Advanced Synthesis of Critical Mesotrione Impurity for Global Agrochemical Compliance
The agrochemical industry faces increasingly stringent regulatory hurdles regarding impurity profiling, particularly for high-volume herbicides like Mesotrione. Patent CN112939922B, published in early 2023, introduces a pivotal breakthrough in the synthesis of 1-cyano-6-methylsulfonyl-7-nitro-9H-xanthene-9-ketone, a critical toxic impurity associated with Mesotrione production. This technical disclosure addresses a significant gap in the supply chain for regulatory reference standards, offering a robust, scalable, and economically viable pathway that replaces unfeasible extraction methods. For R&D directors and quality assurance teams, the ability to synthesize this specific xanthene derivative with high fidelity is essential for meeting European standards which mandate impurity levels below 0.0002% (w/w). The patent outlines a concise five-step sequence starting from inexpensive commodity chemicals, demonstrating a clear commitment to process efficiency and cost containment without compromising structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of specific process-related impurities for agrochemical registration has been plagued by significant logistical and technical challenges. In the case of Mesotrione, the target impurity, 1-cyano-6-methylsulfonyl-7-nitro-9H-xanthene-9-ketone, is formed in such minute quantities during standard manufacturing that isolation via extraction is technically impractical and economically prohibitive. Attempting to isolate trace components from tons of crude active ingredient requires massive solvent volumes, complex chromatographic separations, and yields negligible amounts of the target material. Furthermore, relying on extraction introduces variability in purity and isotopic composition, which can compromise the accuracy of analytical methods used for batch release testing. This bottleneck often delays regulatory filings and complicates the supply of certified reference materials needed for global market access.
The Novel Approach
The methodology described in patent CN112939922B circumvents these extraction bottlenecks by constructing the molecule de novo from readily available precursors. The strategy leverages 2-chloro-4-fluoro-5-nitrobenzoic acid as a foundational building block, utilizing its inherent reactivity to install the necessary functional groups in a logical, stepwise fashion. By shifting from an isolation paradigm to a constructive synthesis paradigm, the process guarantees a consistent supply of the impurity independent of Mesotrione production batches. This approach not only secures the supply chain for reference standards but also allows for the optimization of each chemical transformation to maximize yield and purity. The route is characterized by mild reaction conditions in the early stages and robust coupling strategies in the later stages, ensuring that the complex xanthene core is assembled with precision.
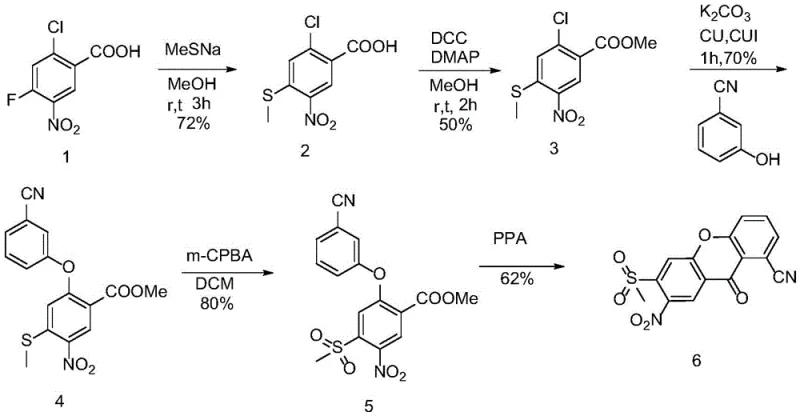
Mechanistic Insights into the Multi-Step Synthetic Cascade
The synthetic pathway initiates with a nucleophilic aromatic substitution where the fluorine atom of the starting benzoic acid is displaced by a methylthio group using sodium methyl mercaptide. This transformation is highly selective and proceeds efficiently at room temperature in methanol, establishing the sulfur handle required for the final sulfone moiety. Following this, the carboxylic acid is protected as a methyl ester using DCC and DMAP, a standard yet effective activation strategy that prevents interference during subsequent coupling reactions. The core structural complexity is introduced in the third step via a copper-catalyzed Ullmann-type coupling. Here, the chlorine atom on the aromatic ring is displaced by 3-hydroxybenzonitrile, forging the critical diaryl ether linkage that defines the xanthene scaffold. This step requires careful thermal management at 120-130°C to overcome the activation energy barrier for C-O bond formation.
The final structural refinement involves two critical transformations: oxidation and cyclization. The methyl sulfide intermediate is oxidized to the corresponding sulfone using m-chloroperoxybenzoic acid (m-CPBA) in dichloromethane. This oxidation state change is crucial as the electron-withdrawing sulfone group influences the electronic density of the aromatic ring, facilitating the final ring closure. The synthesis concludes with an intramolecular Friedel-Crafts acylation mediated by concentrated sulfuric acid. Under elevated temperatures, the ester carbonyl attacks the activated aromatic ring, closing the tricyclic xanthene-9-one system. This cascade demonstrates a sophisticated understanding of electronic effects, where the nitro and sulfone groups direct the cyclization to the correct position, ensuring the formation of the desired regioisomer essential for accurate impurity profiling.
How to Synthesize 1-Cyano-6-Methylsulfonyl-7-Nitro-9H-Xanthene-9-Ketone Efficiently
Executing this synthesis requires precise control over reaction parameters to maintain high purity suitable for analytical standards. The process begins with the dissolution of the starting acid in methanol followed by the batch addition of the thiolate nucleophile, requiring strict pH control during workup to isolate the intermediate acid. Subsequent steps involve standard organic operations such as spin-drying and column chromatography, which are effective at the laboratory scale for removing side products like urea derivatives from the DCC coupling or unreacted phenols from the coupling step. The oxidation step demands temperature control below 0°C initially to manage the exotherm of the peracid reaction, ensuring safety and selectivity.
- Perform nucleophilic substitution on 2-chloro-4-fluoro-5-nitrobenzoic acid using sodium methyl mercaptide in methanol to obtain Compound 2.
- Esterify Compound 2 using DCC and DMAP in methanol to form the methyl ester Compound 3.
- Execute a copper-catalyzed coupling reaction between Compound 3 and 3-hydroxybenzonitrile to generate the ether linkage in Compound 4.
- Oxidize the methyl sulfide group in Compound 4 to a sulfone using m-CPBA in DCM to yield Compound 5.
- Conduct an intramolecular cyclization of Compound 5 using sulfuric acid at elevated temperatures to finalize the xanthene-9-ketone structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits over reliance on extraction or complex custom synthesis from exotic precursors. The primary advantage lies in the utilization of 2-chloro-4-fluoro-5-nitrobenzoic acid, a commodity chemical that is widely available and cost-effective, significantly reducing the raw material cost basis. By avoiding the need for specialized, high-cost catalysts or cryogenic conditions in the initial steps, the process minimizes energy consumption and equipment requirements. The modular nature of the synthesis allows for flexible production scheduling, where intermediates can be stockpiled if necessary, enhancing supply chain resilience against demand fluctuations in the agrochemical sector.
- Cost Reduction in Manufacturing: The elimination of expensive extraction processes and the use of common solvents like methanol and dichloromethane drastically lower the operational expenditure. The route avoids the use of precious metal catalysts in the coupling step, opting instead for economical copper powder and copper iodide, which translates to direct savings in catalyst procurement and waste disposal costs. Furthermore, the high selectivity of the reactions reduces the burden on downstream purification, minimizing solvent usage and waste generation associated with extensive recrystallization or chromatography.
- Enhanced Supply Chain Reliability: Dependence on a synthetic route rather than extraction decouples the supply of this critical impurity from the production volume of Mesotrione itself. This independence ensures that reference standards can be produced on demand, regardless of the seasonal manufacturing cycles of the herbicide. The robustness of the chemistry, particularly the room temperature operations in the first two steps, reduces the risk of batch failures due to equipment malfunction, thereby guaranteeing consistent lead times for customers requiring urgent regulatory documentation.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to industrial reactors. The workup procedures, involving simple pH adjustments and phase separations, are amenable to continuous processing or large-batch operations. Additionally, the avoidance of heavy metals other than copper, which is easier to remove and recycle than palladium or platinum, simplifies compliance with environmental regulations regarding heavy metal residues in fine chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized xanthene derivative. These insights are derived directly from the experimental data and process descriptions found in the patent literature, ensuring accuracy for technical decision-makers. Understanding these nuances is vital for integrating this material into your quality control workflows.
Q: Why is the synthesis of 1-cyano-6-methylsulfonyl-7-nitro-9H-xanthene-9-ketone necessary?
A: This compound is a known toxic impurity generated during the production of Mesotrione, a widely used corn field herbicide. Regulatory bodies, particularly in Europe, mandate strict control limits (less than 0.0002% w/w) for this impurity. Since natural extraction from the production process is unfeasible due to trace levels, a dedicated synthetic route is required to produce reference standards for quality control and registration dossiers.
Q: What are the key advantages of the patented synthesis route over traditional extraction methods?
A: Traditional extraction is impossible because the impurity exists in negligible quantities within the crude Mesotrione mixture. The patented method utilizes cheap, commercially available starting materials like 2-chloro-4-fluoro-5-nitrobenzoic acid. The route is designed to be simple and economic, avoiding complex purification steps associated with isolating trace components from bulk API production, thereby ensuring a reliable supply of high-purity reference material.
Q: How does the process ensure high purity for analytical standards?
A: The synthesis employs distinct purification strategies at each stage, including pH adjustment, filtration, and column chromatography. The stepwise construction of the molecule allows for the removal of intermediates and by-products before the final cyclization. This controlled approach ensures that the final 1-cyano-6-methylsulfonyl-7-nitro-9H-xanthene-9-ketone meets the rigorous purity specifications required for impurity profiling and regulatory submission.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Cyano-6-Methylsulfonyl-7-Nitro-9H-Xanthene-9-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurity standards play in the global registration of agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for R&D screening or large-scale QC validation. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our materials meet the exacting standards required by international regulatory bodies.
We invite you to collaborate with us to secure your supply chain for Mesotrione impurity standards. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis of 1-cyano-6-methylsulfonyl-7-nitro-9H-xanthene-9-ketone can support your compliance goals efficiently and economically.
