Advanced Palladium-Catalyzed Synthesis of Diaryl Pyrroles for Commercial Scale-Up and Pharmaceutical Applications
Introduction to Novel Diaryl Pyrrole Synthesis Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly pyrrole derivatives, which serve as critical scaffolds in numerous bioactive molecules. A significant technological breakthrough in this domain is documented in Chinese Patent CN108129373B, which discloses a highly efficient, three-step synthetic route for preparing diaryl pyrrole compounds. This patent represents a paradigm shift from traditional multicomponent reactions that often suffer from poor regioselectivity and harsh conditions. By leveraging a sophisticated palladium-catalyzed cascade involving cross-coupling and oxidative cyclization, this method achieves superior yields and purity profiles. For R&D directors and process chemists, understanding the nuances of this pathway is essential for developing next-generation active pharmaceutical ingredients (APIs). The invention not only simplifies the structural assembly of complex pyrroles but also addresses the critical need for reliable diaryl pyrrole supplier capabilities in the global market.
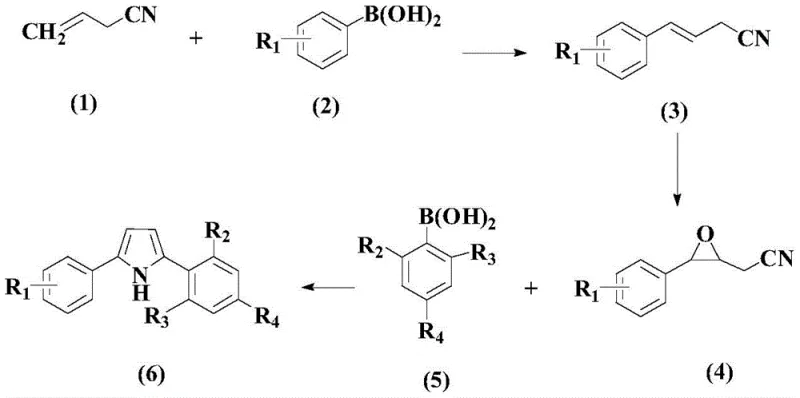
The core innovation lies in the strategic disconnection of the pyrrole ring construction, utilizing readily available allyl cyanide and arylboronic acids as starting materials. This approach circumvents the limitations of earlier methods that relied on precious metal catalysts like rhodium or ruthenium, which are not only cost-prohibitive but also pose challenges in residual metal removal for pharmaceutical grades. The patent details a meticulous optimization of reaction parameters, including catalyst loading, ligand selection, and solvent systems, ensuring that the process is both economically viable and environmentally considerate. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a compelling solution for cost reduction in pharmaceutical intermediates manufacturing, aligning perfectly with the strategic goals of modern chemical enterprises aiming for sustainable growth.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyrroles has been fraught with challenges related to regiocontrol and substrate scope. Prior art, such as the methods disclosed in CN201610073850.3 and CN201610064159.9, often necessitates the use of specialized substrates or suffers from limited functional group tolerance. Furthermore, literature precedents involving rhodium-catalyzed C-H activation, while elegant, typically require expensive catalysts and stringent anhydrous conditions that complicate scale-up. For instance, methods utilizing Rh(II) catalysts for formal [3+2] cycloadditions often encounter issues with catalyst deactivation and the formation of complex impurity profiles that are difficult to separate. Additionally, traditional Paal-Knorr syntheses, though classic, lack the flexibility to introduce diverse aryl groups at specific positions without extensive protecting group chemistry. These limitations result in prolonged development timelines and inflated production costs, creating a bottleneck for the commercial scale-up of complex pharmaceutical intermediates.
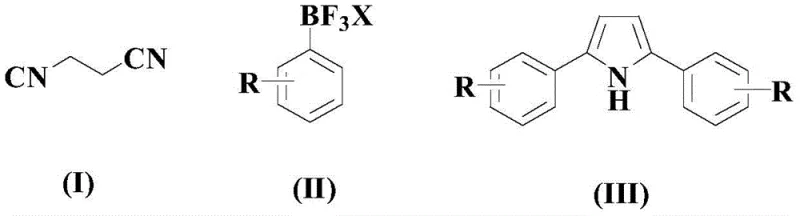
The Novel Approach
In stark contrast, the novel approach outlined in CN108129373B introduces a modular strategy that decouples the formation of the carbon framework from the ring closure event. The process begins with a palladium-catalyzed Heck-type coupling between allyl cyanide and an arylboronic acid to generate a cinnamyl nitrile intermediate. This is followed by a selective oxidation to form an epoxy nitrile, which then undergoes a final palladium-catalyzed coupling with a second arylboronic acid to close the pyrrole ring. This stepwise progression allows for precise control over the substitution pattern, enabling the synthesis of both symmetrical and asymmetrical diaryl pyrroles with high fidelity. The use of palladium acetate in conjunction with specific bipyridine ligands ensures high turnover numbers, while the employment of aqueous-alcoholic solvent systems in the final step enhances the green chemistry profile of the synthesis. This novel pathway effectively resolves the regioselectivity issues plaguing older methods and provides a versatile platform for generating diverse pyrrole libraries.
Mechanistic Insights into Palladium-Catalyzed Cyclization
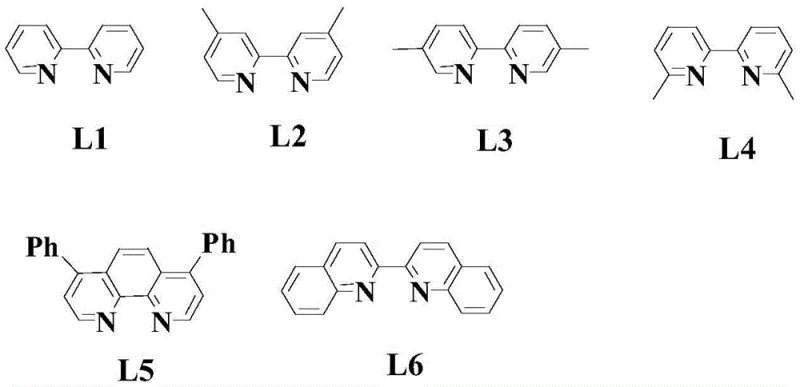
The mechanistic underpinning of this synthesis relies heavily on the interplay between the palladium center and the tailored organic ligands. In the initial coupling step, the palladium catalyst facilitates the activation of the arylboronic acid, promoting transmetallation and subsequent insertion into the alkene moiety of the allyl cyanide. The presence of silver acetate as an oxidant is crucial for regenerating the active palladium species, ensuring the catalytic cycle proceeds efficiently. Detailed investigation reveals that the choice of ligand is paramount; specifically, the use of 2,2'-bipyridine derivatives (such as Ligand L1 shown in the patent data) significantly enhances the stability of the palladium complex and directs the regioselectivity of the addition. Without these specific ligands, the reaction tends to produce linear byproducts or fails to proceed altogether, highlighting the non-obvious nature of this optimization.
Furthermore, the final cyclization step involves a delicate balance of acidity and solvation. The patent data indicates that p-toluenesulfonic acid monohydrate acts as a proton source to facilitate the ring closure while preventing the hydrolysis of sensitive intermediates. The solvent system, preferably a mixture of isopropanol and water, plays a dual role: it solubilizes the inorganic bases and boronic acids while providing a medium that supports the transition state of the cyclization. Impurity control is achieved through the precise stoichiometric control of the oxidant in the second step; using m-chloroperoxybenzoic acid (m-CPBA) ensures clean epoxidation without over-oxidation of the nitrile group. This mechanistic clarity allows process chemists to predict and mitigate potential side reactions, ensuring that the final product meets the stringent purity specifications required for high-purity OLED material or pharmaceutical intermediate applications.
How to Synthesize Diaryl Pyrrole Efficiently
Executing this synthesis requires strict adherence to the optimized conditions defined in the patent to maximize yield and minimize waste. The process is divided into three distinct operational stages, each requiring specific attention to temperature, atmosphere, and workup procedures. The initial coupling demands an inert atmosphere to prevent catalyst oxidation, while the subsequent oxidation step requires careful temperature control to avoid exotherms. The final cyclization benefits from the unique solubility properties of the alcohol-water mixture, which aids in product isolation. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Step 1: React allyl cyanide with arylboronic acid using Pd(OAc)2, 2,2'-bipyridine, and silver acetate in THF to form the cinnamyl nitrile intermediate.
- Step 2: Perform oxidative cyclization of the nitrile intermediate using m-CPBA in dichloromethane to generate the epoxy nitrile precursor.
- Step 3: Conduct the final palladium-catalyzed coupling with a second arylboronic acid using ligand L1 and p-toluenesulfonic acid in isopropanol-water to yield the target diaryl pyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers transformative advantages that directly impact the bottom line and operational resilience. The primary benefit stems from the utilization of commodity chemicals as starting materials. Allyl cyanide and various arylboronic acids are produced on a massive industrial scale, ensuring a stable and continuous supply chain that is not subject to the volatility associated with exotic reagents. This accessibility translates into significant cost reduction in pharmaceutical intermediates manufacturing, as the raw material costs are inherently lower and more predictable compared to routes requiring custom-synthesized precursors. Moreover, the elimination of expensive rhodium or ruthenium catalysts removes a major cost driver and simplifies the supply chain logistics, as palladium catalysts are more readily available and easier to recover.
- Cost Reduction in Manufacturing: The process eliminates the need for precious metal catalysts like rhodium, which are subject to extreme price fluctuations and supply constraints. By switching to palladium-based systems with high turnover efficiency, manufacturers can drastically reduce the cost of goods sold (COGS). Additionally, the use of standard solvents like THF and isopropanol, which are easily recycled, further lowers operational expenditures. The high yields reported in the patent examples mean less raw material is wasted, enhancing the overall atom economy and reducing the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on widely available boronic acids and simple nitriles mitigates the risk of supply disruptions. Unlike specialized heterocyclic building blocks that may have single-source suppliers, the inputs for this process are commoditized. This diversification of the supply base ensures that production schedules can be maintained even during market shortages of specific fine chemicals. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, providing flexibility in global sourcing strategies.
- Scalability and Environmental Compliance: The reaction conditions are mild, operating at temperatures between 70°C and 100°C, which reduces energy consumption and safety risks associated with high-pressure or cryogenic processes. The final step utilizes an aqueous solvent system, which significantly reduces the volume of hazardous organic waste generated. This alignment with green chemistry principles simplifies waste treatment and disposal, lowering environmental compliance costs. The straightforward workup procedures, involving standard extractions and chromatography, are easily adaptable to large-scale continuous flow reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and applicability.
Q: What are the key advantages of this palladium-catalyzed route over traditional rhodium methods?
A: This method utilizes widely available palladium catalysts and arylboronic acids instead of expensive rhodium complexes, significantly reducing raw material costs while maintaining high regioselectivity and yield.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The process employs specific ligand optimization (Ligand L1) and controlled acidic conditions (p-toluenesulfonic acid) which minimize side reactions and byproduct formation, facilitating easier purification via standard column chromatography.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction conditions utilize common organic solvents like THF and isopropanol and operate at moderate temperatures (70-100°C), making the process highly amenable to scale-up from laboratory to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN108129373B for the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless. We are committed to delivering high-purity diaryl pyrrole compounds that meet rigorous quality standards, supported by our state-of-the-art rigorous QC labs and analytical capabilities. Our infrastructure is designed to handle complex palladium-catalyzed reactions with precision, guaranteeing consistent batch-to-batch quality and stringent purity specifications essential for regulatory approval.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments for your target molecules. Let us help you optimize your supply chain and accelerate your drug development pipeline with our reliable diaryl pyrrole supplier services and deep technical expertise.
