the need for chromatographic purification. This mechanistic elegance translates directly into commercial value, offering a scalable and robust manufacturing route that mitigates the risks associated with batch-to-batch variability.
How to Synthesize Cefetamet Pivoxil Hydrochloride Efficiently
The implementation of this advanced synthesis route requires careful attention to process parameters to fully realize its potential for yield optimization and impurity control. The procedure begins with the dissolution of the cefetamet pivoxil starting material in the selected saturated monobasic ester solvent, with temperature control being a critical variable to ensure complete solubility prior to acid addition. The subsequent addition of the non-aqueous hydrogen chloride solution must be managed meticulously, preferably in multiple portions or via a controlled dropwise addition, to manage the exothermic nature of the salification reaction and prevent thermal degradation of the product. Detailed standardized synthetic steps see the guide below.
- Dissolve crude cefetamet pivoxil in a saturated monobasic ester solvent such as ethyl acetate under controlled temperature conditions between 30°C and 40°C.
- Slowly add a non-aqueous hydrogen chloride solution, preferably in ethyl acetate, in multiple portions to control exothermic reaction and ensure complete salification.
- Cool the reaction mixture to 0-5°C to induce crystallization, filter the resulting white powder, and dry under vacuum to obtain high-purity cefetamet pivoxil hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented manufacturing process offers transformative benefits that extend far beyond simple chemical yield improvements. The elimination of intermediate purification steps represents a significant reduction in operational complexity, directly translating to lower capital expenditure on equipment and reduced consumption of auxiliary materials such as filtration media and decolorizing agents. By streamlining the production workflow, manufacturers can achieve a drastic simplification of the supply chain, reducing the number of unit operations required to bring the product from raw material to finished goods. This efficiency gain allows for faster turnaround times and greater flexibility in responding to market demand fluctuations, ensuring that pharmaceutical partners can maintain adequate inventory levels without the burden of excessive safety stock. Additionally, the use of common, industrially available solvents like ethyl acetate enhances supply chain reliability, as these materials are less subject to the volatility and regulatory restrictions often associated with specialized or hazardous reagents used in older methodologies.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the removal of costly recrystallization cycles that traditionally consume vast amounts of solvent and energy. By achieving high purity directly from the reaction mixture, the process eliminates the yield losses inherent in multiple purification steps, where significant amounts of valuable product are often discarded in mother liquors. Furthermore, the improved physical properties of the product reduce processing time during drying and milling, leading to substantial cost savings in utility consumption and labor hours. The ability to use lower purity starting materials without compromising the final product quality also opens up opportunities for sourcing more cost-effective raw materials, further driving down the overall cost of goods sold and enhancing profit margins for all stakeholders in the value chain.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthesis method ensures a more predictable and stable supply of high-purity cefetamet pivoxil hydrochloride. The reduced sensitivity to starting material quality means that supply disruptions caused by the unavailability of ultra-high-grade intermediates are minimized, allowing for greater flexibility in vendor selection and raw material procurement. The consistent production of a free-flowing white powder also improves packaging efficiency and reduces the risk of product degradation during storage and transport, ensuring that the material arrives at the formulation site in optimal condition. This reliability is critical for maintaining uninterrupted production schedules for finished dosage forms, safeguarding against stockouts that could impact patient access to essential antibiotic therapies in global markets.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing equipment and conditions that are easily transferable from pilot plant to full-scale industrial production. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the burden on waste treatment facilities and lowering the carbon footprint of the manufacturing operation. By minimizing the release of organic volatiles and hazardous byproducts, this method supports a greener manufacturing profile that is increasingly valued by regulatory bodies and end consumers alike. This environmental compliance not only mitigates regulatory risk but also enhances the brand reputation of companies adopting this technology as responsible leaders in sustainable pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality assurance of cefetamet pivoxil hydrochloride using this advanced non-aqueous method. These insights are derived directly from the experimental data and beneficial effects reported in the underlying patent literature, providing a transparent view of the technology's capabilities. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing portfolios.
Q: Why is non-aqueous hydrogen chloride preferred over aqueous hydrochloric acid for this synthesis?
A: Using non-aqueous hydrogen chloride prevents the hydrolysis of the sensitive pivaloyloxymethyl ester group, which can occur in the presence of water, thereby maintaining high chemical purity and stability of the final antibiotic intermediate.
Q: How does this method improve the physical properties of the product compared to conventional techniques?
A: The novel solvent system prevents the agglomeration and discoloration often seen in traditional isopropanol-based methods, resulting in a free-flowing white powder that is easier to handle, package, and formulate in downstream pharmaceutical manufacturing.
Q: Is this process suitable for large-scale industrial production without intermediate purification?
A: Yes, the process is specifically designed to tolerate lower purity starting materials while still delivering a finished product with over 99% purity, eliminating the need for costly and time-consuming intermediate recrystallization steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefetamet Pivoxil Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes requires a partner with deep technical expertise and proven industrial capability. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented synthesis are fully realized in practical, large-volume output. Our facilities are equipped with state-of-the-art reaction vessels and precision temperature control systems necessary to execute the non-aqueous salification process with exacting standards. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of cefetamet pivoxil hydrochloride meets the highest global pharmacopoeial requirements, providing our clients with the confidence needed to streamline their own regulatory filings and product launches.
We invite you to collaborate with us to optimize your antibiotic supply chain and leverage these technological advancements for your commercial success. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets, demonstrating exactly how this process can improve your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and expert evaluation. Together, we can ensure a stable, high-quality supply of this critical intermediate to support the global fight against bacterial infections.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
