Scalable Synthesis of 3',4',5'-Trifluoro-2-Nitro-1,1'-Biphenyl via Advanced Palladium Catalysis
The global demand for high-performance fungicides continues to drive innovation in the synthesis of critical agrochemical intermediates. A pivotal development in this sector is disclosed in patent CN111606808A, which details a highly efficient synthetic method for 3',4',5'-trifluoro-2-nitro-1,1'-biphenyl. This compound serves as a vital building block for Fluxapyroxad, a broad-spectrum fungicide developed by BASF. The structural complexity of this molecule, featuring a polyfluorinated biphenyl backbone with a sensitive nitro group, presents significant challenges for traditional coupling methodologies. As illustrated below, the target molecule requires precise control over regioselectivity and functional group tolerance.
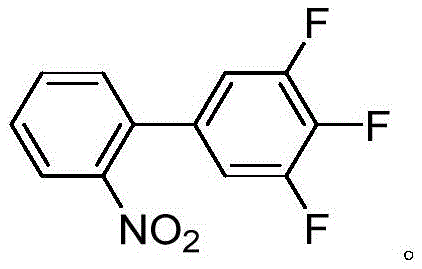
The significance of this patent lies in its ability to overcome the limitations of existing synthetic routes, offering a pathway that is not only chemically robust but also economically viable for large-scale production. By utilizing a Suzuki-Miyaura coupling strategy with novel palladium catalysts, the method achieves near-quantitative yields under remarkably mild conditions. For procurement managers and supply chain directors, this represents a shift towards more reliable sourcing of high-purity agrochemical intermediates, minimizing the risks associated with complex, multi-step syntheses that often suffer from yield erosion and impurity accumulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyfluorinated biphenyls has been plagued by reliance on sophisticated and often impractical catalytic systems. Prior art, such as PCT 2018/035685 A1, describes methods utilizing palladium acetate paired with extremely expensive ligands like 4-(9-anthryl)-3-tert-butyl-2,3-dihydrobenzo[d][1,3]oxaphospholane, which can cost upwards of 1500 yuan per 500 mg. Furthermore, other approaches, like those found in Chinese patent CN 105399635 B, depend on palladium/graphene catalysts that are not commercially available and require microwave irradiation at 100°C. These conditions are inherently difficult to scale; microwave reactors are rarely feasible for tonnage production, and air-sensitive catalysts necessitate costly inert atmosphere handling throughout the entire manufacturing process. Such constraints create bottlenecks in the supply chain, leading to higher costs and longer lead times for the final active pharmaceutical or agrochemical ingredients.
The Novel Approach
In stark contrast, the methodology presented in CN111606808A introduces a library of stable, commercially accessible N-heterocyclic carbene (NHC) palladium complexes, designated as Formulas (1) through (24). These catalysts exhibit exceptional stability and activity, allowing the reaction to proceed efficiently at temperatures ranging from 40°C to 100°C without the need for microwave assistance or extreme pressures. The versatility of this system is demonstrated by the wide array of ligand structures available, enabling fine-tuning of the electronic and steric environment around the palladium center to optimize the coupling of the electron-deficient 2-nitrochlorobenzene with 3,4,5-trifluorobenzeneboronic acid.
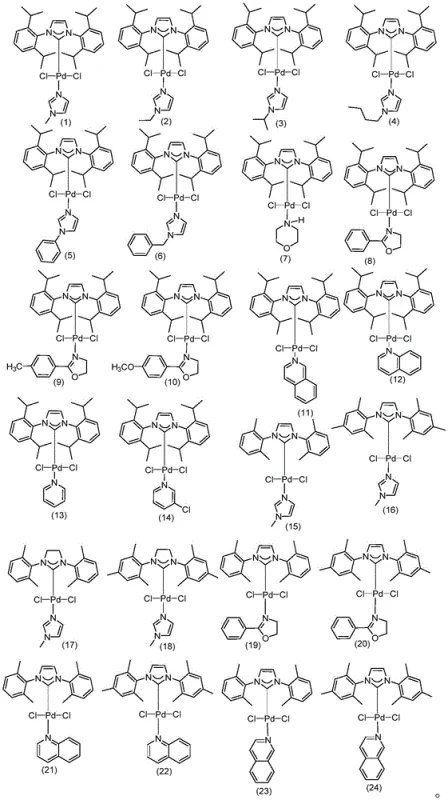
This novel approach drastically simplifies the operational workflow. The reaction can be conducted in a variety of solvents, including environmentally friendlier options like water mixed with alcohols or ethers, further reducing the environmental footprint and waste disposal costs. The ability to use inexpensive starting materials like 2-nitrochlorobenzene, combined with catalyst loadings as low as 0.000005 molar equivalents, translates directly into substantial cost reductions in agrochemical intermediate manufacturing. This shift from exotic, custom-synthesized catalysts to robust, off-the-shelf solutions is a game-changer for industrial scalability.
Mechanistic Insights into NHC-Pd Catalyzed Suzuki-Miyaura Coupling
The core of this technological breakthrough is the utilization of bulky, electron-rich N-heterocyclic carbene ligands coordinated to palladium. These ligands stabilize the active Pd(0) species, preventing the formation of inactive palladium black, which is a common failure mode in Suzuki couplings involving sterically hindered or electron-poor substrates. The catalytic cycle initiates with the oxidative addition of the aryl chloride (2-nitrochlorobenzene) to the Pd(0) center. The presence of the nitro group ortho to the chloride makes this step challenging due to electronic deactivation, but the strong sigma-donating capability of the NHC ligand facilitates this critical bond cleavage. Following oxidative addition, transmetallation occurs with the 3,4,5-trifluorobenzeneboronic acid, activated by the base present in the reaction mixture.
Impurity control is paramount in the synthesis of fungicide intermediates to ensure the efficacy and safety of the final crop protection product. The high selectivity of these NHC-Pd catalysts minimizes side reactions such as homocoupling of the boronic acid or dehalogenation of the nitro-chlorobenzene. The patent data indicates that liquid chromatography purity consistently exceeds 98%, often reaching >99% in optimized examples. This high level of purity is achieved because the catalyst system tolerates the sensitive nitro group without reduction, a common side reaction with less selective catalysts. Furthermore, the low catalyst loading reduces the burden on downstream purification processes, as there is less residual palladium to remove, thereby streamlining the path to high-purity biphenyl derivatives.
How to Synthesize 3',4',5'-Trifluoro-2-Nitro-1,1'-Biphenyl Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-yield results in a laboratory or pilot plant setting. The process is designed to be operationally simple, requiring standard glassware and heating equipment rather than specialized high-pressure reactors. By adhering to the specified molar ratios and temperature ranges, manufacturers can reliably produce the target intermediate with minimal batch-to-batch variation. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Under nitrogen or inert gas protection, mix 2-nitrochlorobenzene, 3,4,5-trifluorobenzeneboronic acid, a specific NHC-Pd catalyst (from formulas 1-24), an alkaline substance, and a reaction solvent.
- Heat the reaction mixture at a mild temperature range of 40-100°C for a duration of 3 to 24 hours to obtain the crude reaction solution.
- Perform post-treatment on the reaction liquid via extraction, concentration, or recrystallization to isolate the high-purity 3',4',5'-trifluoro-2-nitro-1,1'-biphenyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the implications of this patent are profound. The transition to this synthetic route addresses several critical pain points in the current supply chain for agrochemical intermediates. By eliminating the dependency on proprietary, non-commercial catalysts and expensive ligands, the overall cost structure of the manufacturing process is significantly optimized. The use of commodity chemicals like 2-nitrochlorobenzene ensures a stable supply of raw materials, insulating production from the volatility often seen with specialty reagents.
- Cost Reduction in Manufacturing: The economic benefits are driven primarily by the drastic reduction in catalyst cost and loading. Traditional methods often require stoichiometric or near-stoichiometric amounts of expensive reagents, whereas this method operates effectively with ppm-level catalyst loadings. Additionally, the elimination of microwave equipment and the ability to run reactions at atmospheric pressure reduce capital expenditure (CAPEX) on specialized reactor infrastructure. The simplified workup procedures, often involving simple filtration or crystallization rather than complex chromatography, further lower operational expenditures (OPEX) by reducing solvent consumption and processing time.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available catalysts (Formulas 1-24) and common solvents. Unlike methods relying on custom-synthesized ligands that may have long lead times or single-source suppliers, the reagents for this process are readily accessible from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the robustness of the reaction conditions means that manufacturing can be transferred between different sites or contract manufacturing organizations (CMOs) with greater ease, ensuring continuous availability of the intermediate for downstream formulation.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the mild reaction conditions (40-100°C) and the option to use water-containing solvent systems align well with green chemistry principles. Lower energy consumption for heating and cooling translates to a reduced carbon footprint. The high atom economy and selectivity of the reaction minimize the generation of hazardous waste streams, simplifying compliance with increasingly stringent environmental regulations. The process is inherently scalable from gram to multi-ton quantities without the engineering challenges associated with scaling microwave chemistry or handling pyrophoric catalysts, making it a sustainable choice for long-term commercial production.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the practical implementation of this synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development scientists.
Q: What are the key advantages of the catalysts described in CN111606808A compared to prior art?
A: Unlike prior art methods that require expensive, air-sensitive ligands or non-commercial palladium/graphene complexes, the catalysts in this patent (Formulas 1-24) are stable, commercially available, and effective at extremely low loadings (down to 0.000005 molar ratio), significantly reducing heavy metal residue and cost.
Q: What yields and purity levels can be expected from this synthesis method?
A: The patent reports that the highest yield of the product is basically quantitative, with typical experimental examples showing total yields ranging from 90% to 99% and liquid chromatography purity consistently exceeding 98%, making it highly suitable for pharmaceutical and agrochemical grade applications.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process operates under mild conditions (40-100°C) without the need for specialized equipment like microwave reactors or high-pressure vessels. The use of common solvents (including water mixtures) and inexpensive starting materials like 2-nitrochlorobenzene makes it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3',4',5'-Trifluoro-2-Nitro-1,1'-Biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation agrochemicals. Our technical team has thoroughly analyzed the synthetic route described in CN111606808A and is fully prepared to leverage this advanced catalytic technology for your production needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3',4',5'-trifluoro-2-nitro-1,1'-biphenyl meets the exacting standards required for fungicide synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this efficient catalytic method can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of this essential agrochemical intermediate.
