Revolutionizing Phosphine Ligand Production: A Safe, Scalable Route for High-Purity Intermediates
The landscape of organophosphorus chemistry is undergoing a significant transformation with the disclosure of patent CN113105502B, which introduces a groundbreaking method for synthesizing tert-butyldiphenylphosphine. This compound serves as a critical ligand in palladium-catalyzed cross-coupling reactions, essential for constructing complex molecular architectures found in active pharmaceutical ingredients (APIs) and advanced functional materials. Historically, the production of such sterically hindered phosphines has been plagued by safety hazards and economic inefficiencies, but this new technology leverages a direct dehydration strategy between tert-butanol and diphenylphosphine. By shifting away from hazardous organometallic reagents towards benign alcohol precursors, the patent outlines a pathway that not only enhances operator safety but also drastically simplifies the supply chain logistics for fine chemical manufacturers. The implications for R&D directors and procurement strategists are profound, as this method promises to stabilize the availability of high-performance ligands while mitigating the volatility associated with traditional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the standard industrial protocols for generating tert-butyldiphenylphosphine have relied heavily on the nucleophilic substitution of diphenylphosphine with tert-butyl halides or the lithiation of phosphines followed by alkylation. These conventional pathways suffer from severe drawbacks, primarily centered around the use of tert-butyl lithium, a reagent known for its pyrophoric nature and requirement for stringent cryogenic conditions below -78°C. Such extreme operating parameters necessitate specialized equipment and rigorous safety protocols, driving up capital expenditure and operational costs significantly. Furthermore, alternative routes utilizing tert-butyl iodide introduce substantial atom economy issues due to the generation of stoichiometric amounts of iodine-containing waste streams, which are environmentally burdensome and costly to treat. The combination of high reagent costs, dangerous handling requirements, and complex waste management has long created a bottleneck in the reliable sourcing of this valuable ligand for the pharmaceutical and agrochemical sectors.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN113105502B employs a catalytic dehydration reaction that directly couples tert-butanol with diphenylphosphine. This innovative approach utilizes inexpensive and widely available metal oxide catalysts, such as manganese dioxide, aluminum oxide, or silicon dioxide, to facilitate the formation of the carbon-phosphorus bond under mild thermal conditions. The reaction proceeds efficiently in common organic solvents like toluene at temperatures ranging from 80 to 100°C, completely eliminating the need for cryogenic cooling or the handling of moisture-sensitive organolithium species. 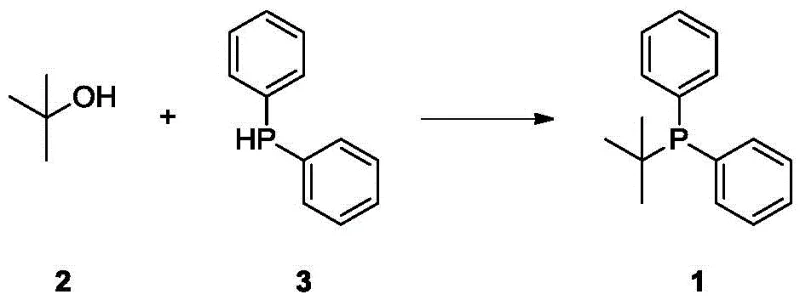 This direct condensation strategy not only streamlines the synthetic sequence by reducing the number of unit operations but also ensures a cleaner reaction profile with fewer by-products. By replacing high-cost halides and dangerous lithiation reagents with bulk commodity alcohols, this novel route offers a compelling value proposition for manufacturers seeking to optimize their production economics without compromising on product quality or purity standards.
This direct condensation strategy not only streamlines the synthetic sequence by reducing the number of unit operations but also ensures a cleaner reaction profile with fewer by-products. By replacing high-cost halides and dangerous lithiation reagents with bulk commodity alcohols, this novel route offers a compelling value proposition for manufacturers seeking to optimize their production economics without compromising on product quality or purity standards.
Mechanistic Insights into Heterogeneous Catalytic Dehydration
The core of this technological breakthrough lies in the activation of the hydroxyl group of tert-butanol by the surface acid sites of the heterogeneous metal oxide catalyst. Under an inert argon atmosphere, the catalyst facilitates the elimination of water from the alcohol, generating a reactive carbocation-like intermediate or activating the phosphine for a direct nucleophilic attack. This mechanism avoids the formation of free radical species that often lead to polymerization or degradation in traditional high-temperature processes. The use of solid catalysts like MnO2 or SiO2 provides a distinct advantage in terms of selectivity, as the surface properties can be tuned to favor the formation of the desired P-C bond while suppressing side reactions such as the oxidation of the phosphine to phosphine oxide. This controlled environment is crucial for maintaining the electronic integrity of the phosphine ligand, ensuring it retains the necessary electron-rich character required for effective coordination with transition metals in downstream catalytic applications.
Furthermore, the heterogeneous nature of the catalytic system plays a pivotal role in impurity control and downstream processing efficiency. Unlike homogeneous catalysts that require complex aqueous workups or chromatographic separation to remove trace metals, the solid catalyst in this process can be simply removed via filtration upon reaction completion. This physical separation step significantly reduces the risk of metal contamination in the final product, a critical parameter for pharmaceutical intermediates where residual metal limits are strictly regulated. The robustness of the catalyst also allows for potential recycling or regeneration, further enhancing the sustainability profile of the process. By minimizing the introduction of extraneous reagents and simplifying the purification train, this method ensures a high-purity product profile that meets the stringent specifications demanded by global API manufacturers and fine chemical consumers.
How to Synthesize Tert-Butyldiphenylphosphine Efficiently
The implementation of this synthesis route involves a straightforward sequence of operations designed for maximum efficiency and safety in a standard chemical plant setting. The process begins with the preparation of a dry reactor under an inert atmosphere, followed by the sequential addition of the solvent, alcohol, phosphine, and catalyst. The reaction is then driven to completion through moderate heating, after which the product is isolated through standard separation techniques.
- Prepare a dry reactor under argon protection and add toluene solvent, followed by tert-butanol, diphenylphosphine, and a metal oxide catalyst such as MnO2 or SiO2.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 6 to 8 hours to ensure complete conversion.
- Upon completion, cool the system, filter off the solid catalyst, and proceed with extraction, drying, and vacuum distillation to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method addresses several critical pain points currently affecting the global supply chain for organophosphorus ligands. The shift from specialized, hazardous reagents to commodity chemicals fundamentally alters the cost structure and risk profile of production. For procurement managers, this translates into a more predictable pricing model and reduced exposure to supply disruptions caused by the limited availability of niche reagents like tert-butyl lithium. The simplified operational requirements also mean that production can be scaled up more rapidly to meet surging demand from the pharmaceutical and electronics sectors, ensuring continuity of supply for key customers.
- Cost Reduction in Manufacturing: The replacement of expensive tert-butyl iodide and tert-butyl lithium with low-cost tert-butanol results in a dramatic decrease in raw material expenditures. Additionally, the elimination of cryogenic cooling systems and the reduction in energy consumption due to milder reaction temperatures contribute to significant operational savings. The ability to use simple filtration for catalyst removal further lowers processing costs by reducing solvent usage and waste treatment fees associated with complex aqueous workups.
- Enhanced Supply Chain Reliability: Tert-butanol is a bulk chemical produced on a massive scale for various industries, ensuring a stable and abundant supply source that is not subject to the same geopolitical or manufacturing constraints as specialized organometallic reagents. This abundance mitigates the risk of raw material shortages and allows for more flexible inventory management strategies. Furthermore, the reduced safety hazards associated with the new process lower insurance premiums and regulatory compliance costs, adding another layer of financial stability to the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard batch reactor operations and common solvents like toluene. The use of heterogeneous catalysts minimizes the generation of heavy metal waste, aligning with increasingly stringent environmental regulations regarding effluent discharge. The high atom economy of the dehydration reaction means less waste is generated per kilogram of product, supporting corporate sustainability goals and reducing the environmental footprint of chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process advantages.
Q: What are the primary safety advantages of this new synthesis method compared to traditional routes?
A: Traditional methods often rely on tert-butyl lithium, which is pyrophoric and requires cryogenic conditions, or tert-butyl iodide, which generates hazardous iodine waste. This new method utilizes tert-butanol, a stable and non-hazardous bulk chemical, significantly reducing operational risks and eliminating the need for extreme temperature controls.
Q: How does the use of heterogeneous catalysts like MnO2 or SiO2 impact the purification process?
A: The use of solid metal oxide catalysts allows for a simple filtration step to remove the catalyst after the reaction is complete. This simplifies the downstream processing compared to homogeneous catalysis, reducing solvent usage and minimizing the risk of metal contamination in the final high-purity ligand product.
Q: Is this synthesis route suitable for large-scale industrial production of phosphine ligands?
A: Yes, the process operates at mild temperatures (80-100°C) using common solvents like toluene and inexpensive, readily available raw materials. The high yields (>85%) and simple workup procedures make it highly scalable and economically viable for commercial manufacturing of complex organophosphorus compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyldiphenylphosphine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the production of high-value phosphine ligands. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality supply regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tert-butyldiphenylphosphine performs optimally in your critical cross-coupling reactions.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project, demonstrating how this efficient route can optimize your overall manufacturing budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global market.
