Advanced Synthetic Strategy for Pyroxasulfone Intermediates: Enhancing Yield and Safety in Herbicide Manufacturing
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key agrochemical intermediates. Patent CN111393427A introduces a groundbreaking synthetic method for sulfuryl pyraflufen, commonly known as pyroxasulfone, addressing critical bottlenecks in traditional manufacturing. This technology represents a significant leap forward for any reliable agrochemical intermediate supplier seeking to optimize production lines. The patent details a robust pathway that not only enhances total yield to a range of 31-38% but also fundamentally alters the safety profile of the reaction sequence. By replacing hazardous reagents with more stable alternatives and streamlining the oxidation steps, this method offers a compelling value proposition for large-scale chemical manufacturing. The following analysis dissects the technical nuances and commercial implications of this proprietary process.
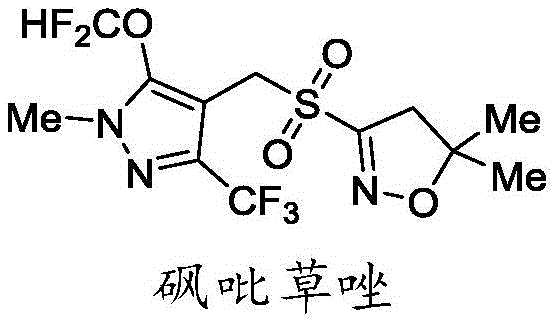
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyroxasulfone has been plagued by significant operational hazards and economic inefficiencies. As illustrated in prior art such as WO2007071900a1, traditional routes often rely on the use of isobutene gas during the formation of the dihydroisoxazole intermediate, posing severe explosion risks in large-scale reactors. Furthermore, these legacy processes frequently employ expensive halogenating agents like N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS), which drastically inflate raw material costs. Another critical drawback is the reliance on sodium methyl mercaptide, a compound notorious for its intense, unpleasant odor and high toxicity, complicating waste management and worker safety protocols. Additionally, the conventional pathway involves redundant multi-step oxidation and reduction sequences, which not only lower the overall atom economy but also increase the generation of hazardous waste streams, making cost reduction in herbicide manufacturing increasingly difficult under modern environmental regulations.
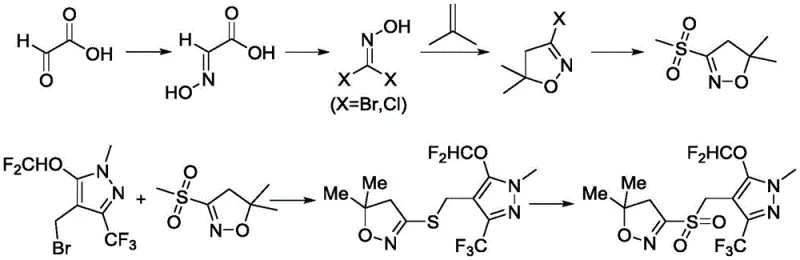
The Novel Approach
In stark contrast, the methodology disclosed in CN111393427A presents a streamlined and economically superior alternative. This novel approach initiates with a safe cyclization reaction to form Intermediate II, followed by a highly efficient chlorination using phosphorus pentachloride or phosphorus oxychloride, which are far more cost-effective than NBS or NCS. The subsequent reaction with thiourea to form the hydrochloride Intermediate IV avoids the use of malodorous mercaptides entirely. Perhaps most importantly, the final oxidation step utilizes a green catalytic system comprising hydrogen peroxide and sodium tungstate. This eliminates the need for hazardous oxidants like m-chloroperoxybenzoic acid (m-CPBA) and prevents the formation of difficult-to-remove byproducts. The result is a process that is not only safer and cleaner but also delivers a higher purity product with simplified post-treatment, marking a substantial advancement for the commercial scale-up of complex heterocyclic intermediates.
Mechanistic Insights into Sodium Tungstate-Catalyzed Oxidation
The cornerstone of this improved synthesis lies in the final oxidation stage, where the thioether linkage is converted into the critical sulfone functionality. In traditional methods, oxidizing a thioether to a sulfone without over-oxidation or stopping at the sulfoxide stage is challenging. The patent specifies a catalytic system using sodium tungstate and an acid, such as sulfuric acid, in the presence of hydrogen peroxide. Mechanistically, the tungstate species likely forms a peroxotungstate complex in situ, which acts as a potent oxygen transfer agent. This complex facilitates the direct and selective oxidation of the sulfur atom from the sulfide state to the sulfone state. This selectivity is crucial because it minimizes the formation of the sulfoxide intermediate, which would otherwise require a second oxidation step or difficult separation, thereby dragging down the overall yield. The acidic environment stabilizes the active oxidant and ensures the reaction proceeds smoothly at mild temperatures, typically between 0-70°C, preserving the integrity of the sensitive heterocyclic rings.
Furthermore, this mechanistic pathway offers distinct advantages regarding impurity control and downstream processing. By avoiding m-CPBA, the process completely sidesteps the generation of m-chlorobenzoic acid, a common byproduct that often co-crystallizes with the product and requires extensive washing or recrystallization to remove. The absence of this acidic byproduct simplifies the workup procedure significantly; the product can often be isolated by simple filtration or extraction, yielding a white solid with a content exceeding 99%. This high level of purity is achieved without the need for energy-intensive recrystallization steps, which directly translates to lower energy consumption and reduced solvent usage. For R&D teams focused on impurity profiles, this route provides a cleaner reaction matrix, reducing the burden on analytical QC labs and ensuring a more consistent quality of high-purity agrochemical intermediates for final formulation.
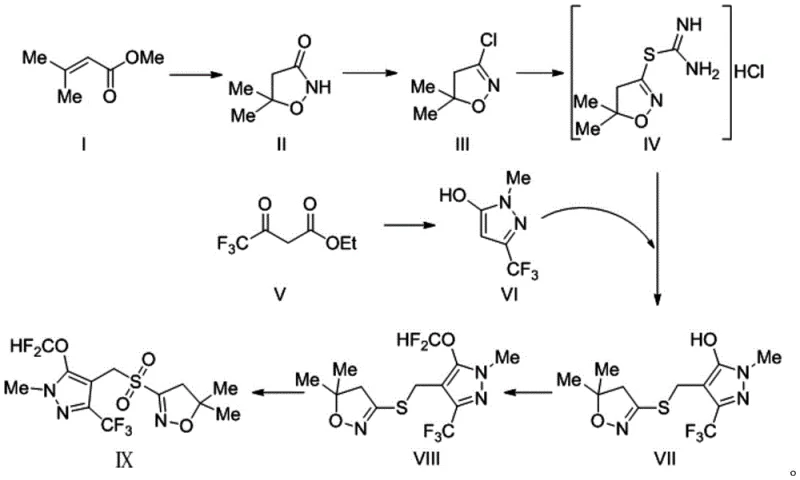
How to Synthesize Pyroxasulfone Efficiently
The synthesis of this critical herbicide intermediate involves a logical sequence of heterocyclic construction and functional group manipulation. The process begins with the cyclization of an acrylate derivative with hydroxyurea, followed by activation via chlorination. The resulting chloro-intermediate is then coupled with a thiourea derivative to introduce the sulfur handle, which is subsequently linked to the pyrazole core. The final and most critical transformation is the catalytic oxidation. While the general flow is straightforward, precise control over reaction parameters such as temperature, stoichiometry, and addition rates is essential to maximize the reported 31-38% total yield. The detailed standardized synthesis steps, including specific molar ratios and solvent choices optimized for industrial throughput, are outlined in the guide below.
- Perform cyclization of Compound I with hydroxyurea to form Intermediate II, followed by chlorination using phosphorus pentachloride to yield Intermediate III.
- React Intermediate III with thiourea to generate the hydrochloride Intermediate IV, ensuring high purity through recrystallization.
- Couple Intermediate IV with pyrazole derivative VI using formaldehyde, followed by difluoromethoxylation and final catalytic oxidation with hydrogen peroxide and sodium tungstate to obtain Target IX.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for specialized and volatile reagents like isobutene gas and expensive halogenating agents, the supply chain becomes more resilient and less susceptible to market fluctuations. The substitution of these materials with commodity chemicals like phosphorus pentachloride and hydrogen peroxide ensures a steady and reliable supply, reducing lead time for high-purity agrochemical intermediates. Moreover, the removal of toxic and malodorous compounds like sodium methyl mercaptide significantly lowers the operational costs associated with safety containment, personal protective equipment, and hazardous waste disposal, contributing to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of high-cost reagents with inexpensive, bulk-available alternatives. The elimination of NBS and NCS, which are premium-priced fine chemicals, in favor of phosphorus chlorides results in a direct reduction in bill of materials costs. Additionally, the streamlined oxidation step avoids the use of m-CPBA, another costly oxidant, and reduces the number of purification steps required. The ability to achieve over 99% purity without recrystallization saves significant amounts of solvent and energy, further driving down the cost per kilogram. These cumulative efficiencies allow for a more competitive pricing structure in the global agrochemical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a logistics perspective, the new route mitigates several high-risk factors. The avoidance of gaseous isobutene removes the need for specialized pressure vessels and gas handling infrastructure, simplifying facility requirements and reducing capital expenditure. The use of stable liquid and solid reagents simplifies storage and transportation, minimizing the risk of supply disruptions due to regulatory restrictions on hazardous gases. Furthermore, the robustness of the reaction conditions, which tolerate a broader range of temperatures and mixing rates compared to the sensitive prior art, ensures consistent batch-to-batch performance. This reliability is critical for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by multinational agrochemical companies.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this method is inherently designed for expansion. The exothermic profiles of the reactions are manageable, and the use of aqueous workups and recyclable solvents aligns with green chemistry principles. The reduction in hazardous waste generation, particularly the absence of heavy metal catalysts or persistent organic byproducts, simplifies compliance with increasingly stringent environmental regulations. This 'green' profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturer, making the supply chain more attractive to environmentally conscious partners and stakeholders in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production facilities.
Q: What are the safety advantages of the new pyroxasulfone synthesis route compared to conventional methods?
A: The novel route eliminates the use of dangerous isobutene gas and toxic, malodorous sodium methyl mercaptide found in prior art. It utilizes safer chlorinating agents like phosphorus pentachloride and a green oxidation system, significantly reducing industrial safety hazards.
Q: How does the catalytic oxidation step improve product purity?
A: By employing a sodium tungstate and acid catalyst system with hydrogen peroxide, the process directly oxidizes the thioether to the sulfone. This avoids the formation of sulfoxide byproducts and m-chlorobenzoic acid waste associated with m-CPBA oxidation, yielding over 99% content without recrystallization.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is designed for industrial scalability. It features simple reaction conditions (often 20-30°C), easily obtainable raw materials, and simplified post-treatment processes like distillation and extraction, leading to a total yield improvement to 31-38%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyroxasulfone Supplier
The technological advancements detailed in patent CN111393427A underscore the potential for more efficient and sustainable production of vital agrochemical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust industrial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in a commercial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Pyroxasulfone intermediate meets the highest international standards, providing our partners with a secure and high-quality supply source.
We invite global agrochemical manufacturers to collaborate with us to leverage this optimized synthesis route for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive edge through superior chemistry and reliable supply chain execution.
