Advanced Enzymatic Synthesis of 7-Dehydrocholesterol Intermediates for Pharmaceutical Scale-Up
Advanced Enzymatic Synthesis of 7-Dehydrocholesterol Intermediates for Pharmaceutical Scale-Up
The global demand for high-purity Vitamin D3 precursors continues to surge, driven by the expanding nutraceutical and pharmaceutical sectors. A pivotal breakthrough in this domain is detailed in patent CN115820782A, which discloses a novel preparation method for 7-dehydrocholesterol intermediates utilizing a sophisticated biocatalytic approach. This technology represents a paradigm shift from traditional harsh chemical synthesis to a greener, enzyme-mediated pathway that operates under mild physiological conditions. By leveraging a dual-enzyme system comprising ketoreductase and ester hydrolase, coupled with an efficient coenzyme regeneration cycle, this method effectively resolves long-standing issues regarding isomer impurities and low yields that have plagued the industry for decades. For R&D directors and procurement strategists, understanding this transition is critical, as it offers a reliable pharmaceutical intermediate supplier pathway that aligns with modern green chemistry mandates while ensuring robust supply chain continuity for critical vitamin synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-dehydrocholesterol has relied heavily on chemically aggressive routes that pose significant operational and environmental challenges. The classical bromination-dehydrobromination process, for instance, utilizes elemental bromine, a highly corrosive and toxic reagent that requires specialized containment and extensive waste treatment infrastructure. As illustrated in the traditional reaction pathways, this method often results in the bromination of the side chain double bond, creating complex byproduct profiles that are notoriously difficult to separate from the target molecule. Furthermore, alternative oxidation-reduction strategies frequently employ expensive oxidants or high-pressure hydrogenation, the latter carrying inherent safety risks due to the explosive nature of hydrogen gas. These conventional methodologies typically suffer from low atom economy and generate substantial hazardous waste, leading to inflated production costs and regulatory scrutiny that can disrupt the supply of high-purity OLED material or pharmaceutical precursors.
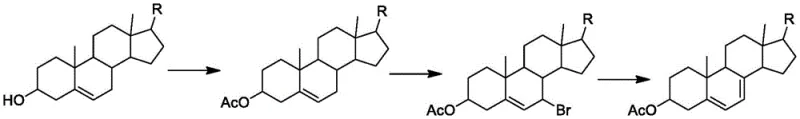
The Novel Approach
In stark contrast, the innovative process outlined in the patent introduces a streamlined synthetic route that bypasses these hazardous chemical transformations entirely. The core of this novelty lies in the construction of a specific intermediate, Compound V, through a highly selective enzymatic reduction of Compound IV. This approach not only eliminates the need for toxic halogens and heavy metal catalysts but also operates at near-neutral pH levels and moderate temperatures ranging from 25-35°C. The integration of a coenzyme regeneration system ensures that the expensive NAD+ cofactor is continuously recycled in situ, drastically reducing raw material consumption. This method effectively improves the yield and purity of the intermediate, creating a stable foundation for the subsequent synthesis of 7-dehydrocholesterol. By adopting this route, manufacturers can achieve cost reduction in API manufacturing through simplified downstream processing and reduced waste disposal liabilities.
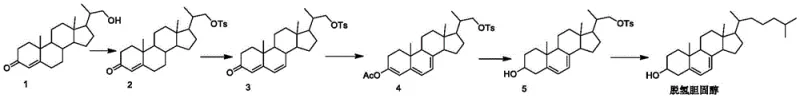
Mechanistic Insights into Dual-Enzyme Catalytic Reduction
The mechanistic elegance of this process is rooted in the synergistic action of ketoreductase and ester hydrolase within a carefully engineered solvent matrix. The reaction begins with Compound IV, which possesses a conjugated diene system that is inherently unstable under harsh chemical conditions. The ketoreductase specifically targets the carbonyl group at the C3 position, stereoselectively reducing it to the desired 3-beta-hydroxy configuration while leaving the sensitive 5,7-diene system intact. Simultaneously, the ester hydrolase manages the protecting groups, ensuring that the hydrolysis occurs without disrupting the delicate steroid backbone. This dual-enzyme cascade is supported by a glucose-glucose dehydrogenase system that regenerates the reduced form of the coenzyme (NADH), driving the equilibrium towards product formation with high efficiency. The specificity of these biological catalysts ensures that side reactions, such as the reduction of the double bonds, are virtually non-existent, thereby maintaining the structural integrity required for Vitamin D3 activity.
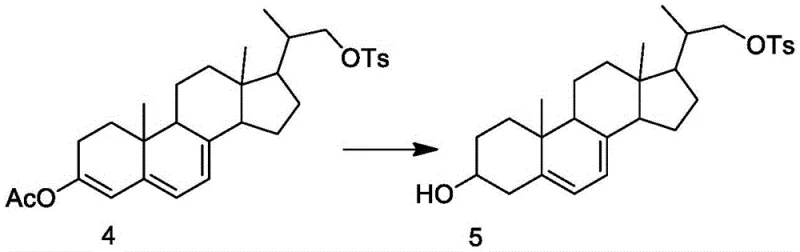
Furthermore, the patent highlights the critical role of the solvent system in stabilizing the transition state of the enzymatic reaction. The use of tert-butanol mixed with co-solvents like dimethyl tetrahydrofuran or ethyl acetate creates a biphasic environment that enhances the solubility of the hydrophobic steroid substrate while maintaining the hydration shell necessary for enzyme activity. This specific solvent composition prevents the aggregation of the substrate and protects the conjugated triene system from isomerization, a common failure mode in chemical reductions. By controlling the pH strictly between 7.0 and 7.5 using sodium carbonate, the process maintains the optimal ionization state for the enzyme active sites. This precise control over the reaction microenvironment allows for the production of Compound V with purity exceeding 99%, demonstrating a level of selectivity that chemical reagents simply cannot match without extensive purification steps.
How to Synthesize 7-Dehydrocholesterol Intermediate Efficiently
The practical implementation of this synthesis involves a sequence of well-defined chemical and biological transformations that can be scaled from laboratory to industrial production. The process initiates with the protection and activation of the starting sterol, followed by the introduction of the 5,7-diene system through dehydrogenation. The crucial step involves the biocatalytic reduction described previously, which converts the keto-intermediate into the hydroxy-intermediate with high stereochemical fidelity. Following isolation and purification of this key intermediate, the final carbon skeleton is completed via a Grignard coupling reaction with a halogenated isopentane derivative. The detailed standardized synthesis steps for this high-efficiency pathway are provided in the guide below.
- Prepare Compound IV through sequential esterification of Compound I with p-toluenesulfonyl chloride, followed by dehydrogenation using trimethyl orthoacetate and PTS, and finally acylation with acetic anhydride.
- Conduct the biocatalytic reduction by mixing Compound IV with a specific solvent system containing tert-butanol, water, ketoreductase, ester hydrolase, and a glucose-based coenzyme regeneration system at 25-35°C.
- Isolate the resulting Compound V through filtration and recrystallization, then react with a Grignard reagent derived from halogenated isopentane to finalize the 7-dehydrocholesterol structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic benefits that extend beyond simple yield metrics. The shift away from hazardous reagents like bromine and sodium borohydride significantly reduces the regulatory burden associated with handling dangerous goods, thereby simplifying logistics and storage requirements. Moreover, the mild reaction conditions reduce the wear and tear on reactor vessels and eliminate the need for high-pressure equipment, leading to lower capital expenditure and maintenance costs over the lifecycle of the production facility. This process optimization facilitates the commercial scale-up of complex polymer additives and pharmaceutical intermediates by providing a more predictable and controllable manufacturing environment.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous chemical reagents, combined with the efficient recycling of cofactors through the glucose dehydrogenase system, leads to substantial cost savings in raw material procurement. Additionally, the high purity of the crude product reduces the load on downstream purification units, such as chromatography columns or crystallization tanks, lowering energy consumption and solvent usage. This holistic reduction in operational complexity allows for a more competitive pricing structure for the final 7-dehydrocholesterol product, enhancing margin potential for downstream Vitamin D3 manufacturers.
- Enhanced Supply Chain Reliability: By utilizing robust enzymatic catalysts that function in aqueous-organic mixed solvents, the process becomes less susceptible to the supply volatility often seen with specialty chemical reagents. The ability to source enzymes from established biotech suppliers ensures a steady flow of critical catalysts, mitigating the risk of production stoppages due to raw material shortages. Furthermore, the stability of the intermediate products under the described storage and handling conditions reduces the risk of degradation during transit, ensuring that reducing lead time for high-purity pharmaceutical intermediates is achievable even across global distribution networks.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic step significantly reduces the volume of organic waste generated per kilogram of product, aligning perfectly with increasingly stringent environmental regulations. The process avoids the generation of heavy metal waste streams and acidic off-gases, simplifying wastewater treatment and废气 abatement protocols. This environmental compatibility not only future-proofs the manufacturing site against regulatory changes but also enhances the corporate sustainability profile, a key metric for modern supply chain audits and ESG reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, impurity profiles, and process robustness. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the primary advantages of the enzymatic reduction method over traditional bromination?
A: The enzymatic method eliminates the use of hazardous elemental bromine and avoids the formation of difficult-to-separate isomer impurities common in chemical reduction, resulting in significantly higher purity (>99%) and improved environmental safety.
Q: How does the specific solvent system impact the reaction yield?
A: The use of a mixed solvent system containing tert-butanol and co-solvents like dimethyl tetrahydrofuran stabilizes the conjugated triene system of the substrate during enzyme catalysis, preventing structural degradation and maximizing the conversion rate to the target intermediate.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates under mild conditions (25-35°C, pH 7.0-7.5) and utilizes a coenzyme regeneration system that minimizes cofactor consumption, making it highly scalable and cost-effective for industrial production compared to high-pressure hydrogenation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Dehydrocholesterol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route described in patent CN115820782A for the production of high-value steroid intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 7-dehydrocholesterol intermediate meets the exacting standards required for pharmaceutical and nutraceutical applications. We are committed to delivering consistent quality and reliability, positioning ourselves as a trusted ally in your supply chain.
We invite you to collaborate with our technical team to explore how this advanced biocatalytic process can optimize your specific manufacturing requirements. By engaging with us, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener methodology. Please contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your project timelines and volume needs.
