Advanced Synthesis of Disubstituted Perfluoroalkyl Phthalonitriles for High-Performance Fluorine Catalysts
Advanced Synthesis of Disubstituted Perfluoroalkyl Phthalonitriles for High-Performance Fluorine Catalysts
The chemical industry is witnessing a paradigm shift towards fluorous phase chemistry, driven by the demand for separable and reusable catalytic systems that align with green chemistry principles. Patent CN103864645A represents a significant technological breakthrough in this domain by disclosing a robust method for synthesizing disubstituted perfluoroalkyl phthalonitrile compounds. These specialized intermediates serve as the critical precursors for generating perfluoroalkyl phthalocyanines, which function as novel fluorous phase catalysts and functional dye materials with exceptional thermal and chemical stability. The innovation lies not merely in the final molecule but in the efficient construction of the carbon-fluorine rich scaffold using a copper-mediated coupling strategy that bypasses the limitations of traditional fluorination techniques. For R&D directors and procurement specialists alike, this patent outlines a pathway to access high-value fluorinated building blocks that were previously difficult to manufacture at scale with consistent purity.
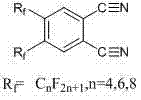
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of perfluoroalkyl-substituted aromatic compounds has been plagued by harsh reaction conditions and poor selectivity, often requiring aggressive fluorinating agents that compromise sensitive functional groups like nitriles. Conventional routes to phthalocyanine derivatives frequently involve post-synthetic modification, where the fluorinated chains are attached after the macrocycle formation, a process that is sterically hindered and yields are notoriously low. Furthermore, traditional methods often rely on expensive noble metal catalysts or generate substantial amounts of hazardous waste due to the use of volatile organic solvents that are difficult to recover from fluorous products. The inability to easily separate the catalyst from the product in homogeneous systems has also been a persistent bottleneck, leading to metal contamination in the final API or electronic material, which necessitates costly and time-consuming purification steps that erode profit margins.
The Novel Approach
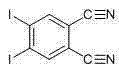
The methodology described in the patent introduces a streamlined, direct coupling reaction that utilizes 4,5-diiodophthalonitrile and perfluoroalkyl iodides as primary raw materials, mediated by activated copper powder in an anhydrous DMSO system. This approach fundamentally alters the synthetic landscape by enabling the direct installation of perfluoroalkyl chains (where n=4, 6, 8) onto the phthalonitrile core prior to cyclization, ensuring precise control over the substitution pattern. The use of activated copper powder acts as a cost-effective alternative to palladium or nickel catalysts, significantly reducing the raw material costs while maintaining high catalytic efficiency for the carbon-carbon bond formation. By conducting the reaction at moderate temperatures of 110 to 115 degrees Celsius under nitrogen protection, the process minimizes thermal degradation of the reactants and ensures a clean reaction profile that simplifies downstream processing.
Mechanistic Insights into Copper-Catalyzed Coupling
The success of this synthesis hinges on the meticulous activation of the copper catalyst, which transforms inert metallic copper into a highly reactive species capable of initiating the coupling cycle. The patent details a specific activation protocol where copper powder is treated with an iodo-acetone solution to form cupric iodide in situ, followed by dissolution of the oxide layer using a hydrochloric acid-acetone mixture. This dual-treatment process ensures that the copper surface is free from passivating oxides, exposing fresh active sites that facilitate the oxidative addition of the perfluoroalkyl iodide. The resulting organocopper intermediate then undergoes transmetallation or radical recombination with the aryl iodide moiety of the phthalonitrile, forging the critical C-C bond that links the fluorous tail to the aromatic head. This mechanistic pathway is particularly advantageous because it avoids the formation of stable copper-fluorine bonds that could otherwise deactivate the catalyst, thereby sustaining the catalytic turnover throughout the 4-hour reaction window.
Impurity control is inherently built into this mechanism through the choice of solvent and the stoichiometry of the reagents. The use of anhydrous DMSO, refined via reflux with calcium oxide and distillation, eliminates trace water that could hydrolyze the nitrile groups or quench the reactive copper species. Furthermore, the molar ratio of diiodophthalonitrile to activated copper powder is maintained at 1:6, providing a large excess of catalyst surface area to drive the reaction to completion and minimize the presence of mono-substituted byproducts. The subsequent purification via column chromatography effectively separates the desired disubstituted product from any unreacted starting materials or homocoupled side products, ensuring that the final intermediate meets the stringent purity specifications required for downstream phthalocyanine synthesis. This level of control over the impurity profile is critical for R&D teams aiming to produce consistent batches of fluorous catalysts with reproducible performance characteristics.
How to Synthesize 4,5-Bis-perfluoroalkyl Phthalonitrile Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for manufacturing these high-value intermediates, emphasizing the importance of reagent preparation and atmospheric control. The process begins with the rigorous activation of copper powder and the drying of DMSO, setting the stage for a moisture-sensitive coupling reaction that proceeds under an inert nitrogen atmosphere. Operators must carefully monitor the temperature ramp to 110-115 degrees Celsius to balance reaction kinetics with thermal stability, followed by a straightforward aqueous workup and ether extraction to isolate the crude product. While the patent provides specific gram-scale examples, the underlying chemistry is robust enough to be adapted for larger scale production with appropriate engineering controls for solvent handling and heat dissipation.
- Activate copper powder by treating with iodo-acetone solution followed by hydrochloric acid-acetone washing to remove oxide layers, then dry under vacuum.
- Perform the coupling reaction by mixing 4,5-diiodophthalonitrile, activated copper powder, and perfluoroalkyl iodide in anhydrous DMSO under nitrogen protection at 110-115°C for 4 hours.
- Quench the reaction with ice water, extract the product with ether, concentrate the organic layer, and purify the final yellow solid via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed synthesis route offers tangible strategic advantages that extend beyond mere technical feasibility. The shift from noble metal catalysts to activated copper powder represents a fundamental decoupling from the volatile pricing dynamics of precious metals, thereby stabilizing the cost structure of the manufacturing process. Additionally, the use of commodity chemicals like 4,5-diiodophthalonitrile and perfluoroalkyl iodides ensures a reliable supply chain, as these raw materials are produced by multiple global vendors, reducing the risk of single-source bottlenecks. The simplified workup procedure, which relies on standard filtration and extraction techniques rather than complex distillation or crystallization trains, translates to reduced processing time and lower energy consumption per kilogram of product. These factors collectively contribute to a more resilient and cost-efficient supply chain capable of meeting the fluctuating demands of the electronic and specialty chemical markets.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or platinum catalysts in favor of activated copper powder drastically reduces the direct material costs associated with the synthesis. Furthermore, the ability to recycle the solvent system and the simplicity of the purification process minimize waste disposal fees and operational overhead. By avoiding the need for specialized high-pressure equipment often required for direct fluorination, capital expenditure for plant setup is also significantly lowered, allowing for faster ROI on production assets.
- Enhanced Supply Chain Reliability: Sourcing strategies are strengthened by the reliance on widely available bulk chemicals rather than bespoke reagents that may have long lead times. The robustness of the reaction conditions means that production schedules are less susceptible to delays caused by sensitive parameter deviations, ensuring consistent on-time delivery to downstream customers. This reliability is paramount for clients in the semiconductor and display industries who require uninterrupted supplies of high-purity precursors to maintain their own manufacturing continuity.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of standard batch reactor technology and the absence of hazardous gaseous reagents. The environmental footprint is reduced through the use of DMSO, which has a high boiling point and low volatility, minimizing VOC emissions compared to traditional chlorinated solvents. Moreover, the copper residues can be managed through standard wastewater treatment protocols, ensuring compliance with increasingly stringent environmental regulations without the need for exotic remediation technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these disubstituted perfluoroalkyl phthalonitriles. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a transparent view of the technology's capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this material into their existing product portfolios or research pipelines.
Q: Why is activated copper powder critical for this synthesis?
A: Activated copper powder is essential because standard copper powder often possesses a surface oxide layer that inhibits the initiation of the coupling reaction. The patent specifies a rigorous activation protocol involving iodo-acetone and hydrochloric acid treatment to generate fresh, reactive cuprous species in situ, ensuring high conversion rates of the diiodophthalonitrile substrate.
Q: What is the role of DMSO in the reaction mechanism?
A: Dimethyl sulfoxide (DMSO) serves as a high-boiling polar aprotic solvent that stabilizes the transition states during the nucleophilic substitution or radical coupling process. Its ability to dissolve both the organic diiodide and the perfluoroalkyl iodide while maintaining stability at elevated temperatures (110-115°C) is crucial for driving the reaction to completion without requiring high-pressure equipment.
Q: How does this method improve upon traditional phthalocyanine precursor synthesis?
A: Traditional methods often struggle to introduce long perfluoroalkyl chains directly onto the phthalonitrile core without degrading the sensitive nitrile groups. This novel approach utilizes a robust Ullmann-type coupling that tolerates the nitrile functionality, allowing for the direct attachment of diverse perfluoroalkyl chains (n=4, 6, 8) with high structural fidelity and simplified purification via column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disubstituted Perfluoroalkyl Phthalonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of fluorous phase chemistry in developing next-generation catalysts and electronic materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of fluorinated intermediates we produce. We understand that consistency is key in high-tech applications, and our quality management systems are designed to deliver the reliability that global supply chains demand.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the rapidly evolving market for advanced fluorine materials.
