Advanced Synthesis of Heteroatom-Containing Cyclohexene Halides for Industrial Scale-Up
Advanced Synthesis of Heteroatom-Containing Cyclohexene Halides for Industrial Scale-Up
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes for complex intermediates that balance high purity with economic viability. Patent CN110563696B introduces a transformative preparation method for heteroatom-containing cyclohexene halides, addressing critical bottlenecks in the synthesis of these valuable building blocks. These compounds serve as pivotal precursors for constructing cyclohexene or cyclohexane derivatives containing heteroatoms, which are extensively utilized in drug discovery, natural product synthesis, and the development of advanced optical materials such as high-speed photoetching recording media. The disclosed technology leverages a strategic combination of halogenating reagents and specific organic base-additive systems to streamline the production workflow. By fundamentally altering the reaction conditions from cryogenic constraints to moderate thermal ranges, this innovation offers a compelling solution for manufacturers aiming to enhance process efficiency while maintaining rigorous quality standards.
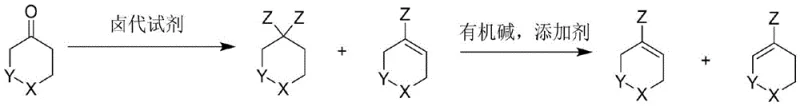
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-heteroatom substituted cyclohexene halides has been plagued by severe operational challenges that hinder industrial adoption. Prior art methods, such as those described in Tetrahedron Letters, often necessitate the use of strong bases like LiHMDS in tetrahydrofuran at ultra-low temperatures of -78°C, requiring absolute exclusion of water and oxygen. Such苛刻 conditions demand specialized cryogenic equipment and impose significant safety risks, particularly when handling reactive reagents like PhNTf2. Furthermore, alternative routes utilizing triphenylphosphine and bromine at -60°C suffer from low molar yields around 62% and generate difficult-to-treat three-waste streams. The reliance on hazardous halogens and the necessity for extensive purification steps, such as silica gel chromatography, drastically inflate production costs and limit the scalability of these traditional processes for commercial manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN110563696B presents a streamlined pathway that circumvents the need for intermediate purification and harsh cryogenic environments. The process initiates by reacting heteroatom-containing cyclohexanone with halogenating agents like thionyl chloride or oxalyl chloride at moderate temperatures ranging from 30°C to 100°C. A key innovation lies in the direct utilization of the resulting crude mixture, which contains both gem-dihalide and alkenyl halide species, without the energy-intensive separation steps typically required. By introducing specific additives into an organic base system for the subsequent dehydrohalogenation step, the method effectively overcomes the kinetic barriers associated with eliminating gem-dichloro compounds. This approach not only simplifies the operational workflow but also maximizes atom utilization, ensuring that the final heteroatom-containing cyclohexene halide is produced with high efficiency and minimal waste generation.
Mechanistic Insights into Additive-Assisted Dehydrohalogenation
The core chemical breakthrough of this patent resides in the sophisticated manipulation of elimination kinetics through the use of amide additives. In conventional dehydrohalogenation scenarios, gem-dibromo compounds eliminate relatively easily, whereas gem-dichloro compounds present a significant kinetic hurdle, often requiring excessive amounts of base and prolonged reaction times exceeding 20 hours. The introduction of additives such as sodium amide (NaNH2) or lithium amide (LiNH2) fundamentally alters this landscape by accelerating the elimination rate, allowing the reaction to reach completion within a practical timeframe of 2 to 5 hours. This acceleration is critical for industrial throughput, as it prevents the accumulation of unreactive intermediates and ensures a consistent flow of material through the production line. The synergy between the organic base, such as DBU or TMG, and the inorganic amide additive creates a highly reactive environment that facilitates the removal of hydrogen halide even from sterically hindered positions.

Beyond mere acceleration, the process offers precise control over regioselectivity, which is paramount for synthesizing specific isomers required for downstream applications. The patent elucidates that the position of the double bond formation is governed by the steric hindrance of the selected organic base. For instance, employing sterically bulky bases like tetramethylguanidine at elevated temperatures favors the formation of Product A, likely due to the preferential abstraction of protons from less hindered positions. Conversely, utilizing different base systems at lower temperatures can direct the reaction towards Product B. This tunability allows chemists to tailor the synthesis towards the desired isomer without altering the core substrate, providing a versatile platform for generating diverse libraries of heteroatom-containing cyclohexene derivatives. The ability to switch between isomers simply by adjusting base sterics and temperature represents a significant advantage in process development.
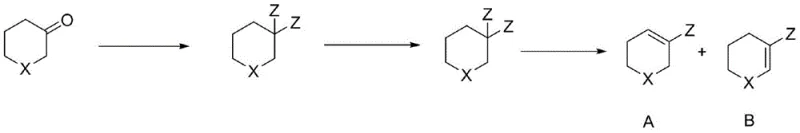
How to Synthesize Heteroatom-Containing Cyclohexene Halide Efficiently
The synthesis protocol described in the patent provides a clear roadmap for executing this transformation with high reproducibility and yield. The procedure begins with the halogenation of the ketone substrate, where careful control of the halogenating agent stoichiometry ensures the formation of the desired gem-dihalide or alkenyl halide mixture. Following the removal of excess halogenating reagent, the crude concentrate is directly subjected to the elimination step without any intermediate workup, which is a critical time-saving measure. The addition of the organic base and the amide additive must be performed under inert atmosphere to prevent moisture interference, followed by heating to the specified temperature range to drive the reaction to completion. Detailed standardized synthesis steps see the guide below.
- React heteroatom-containing cyclohexanone with a halogenating agent such as thionyl chloride or oxalyl chloride at 30-100°C to generate a mixture of gem-dihalide and alkenyl halide intermediates without purification.
- Introduce an organic base like DBU or TMG along with a critical additive such as sodium amide or lithium amide to the reaction mixture.
- Heat the mixture to facilitate dehydrohalogenation, overcoming the kinetic barrier of gem-dichloro elimination to yield the final heteroatom-containing cyclohexene halide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible operational efficiencies and risk mitigation strategies. The elimination of intermediate purification steps, such as column chromatography or complex distillation sequences, significantly reduces the consumption of solvents and stationary phases, leading to substantial cost savings in raw material procurement. Furthermore, the ability to operate at moderate temperatures removes the dependency on expensive cryogenic cooling infrastructure, thereby lowering capital expenditure requirements for new production lines and reducing energy consumption costs. The robustness of the reaction conditions also implies a lower risk of batch failure due to minor fluctuations in temperature or moisture, enhancing overall supply chain reliability and ensuring consistent delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the telescoping of the halogenation and elimination steps, which avoids the isolation and purification of the unstable gem-dihalide intermediate. By processing the crude reaction mixture directly, manufacturers save significant labor hours and reduce the loss of material typically associated with multiple workup procedures. Additionally, the use of commercially available and relatively inexpensive halogenating agents like thionyl chloride, combined with the high atom economy of the elimination step, contributes to a leaner cost structure. The avoidance of precious metal catalysts or exotic reagents further ensures that the cost of goods sold remains competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on common chemical feedstocks and standard reactor equipment enhances the resilience of the supply chain against disruptions. Unlike processes requiring specialized low-temperature reactors or rare reagents, this method can be implemented in most multipurpose chemical plants with minimal retrofitting. The shortened reaction times, achieved through the use of amide additives, increase the throughput capacity of existing facilities, allowing suppliers to respond more rapidly to fluctuating market demands. This agility is crucial for maintaining continuous supply to pharmaceutical clients who require just-in-time delivery of high-quality intermediates for their own production schedules.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates fewer waste streams compared to traditional methods that produce large volumes of silica gel waste or require extensive solvent exchanges. The simplified workflow reduces the overall solvent footprint, aligning with increasingly stringent global environmental regulations and sustainability goals. The scalability of the reaction is demonstrated by the use of standard reflux conditions and common solvents like dichloroethane or dioxane, which are well-understood in large-scale manufacturing contexts. This ease of scale-up minimizes the technical risks associated with technology transfer from laboratory to pilot and commercial plant scales.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and mechanistic explanations provided in the patent documentation, ensuring accuracy and relevance for process chemists. Understanding these nuances is essential for successfully adapting the method to specific substrate variations or production constraints.
Q: How does this process improve upon traditional low-temperature synthesis methods?
A: Traditional methods often require cryogenic conditions (-78°C) and strict anhydrous environments using reagents like LiHMDS. This patented process operates at moderate temperatures (30-100°C) and tolerates crude intermediate mixtures, significantly reducing energy consumption and equipment complexity.
Q: What is the role of additives like sodium amide in this reaction?
A: Additives such as sodium amide or lithium amide are crucial for overcoming the high activation energy required for the elimination of gem-dichloro compounds. They accelerate the dehydrohalogenation rate, allowing the reaction to complete within hours rather than days, while organic bases alone would be insufficient.
Q: Can this method control regioselectivity for different isomer products?
A: Yes, regioselectivity is effectively controlled by the steric hindrance of the chosen organic base. Bulky bases favor the formation of specific alkene isomers at higher temperatures, while different base systems at lower temperatures can direct the formation of alternative regioisomers, providing flexibility for downstream synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heteroatom-Containing Cyclohexene Halide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development and manufacturing programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify identity and potency. By leveraging the innovative processes described in patents like CN110563696B, we can offer you a reliable source of heteroatom-containing cyclohexene halides that are produced with maximum efficiency and minimal environmental impact.
We invite you to collaborate with us to explore how this advanced synthesis technology can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to our optimized process can reduce your overall manufacturing expenses. Please contact us today to request specific COA data for our current inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in driving innovation and efficiency in your supply chain.
