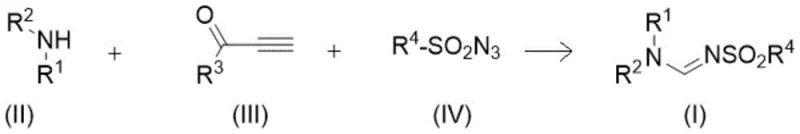
Advanced Silver-Catalyzed Synthesis of Sulfonyl Formamidine Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic methodologies that can deliver complex nitrogen-containing heterocycles with high efficiency and minimal environmental impact. A significant breakthrough in this domain is detailed in patent CN113444056A, which discloses a novel preparation method for sulfonyl formamidine derivatives. This technology leverages a sophisticated silver-catalyzed multicomponent reaction strategy, transforming simple starting materials into high-value intermediates in a single operational step. For R&D directors and process chemists, this represents a paradigm shift from traditional multi-step syntheses to a streamlined, atom-economical process. The ability to construct the sulfonyl formamidine scaffold directly from an amine, a terminal alkynone, and a sulfonyl azide not only simplifies the synthetic route but also drastically reduces the generation of chemical waste. As a leading entity in fine chemical manufacturing, we recognize this methodology as a critical enabler for the rapid development of new active pharmaceutical ingredients (APIs) and crop protection agents, offering a reliable pathway to access these structurally diverse molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amidine and formamidine derivatives has been plagued by significant operational challenges that hinder efficient commercial production. Traditional routes often require the pre-functionalization of starting materials, necessitating multiple isolation and purification steps that accumulate impurities and drive up manufacturing costs. Many prior art methods rely on harsh reaction conditions, such as strong acids or bases and elevated temperatures, which can lead to the decomposition of sensitive functional groups commonly found in drug candidates. Furthermore, the lack of regioselectivity in older protocols frequently results in complex mixtures of isomers, requiring extensive and costly chromatographic separation to achieve the necessary purity standards for pharmaceutical applications. These inefficiencies create bottlenecks in the supply chain, extending lead times and increasing the overall cost of goods sold (COGS) for downstream manufacturers who rely on these key intermediates for their final products.
The Novel Approach
In stark contrast, the methodology described in the patent introduces a highly efficient one-pot three-component coupling reaction that circumvents the drawbacks of legacy techniques. By utilizing a silver catalyst to mediate the reaction between an amine compound, a terminal alkynone, and a sulfonyl azide, the process achieves remarkable selectivity and yield under relatively mild conditions. This approach eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption and processing time. The versatility of this method is evidenced by its tolerance to a wide range of substituents, including halogens, alkoxy groups, and various alkyl chains, allowing for the rapid generation of diverse chemical libraries for structure-activity relationship (SAR) studies. For procurement managers, this translates to a more predictable supply of high-quality intermediates, while for supply chain heads, it意味着 a reduction in the complexity of logistics and inventory management associated with multi-step synthetic sequences.
Mechanistic Insights into Silver-Catalyzed Multicomponent Coupling
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the silver catalyst. The reaction initiates with the nucleophilic addition of the amine compound to the terminal alkynone, forming an enamine intermediate in situ. This enamine then undergoes a 1,3-dipolar cycloaddition with the sulfonyl azide, a step that is critically dependent on the electronic activation provided by the silver species. The resulting triazole intermediate is inherently unstable under the reaction conditions and spontaneously undergoes a ring-opening rearrangement to yield the final N-sulfonyl formamidine derivative. It is crucial to note that the terminal alkynone serves solely as a carbon source in this transformation, highlighting the atom economy of the process. The choice of silver salt plays a pivotal role; soluble silver compounds like silver trifluoroacetate (AgTFA) have shown superior catalytic activity compared to insoluble variants, likely due to better homogeneity and coordination with the reactive species in the solution phase.
Understanding the impurity profile is essential for ensuring the quality of the final product. The high selectivity of this silver-catalyzed system minimizes the formation of side products such as polymerized alkynes or over-oxidized species that are common in non-catalyzed thermal reactions. The mechanism ensures that the sulfonyl group is transferred cleanly to the nitrogen center without scrambling or degradation. This precise control over the reaction trajectory allows for the production of sulfonyl formamidines with purities consistently exceeding 96% as measured by HPLC, meeting the stringent requirements for pharmaceutical intermediates. For R&D teams, this mechanistic clarity provides confidence in scaling the process, as the reaction parameters are well-defined and reproducible across different batches and substrate scopes.
How to Synthesize Sulfonyl Formamidine Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction conditions to maximize yield and purity. The general protocol involves dissolving the amine, terminal alkynone, and sulfonyl azide in a polar organic solvent, with acetonitrile being the preferred medium due to its ability to solubilize both the reactants and the silver catalyst effectively. The molar ratio of the amine to the alkynone and azide is typically maintained between 1:1 and 1:3, ensuring complete conversion of the limiting reagent. The reaction temperature can be tuned from room temperature (25°C) to 120°C depending on the reactivity of the specific substrates involved, with reaction times ranging from 1 to 24 hours. Detailed standardized operating procedures for this synthesis are provided below to guide process engineers in replicating these results.
- Mix amine compound, terminal alkynone, and sulfonyl azide in an organic solvent such as acetonitrile with a silver catalyst.
- Stir the reaction mixture at temperatures between 25°C and 120°C for 1 to 24 hours to facilitate the multicomponent coupling.
- Perform post-treatment via extraction with ethyl acetate and water, followed by silica gel column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this silver-catalyzed methodology offers substantial strategic advantages for organizations focused on cost reduction in pharmaceutical intermediates manufacturing. The elimination of multiple synthetic steps directly correlates to a significant decrease in raw material consumption, energy usage, and labor costs associated with intermediate handling and purification. By consolidating the synthesis into a single pot, manufacturers can reduce the footprint of their production facilities and minimize the volume of hazardous waste generated, aligning with modern green chemistry principles and environmental regulations. This efficiency gain is not merely theoretical; the high yields reported in the patent data, often surpassing 90%, indicate a robust process that maximizes the output from every kilogram of input material, thereby improving the overall margin profile for the final API or agrochemical product.
- Cost Reduction in Manufacturing: The use of commercially available silver catalysts, which can often be recovered or used in low loadings, avoids the expense of exotic transition metals or complex ligand systems. Furthermore, the simplified workup procedure, which typically involves standard extraction and crystallization or column chromatography, removes the need for specialized equipment or tedious distillation processes. This streamlining of the downstream processing phase results in drastic savings in operational expenditures (OPEX) and allows for a more competitive pricing structure for the final sulfonyl formamidine derivatives supplied to the market.
- Enhanced Supply Chain Reliability: The starting materials for this reaction—amines, terminal alkynones, and sulfonyl azides—are widely available commodity chemicals with established global supply chains. This abundance mitigates the risk of supply disruptions that often plague processes relying on bespoke or hard-to-source reagents. For supply chain heads, this means greater predictability in procurement planning and the ability to secure long-term contracts for raw materials at stable prices. The robustness of the reaction conditions also implies that the process can be executed in various geographic locations without requiring highly specialized infrastructure, further diversifying the supply base and reducing geopolitical risks.
- Scalability and Environmental Compliance: The scalability of this process is supported by its exothermic profile, which is manageable under standard cooling conditions, and the absence of highly toxic gases or unstable intermediates that would require containment systems. The high atom economy ensures that the majority of the mass of the reactants ends up in the desired product, minimizing the burden on waste treatment facilities. This environmental compatibility facilitates easier regulatory approval for new manufacturing sites and supports the sustainability goals of multinational corporations seeking to reduce their carbon footprint while maintaining high production volumes of complex fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this sulfonyl formamidine synthesis technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for process chemists and project managers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the optimal catalyst for this sulfonyl formamidine synthesis?
A: According to the patent data, silver trifluoroacetate (AgTFA) demonstrates the highest catalytic efficiency, although other silver salts like silver acetate and silver chloride are also effective.
Q: Can this method be scaled for industrial production of drug intermediates?
A: Yes, the process utilizes readily available raw materials and simple post-treatment steps like extraction and chromatography, making it highly suitable for commercial scale-up.
Q: What represents the typical purity and yield of the final product?
A: The method consistently achieves high yields ranging from 70% to 98% with product purities exceeding 96% as determined by HPLC analysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonyl Formamidine Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure required to translate this innovative patent technology into commercial reality. Our team of experienced chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of sulfonyl formamidine derivative meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate use in downstream synthetic applications without the need for additional repurification.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data for our available catalog compounds and to request comprehensive route feasibility assessments for your custom synthesis projects. Let us collaborate to accelerate your drug development timelines and optimize your supply chain efficiency with our premium chemical solutions.
