Advanced Catalyst-Free Aminomethylation for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
Advanced Catalyst-Free Aminomethylation for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks efficient, scalable, and cost-effective methodologies for constructing complex nitrogen-containing heterocycles, which serve as the backbone for numerous bioactive molecules. A groundbreaking approach detailed in patent CN109761975B introduces a novel synthetic pathway for N-((2-heteroarylimidazo-heteroaryl-3-yl)methyl)benzenesulfonamide compounds, addressing critical bottlenecks in traditional C-H functionalization. This technology leverages a unique oxidative coupling strategy that bypasses the need for precious metal catalysts, utilizing instead a readily available radical generation system involving di-tert-butyl peroxide and potassium permanganate. For R&D directors and process chemists, this represents a paradigm shift towards greener, more economical synthesis of key pharmaceutical intermediates used in the development of treatments for insomnia, anxiety, and HIV. The structural versatility of these imidazoheteroaryl scaffolds makes them indispensable building blocks, and the ability to install aminomethyl groups directly via sp2-sp3 coupling opens new avenues for medicinal chemistry optimization.
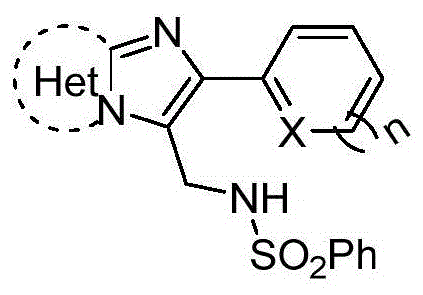
Furthermore, the operational simplicity of conducting these reactions in an air environment rather than under stringent inert atmospheres drastically lowers the barrier for commercial adoption. By employing hexafluoroisopropanol (HFIP) and methanol as a synergistic solvent system, the protocol achieves high conversion rates while maintaining a clean impurity profile, which is paramount for regulatory compliance in API manufacturing. This patent not only enriches the functionalization toolbox for imidazo[1,2-a]pyridine derivatives but also establishes a robust platform for the late-stage modification of complex drug candidates. As we delve deeper into the technical specifics, it becomes evident that this methodology offers substantial advantages in terms of supply chain reliability and production cost efficiency, making it a highly attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of aminomethyl subunits onto heteroaromatic cores has been fraught with significant synthetic challenges, often relying on pre-functionalized starting materials or harsh reaction conditions that limit scalability. Traditional cross-coupling strategies frequently necessitate the use of expensive palladium or copper catalysts, which not only inflate the raw material costs but also introduce the risk of toxic heavy metal residues that require rigorous and costly removal steps to meet pharmacopeial standards. Moreover, many conventional aminomethylation protocols demand strictly anhydrous and oxygen-free environments, requiring specialized equipment and increasing the operational complexity and energy consumption of the manufacturing process. The reliance on unstable organometallic reagents, such as alpha-trifluoroboronic acids or alpha-silylamines, further complicates the supply chain due to their sensitivity to moisture and limited shelf life. These factors collectively contribute to prolonged lead times and elevated production costs, creating a bottleneck for the rapid development and commercialization of new therapeutic agents based on imidazoheteroaryl scaffolds.
The Novel Approach
In stark contrast, the methodology disclosed in CN109761975B presents a streamlined, catalyst-free alternative that utilizes simple, commodity chemicals to achieve direct C-H aminomethylation with remarkable efficiency. By employing di-tert-butyl peroxide (DTBP) as a radical initiator and potassium permanganate as an oxidant in the presence of sodium tert-butoxide, the reaction proceeds smoothly under aerobic conditions, eliminating the need for expensive transition metals and inert gas protection. The use of methanol serves a dual purpose as both a solvent and a carbon source for the methylene bridge, representing a significant innovation in atom economy and resource utilization. This approach not only simplifies the reaction setup but also enhances safety profiles by avoiding pyrophoric reagents, thereby facilitating easier scale-up from laboratory to pilot plant operations. The compatibility of this system with a wide range of heterocyclic substrates, including thiophene and furan derivatives, underscores its versatility and potential for broad application in the synthesis of diverse pharmaceutical intermediates.
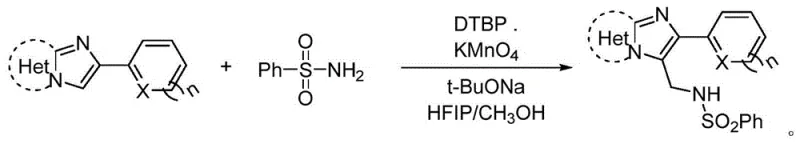
Mechanistic Insights into Oxidative Radical Aminomethylation
The mechanistic underpinning of this transformation involves a sophisticated interplay of radical species generated in situ, which facilitates the challenging sp2-sp3 carbon-carbon bond formation at the C3 position of the imidazoheteroaryl ring. The reaction is initiated by the thermal decomposition of di-tert-butyl peroxide, generating tert-butoxy radicals that abstract hydrogen atoms from the methanol solvent to produce hydroxymethyl radicals. These reactive intermediates are subsequently oxidized by potassium permanganate to form formaldehyde or equivalent electrophilic species, which then undergo nucleophilic attack by the benzenesulfonamide nitrogen. The resulting N-sulfonyl hemiaminal intermediate is further activated by the basic conditions provided by sodium tert-butoxide, leading to the elimination of water and the formation of an N-sulfonyl imine. Finally, a radical addition of the imidazoheteroaryl species to this imine, followed by rearomatization, yields the desired N-((2-heteroarylimidazo-heteroaryl-3-yl)methyl)benzenesulfonamide product. This intricate cascade highlights the elegance of using simple oxidants to drive complex molecular constructions without the need for external catalytic cycles.
From an impurity control perspective, the choice of HFIP as a co-solvent plays a critical role in stabilizing radical intermediates and suppressing side reactions that could lead to over-oxidation or polymerization. The specific ratio of HFIP to methanol allows for fine-tuning of the reaction polarity and hydrogen-bonding network, which is essential for maintaining high selectivity towards the mono-aminomethylated product. Analytical data from the patent examples, such as the synthesis of N-((2-(thiophen-2-yl)imidazo[1,2-a]pyridin-3-yl)methyl)benzenesulfonamide, demonstrates excellent purity profiles with yields reaching up to 74% under optimized conditions. The absence of metal catalysts inherently reduces the complexity of the impurity spectrum, simplifying downstream purification processes such as column chromatography or crystallization. This level of control is vital for ensuring consistent batch-to-batch quality, a key requirement for any reliable pharmaceutical intermediate supplier serving the global market.
![Structure of N-((2-(thiophen-2-yl)imidazo[1,2-a]pyridin-3-yl)methyl)benzenesulfonamide obtained with 73% yield](/insights/img/imidazoheteroaryl-benzenesulfonamide-synthesis-pharma-supplier-20260305125049-05.png)
How to Synthesize N-((2-Heteroarylimidazo-Heteroaryl-3-Yl)Methyl)Benzenesulfonamide Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal parameters to maximize yield and minimize byproduct formation. The standard protocol involves charging a reaction vessel with the 2-heteroarylimidazoheteroaryl substrate and benzenesulfonamide in a molar ratio of approximately 1:2 to 1:3, ensuring an excess of the sulfonamide to drive the equilibrium forward. Following the addition of the oxidant system comprising DTBP, KMnO4, and t-BuONa, the mixture is heated to a temperature range of 100°C to 140°C for a duration of 8 to 12 hours. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Combine 2-heteroarylimidazoheteroaryl substrate and benzenesulfonamide in a reaction vessel under ambient air conditions.
- Add oxidants and base including di-tert-butyl peroxide, potassium permanganate, and sodium tert-butoxide, followed by HFIP/methanol solvent mixture.
- Heat the reaction mixture to 100-140°C for 8-12 hours, then perform chromatographic separation and drying to isolate the target sulfonamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free methodology translates into tangible economic benefits and enhanced operational resilience. The elimination of precious metal catalysts removes a significant cost driver from the bill of materials, while also mitigating the supply risks associated with fluctuating prices of palladium or rhodium. Furthermore, the use of commodity chemicals like potassium permanganate and di-tert-butyl peroxide ensures a stable and secure supply chain, as these reagents are produced in large volumes globally and are not subject to the same geopolitical constraints as rare earth elements. The ability to run reactions in air rather than under nitrogen or argon atmospheres reduces utility costs related to gas consumption and equipment maintenance, contributing to overall cost reduction in pharmaceutical intermediate manufacturing. These factors combined make the process highly attractive for large-scale production where margin optimization is critical.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts fundamentally alters the cost structure of the synthesis, eliminating the need for expensive ligands and the subsequent metal scavenging steps that are often required to meet regulatory limits. This simplification of the downstream processing workflow leads to substantial cost savings in terms of both materials and labor, as fewer unit operations are required to achieve the final purity specifications. Additionally, the high atom economy achieved by using methanol as a C1 source minimizes waste generation, further reducing disposal costs and environmental fees associated with chemical manufacturing. The overall process efficiency allows for competitive pricing strategies without compromising on the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: By relying on widely available industrial chemicals rather than specialized organometallic reagents, manufacturers can significantly reduce lead times and avoid disruptions caused by supplier shortages. The robustness of the reaction conditions, which tolerate air and moisture, means that production can proceed with less stringent environmental controls, reducing the risk of batch failures due to minor deviations in atmospheric conditions. This reliability is crucial for maintaining continuous supply to downstream API producers, ensuring that drug development timelines are met without delay. The stability of the reagents also allows for longer storage periods, enabling strategic stockpiling to buffer against market volatility.
- Scalability and Environmental Compliance: The simplicity of the reaction setup facilitates seamless scale-up from gram to kilogram and ton scales, as the heat transfer and mixing requirements are less demanding compared to sensitive catalytic systems. The use of HFIP and methanol, while requiring appropriate recovery systems, aligns with modern green chemistry principles by reducing the reliance on chlorinated solvents often used in traditional coupling reactions. The absence of heavy metals simplifies wastewater treatment and waste solid disposal, helping facilities meet increasingly stringent environmental regulations. This alignment with sustainability goals enhances the corporate social responsibility profile of the manufacturing site, appealing to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and practical application scenarios. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production lines or new product development projects. The answers reflect the specific advantages and operational parameters defined in the intellectual property documentation.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method described in CN109761975B operates without transition metal catalysts, utilizing a cost-effective system of di-tert-butyl peroxide and potassium permanganate, which significantly reduces raw material costs and eliminates heavy metal residue concerns.
Q: What are the reaction conditions regarding atmosphere and temperature?
A: The process is remarkably robust, proceeding efficiently in an air environment without the need for inert gas protection. The reaction typically runs at temperatures between 100°C and 140°C for 8 to 12 hours, facilitating easier operational handling compared to sensitive anaerobic protocols.
Q: Is the substrate scope limited to specific heterocycles?
A: The method demonstrates broad applicability across various heterocyclic systems, successfully functionalizing imidazo[1,2-a]pyridines and imidazo[2,1-b]thiazoles substituted with thiophene, furan, or phenyl groups, ensuring versatility for diverse drug discovery pipelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-((2-Heteroarylimidazo-Heteroaryl-3-Yl)Methyl)Benzenesulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free aminomethylation technology for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-((2-heteroarylimidazo-heteroaryl-3-yl)methyl)benzenesulfonamide delivered meets the highest international standards for safety and efficacy. We are committed to leveraging our technical expertise to optimize this process further, tailoring it to your specific volume and quality requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits specific to your project scope. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your drug development pipeline while optimizing your supply chain costs. Let us be your partner in turning complex chemical challenges into commercial successes.
