Advanced Synthesis of Glycocyclopyrrole Intermediates for Commercial Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways to access complex heterocyclic scaffolds, particularly those containing nitrogen. Patent CN111518105A discloses a groundbreaking preparation method for glycocyclo-pyrrole cyclic compounds, addressing critical bottlenecks in the synthesis of highly functionalized pyrroles. This technology leverages a sophisticated tandem sequence involving a Ferrier rearrangement followed by a Barton-Zard reaction, transforming readily available 2-nitroglycals into valuable pyranopyrrole derivatives. Unlike traditional methods that often struggle with regioselectivity and harsh conditions, this novel approach offers a streamlined, green, and economically viable route. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the supply chain for nitrogen-containing heterocycles, ensuring high purity while drastically simplifying the manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of β-substituted pyrroles fused with sugar moieties has been a formidable synthetic challenge. Prior art, such as the work by Pathak's group in 2008, relied heavily on vinyl sulfone-modified carbohydrates as starting materials. While effective in a laboratory setting, these vinyl sulfones are notoriously difficult to prepare on a large scale, requiring multi-step syntheses that drive up costs and extend lead times. Furthermore, the subsequent ring-opening steps often necessitated the use of phosphorus oxychloride (POCl3) in DMF, a combination that poses severe safety hazards and generates toxic waste streams. The high equivalent of base required and prolonged reaction times in these conventional routes further exacerbate production costs, making them unsuitable for the rigorous demands of modern industrial pharmaceutical manufacturing where efficiency and environmental compliance are paramount.
The Novel Approach
The methodology outlined in CN111518105A revolutionizes this landscape by utilizing 2-nitroglycals as the primary feedstock, which are far more accessible and stable than vinyl sulfones. The core innovation lies in the seamless integration of an NHC-catalyzed Ferrier rearrangement with a Barton-Zard cyclization. This dual-reaction strategy allows for the direct construction of the pyranopyrrole core under remarkably mild conditions, typically at room temperature in dichloromethane. By eliminating the need for toxic chlorinating agents and reducing the stoichiometric requirement of bases and isonitrile compounds, this process achieves a level of atom economy and operational simplicity that was previously unattainable. 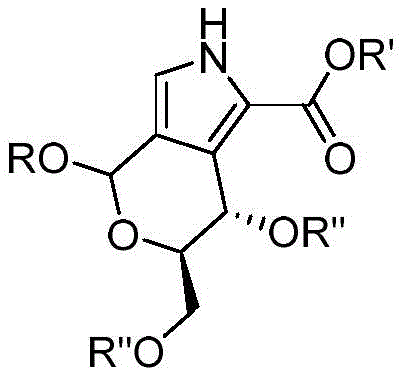
Mechanistic Insights into NHC-Catalyzed Ferrier Rearrangement and Barton-Zard Cyclization
The success of this synthesis hinges on the precise orchestration of two distinct mechanistic events. The first stage involves the Ferrier rearrangement, where an N-heterocyclic carbene (NHC) catalyst activates the 2-nitroglycal substrate. In the presence of a mild base like cesium carbonate and an alcohol nucleophile, the glycal undergoes allylic transposition to form a 2,3-unsaturated glycoside. This step is critical as it establishes the necessary carbon-oxygen framework while preserving the stereochemical integrity of the sugar ring, which serves as a chiral pool for the final molecule. The NHC catalyst plays a pivotal role here, facilitating the reaction at ambient temperatures and minimizing side reactions that could lead to impurity formation.
Following the in-situ generation of the unsaturated glycoside, the system transitions directly into the Barton-Zard reaction without the need for intermediate isolation. The addition of methyl isocyanoacetate and a second equivalent of base triggers a cycloaddition process. The isocyanoacetate acts as a 1,3-dipole equivalent, attacking the electron-deficient double bond of the glycoside. This results in the closure of the pyrrole ring, fused perfectly to the existing pyranose structure. The mild basic conditions ensure that the sensitive acetyl protecting groups on the sugar remain intact, preventing deprotection issues that often plague carbohydrate chemistry. This mechanistic elegance ensures a clean reaction profile, yielding products with high structural fidelity and minimal byproduct generation.
How to Synthesize Glycocyclopyrrole Efficiently
To implement this synthesis effectively, operators must adhere to strict anhydrous conditions and precise stoichiometric controls. The process begins with the dissolution of 2-nitroperacetylated glucosene in dry dichloromethane under an inert nitrogen atmosphere. The addition of the alcohol component, such as benzyl alcohol or methanol, alongside the NHC catalyst and a catalytic amount of cesium carbonate initiates the Ferrier rearrangement. Once TLC monitoring confirms the consumption of the starting material, the reaction mixture is treated directly with methyl isocyanoacetate and a larger excess of base to drive the Barton-Zard cyclization to completion. 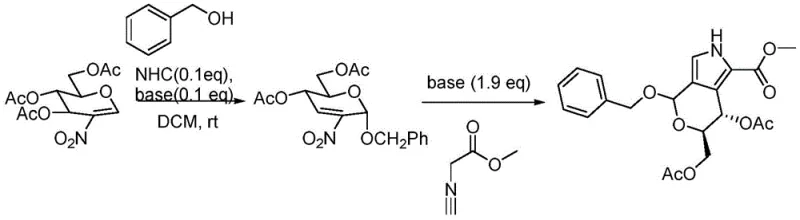
- Perform Ferrier rearrangement of 2-nitroglycal with an alcohol using NHC catalyst and mild base in DCM at room temperature.
- Without isolation, add methyl isocyanoacetate and cesium carbonate to the reaction mixture for the Barton-Zard reaction.
- Monitor reaction completion via TLC, then purify the crude product using column chromatography to obtain the target glycocyclopyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits beyond mere technical feasibility. The shift away from custom-synthesized vinyl sulfones to commodity chemicals like simple alcohols and carbonates fundamentally alters the cost structure of the intermediate. By removing the dependency on complex, multi-step precursor synthesis, manufacturers can secure a more stable and predictable supply of raw materials, insulating production schedules from upstream volatility. Furthermore, the elimination of hazardous reagents like POCl3 simplifies waste management protocols, reducing the overhead associated with environmental compliance and hazardous waste disposal.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the reagent profile. Replacing expensive, hard-to-source vinyl sulfones with inexpensive alcohols and commercially available 2-nitroglycals leads to substantial raw material cost savings. Additionally, the reduction in base equivalents and the ability to run the reaction at room temperature significantly lowers energy consumption and utility costs. The avoidance of toxic reagents also means reduced expenditure on specialized containment equipment and waste treatment facilities, contributing to a leaner overall production budget.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved due to the use of widely available starting materials. Alcohols such as methanol, ethanol, and benzyl alcohol are bulk commodities with robust global supply networks, ensuring consistent availability even during market fluctuations. The simplified two-step one-pot nature of the reaction reduces the number of unit operations required, thereby shortening the overall manufacturing cycle time. This agility allows for faster response to demand spikes and reduces the inventory holding costs associated with long-duration batch processes.
- Scalability and Environmental Compliance: From a scale-up perspective, the mild reaction conditions (0°C to 60°C) and the use of standard organic solvents like dichloromethane make this process highly adaptable to large-scale reactors. There is no need for extreme temperatures or pressures, which minimizes engineering risks during technology transfer. Environmentally, the process aligns with green chemistry principles by avoiding heavy metals and toxic chlorinating agents. This facilitates easier regulatory approval and supports corporate sustainability goals, making the final API or intermediate more attractive to environmentally conscious downstream partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glycocyclopyrrole synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process robustness and applicability.
Q: What are the key advantages of this new synthesis method over traditional vinyl sulfone routes?
A: The new method avoids the use of difficult-to-prepare vinyl sulfone starting materials and eliminates the need for highly toxic reagents like POCl3. It operates under milder conditions (room temperature) with shorter reaction times and lower base equivalents, significantly reducing production costs and environmental impact.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It utilizes cheap and easily obtained reagents such as simple alcohols and carbonates. The reaction conditions are mild (0°C to 60°C) and do not require specialized high-pressure or cryogenic equipment, making it highly suitable for commercial scale-up.
Q: What is the stereochemical outcome of the Ferrier rearrangement in this protocol?
A: The Ferrier rearrangement preserves the chirality of the sugar backbone while introducing a new stereocenter at the anomeric position. The use of NHC catalysts helps control the stereoselectivity, ensuring the formation of the desired 2,3-unsaturated glycoside which is crucial for the subsequent Barton-Zard cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycocyclopyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Ferrier-Barton-Zard cascade in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of glycocyclopyrrole intermediate adheres to the highest quality standards required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis can be integrated into your specific drug development pipeline. By leveraging our expertise, you can achieve significant process optimization and secure a reliable supply of critical building blocks. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project needs.
