Advanced Sulfonation Technology for High-Purity Amino-K Acid Manufacturing
Advanced Sulfonation Technology for High-Purity Amino-K Acid Manufacturing
The chemical industry is constantly evolving towards more sustainable and efficient manufacturing processes, particularly in the synthesis of critical dye intermediates like Amino-K acid. Patent CN107556217B introduces a groundbreaking production process that fundamentally restructures the sulfonation reaction mechanism to address long-standing environmental and efficiency challenges. This innovation replaces the traditional reliance on excessive amounts of 65% fuming sulfuric acid with a precise combination of 100% concentrated sulfuric acid and a chlorosulfonic acid auxiliary agent. By optimizing the molar ratios and reaction temperatures, this method not only achieves superior conversion rates exceeding 99.4% but also drastically mitigates the generation of hazardous waste acid. For global procurement leaders and R&D directors, this represents a significant leap forward in cost reduction in dye intermediate manufacturing, offering a pathway to cleaner production without compromising on the rigorous quality standards required for azo and reactive dye synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Amino-K acid has been plagued by inefficient resource utilization and severe environmental burdens due to the reliance on outdated sulfonation techniques. The conventional process typically employs 65% fuming sulfuric acid as the primary sulfonating agent, necessitating a massive excess—often up to ten times the molar amount of the raw material, amino G acid—to drive the reaction to completion. This excessive addition leads to a cascade of negative outcomes, including the formation of substantial quantities of strong-acid wastewater that requires complex and costly neutralization treatments before disposal. Moreover, the low selectivity inherent in this brute-force approach results in the generation of numerous byproducts, such as sulfone substances and coking materials, which directly degrade the purity and yield of the final product. The accumulation of waste acid not only poses significant regulatory compliance risks but also inflates the operational expenditure associated with waste management and raw material consumption, creating a bottleneck for scalable and sustainable production.
The Novel Approach
In stark contrast, the novel process detailed in the patent data introduces a sophisticated, multi-stage reaction protocol that maximizes atomic economy and minimizes waste generation from the outset. The method begins by slowly adding amino G acid into 100% concentrated sulfuric acid at a controlled molar ratio of 1:3, maintaining a moderate temperature range of 20-65°C to ensure stable initial sulfonation. Crucially, after an initial reaction period of 3-4 hours, a sulfonation auxiliary agent, specifically chlorosulfonic acid, is introduced to the reaction mixture at a molar ratio of 1.3:1. This strategic addition allows the reaction temperature to be carefully managed between 60-70°C before being elevated to 125-130°C for the final completion phase. This refined approach ensures that the effective concentration of sulfuric acid remains optimal throughout the process without the need for volumetric excess, thereby enhancing the selectivity of the sulfonation at the desired positions on the naphthalene ring and significantly boosting the overall yield of the target molecule.
Mechanistic Insights into Chlorosulfonic Acid Assisted Sulfonation
The core innovation of this production process lies in the dynamic management of water content within the reaction system, which is critical for maintaining the strength of the sulfonating agent. In traditional sulfonation, the water produced as a byproduct dilutes the sulfuric acid, necessitating the addition of large volumes of excess acid to maintain reaction kinetics. However, in this novel method, the introduction of chlorosulfonic acid serves a dual purpose: it acts as a potent sulfonating species itself and, more importantly, reacts rapidly with the water generated during the decomposition and sulfonation reactions. This reaction effectively consumes the water and regenerates sulfuric acid in situ, creating a self-sustaining cycle that preserves the high acidity required for the reaction to proceed efficiently. By preventing the dilution of the reaction medium, the process ensures that the sulfonation occurs selectively at the 3, 6, and 8 positions of the naphthalene ring, minimizing the formation of isomeric byproducts and ensuring a high-purity profile for the final Amino-K acid product.
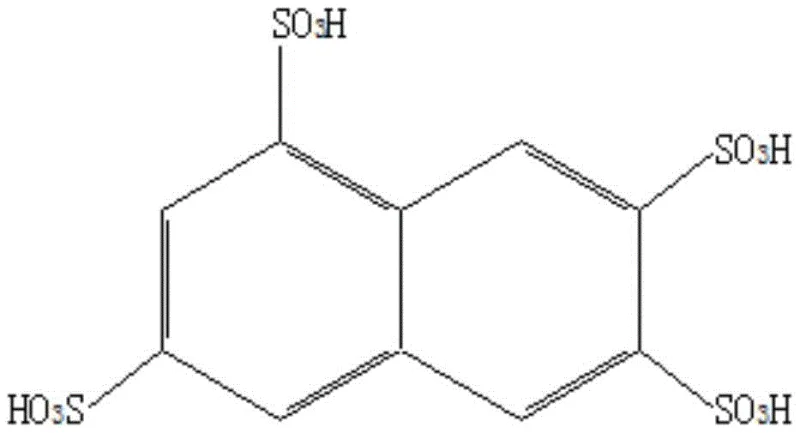
Furthermore, the precise control of temperature gradients throughout the four distinct steps of the process plays a vital role in impurity control and reaction safety. The initial low-temperature phase prevents localized overheating and oxidative degradation of the sensitive amino group, while the subsequent intermediate temperature phase allows for the thorough mixing and reaction of the chlorosulfonic acid auxiliary. The final high-temperature stage ensures the completion of the sulfonation at the more sterically hindered positions, driving the conversion of amino G acid to near completion. This meticulous thermal management, combined with the chemical regulation of acid strength, results in a product with a conversion rate consistently above 99.4% and a selectivity that far exceeds conventional methods. For R&D teams, understanding this mechanism provides a robust framework for scaling up the synthesis of complex sulfonated intermediates while maintaining strict control over the impurity spectrum.
How to Synthesize Amino-K Acid Efficiently
The synthesis of Amino-K acid via this advanced protocol requires precise adherence to the specified reaction conditions to fully realize the benefits of waste reduction and yield improvement. The process is designed to be operationally straightforward yet chemically sophisticated, leveraging the unique properties of chlorosulfonic acid to optimize the sulfonation environment. Operators must carefully monitor the addition rates and temperature profiles to ensure the safety and efficacy of the reaction, particularly during the exothermic addition of the auxiliary agent. The following guide outlines the standardized steps derived from the patent examples, providing a clear roadmap for implementing this technology in a commercial setting. Detailed standardized synthesis steps are provided in the guide below.
- Slowly add amino G acid into 100% concentrated sulfuric acid at a molar ratio of 1: 3, controlling the temperature between 20-65°C for 3-4 hours.
- Add chlorosulfonic acid (sulfonation auxiliary) at a molar ratio of 1.3: 1 to the reaction liquid, maintaining 60-70°C for 1-1.5 hours to consume generated water.
- Raise the temperature to 125-130°C and maintain for 5-7 hours to complete the sulfonation, then cool and centrifuge to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel production process translates into tangible strategic advantages that extend far beyond simple chemical yield metrics. By fundamentally altering the stoichiometry of the reaction to require significantly less sulfuric acid, the process inherently reduces the raw material burden and the logistical complexity associated with handling and storing large volumes of corrosive acids. This shift not only lowers the direct cost of goods sold but also mitigates the risks associated with the supply volatility of bulk chemicals, ensuring a more resilient and predictable production schedule. Additionally, the drastic reduction in waste acid generation simplifies the downstream purification and waste treatment processes, leading to substantial operational savings and a smaller environmental footprint that aligns with increasingly stringent global sustainability regulations.
- Cost Reduction in Manufacturing: The elimination of excessive sulfuric acid usage creates a direct and significant impact on the variable costs associated with Amino-K acid production. By reducing the molar ratio of sulfuric acid from a traditional 10:1 down to a highly efficient 3:1, the process minimizes the expenditure on raw materials while simultaneously lowering the costs related to waste neutralization and disposal. The regeneration of sulfuric acid through the chlorosulfonic acid auxiliary further enhances this efficiency, ensuring that every mole of acid contributes effectively to the reaction rather than becoming waste. This optimized resource utilization allows manufacturers to achieve a more competitive cost structure without sacrificing product quality, providing a distinct market advantage in price-sensitive sectors.
- Enhanced Supply Chain Reliability: The simplified reaction protocol and reduced dependency on massive quantities of fuming sulfuric acid contribute to a more robust and reliable supply chain. With lower volumes of hazardous materials required for each batch, the logistical challenges of transportation and storage are significantly alleviated, reducing the potential for supply disruptions caused by regulatory restrictions or transport limitations. Furthermore, the higher selectivity and yield of the process mean that less raw material is needed to produce the same amount of finished product, effectively increasing the throughput capacity of existing manufacturing facilities. This efficiency gain ensures a steadier flow of high-purity intermediates to downstream customers, reinforcing the reliability of the supply partnership.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily controllable and reproducible on a large industrial scale. The significant reduction in waste acid volume addresses one of the most critical environmental pain points in dye intermediate manufacturing, facilitating easier compliance with environmental protection laws and reducing the liability associated with hazardous waste management. By minimizing the generation of byproducts and waste streams, the process supports a cleaner production model that is increasingly demanded by end-users in the textile and agrochemical industries. This alignment with green chemistry principles not only future-proofs the manufacturing operation but also enhances the brand value of the supplier as a responsible and sustainable partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced Amino-K acid production technology. These insights are derived directly from the patent specifications and experimental data, providing a factual basis for decision-making. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this process for their own manufacturing lines or sourcing strategies.
Q: How does the new process reduce waste acid compared to traditional methods?
A: The novel process utilizes 100% concentrated sulfuric acid with a molar ratio of only 1:3 relative to the raw material, compared to the traditional 10-fold excess of 65% fuming sulfuric acid. Furthermore, the addition of chlorosulfonic acid reacts with water generated during sulfonation to regenerate sulfuric acid, drastically reducing the volume of waste acid requiring treatment.
Q: What is the role of chlorosulfonic acid in this synthesis?
A: Chlorosulfonic acid acts as a sulfonation auxiliary agent that reacts with water produced during the decomposition and sulfonation reactions. This reaction consumes water and generates additional sulfuric acid in situ, maintaining a high effective acid concentration without needing excessive bulk acid, thereby improving selectivity and yield.
Q: What are the purity and yield improvements of this method?
A: By optimizing the acid concentration and minimizing byproduct formation through controlled sulfonation, this method achieves an amino G acid conversion rate of over 99.4% and improves the yield of Amino-K acid by approximately 2-3% compared to conventional processes, while significantly enhancing product selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino-K Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the global fine chemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the chlorosulfonic acid-assisted sulfonation can be seamlessly translated from the laboratory to full-scale manufacturing. We are committed to delivering high-purity Amino-K acid that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our technical expertise, we can help you navigate the complexities of this optimized synthesis, ensuring consistent quality and supply continuity for your dye and pigment applications.
We invite you to engage with our technical procurement team to discuss how this advanced production method can benefit your specific supply chain requirements. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs, and let us provide you with specific COA data and route feasibility assessments. Together, we can drive efficiency and sustainability in your intermediate sourcing strategy, securing a competitive edge in the marketplace.
