Advanced One-Pot Synthesis of 1,4-Bissulfonylated Pyrazoles for Commercial API Production
Advanced One-Pot Synthesis of 1,4-Bissulfonylated Pyrazoles for Commercial API Production
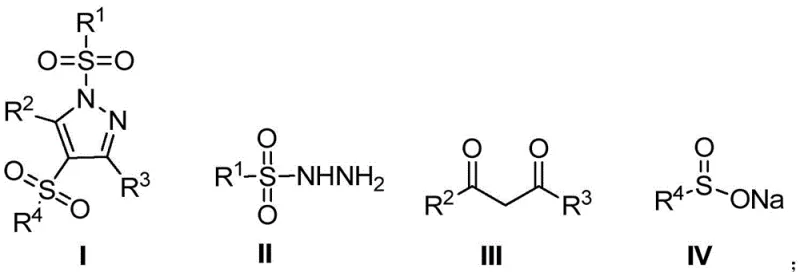
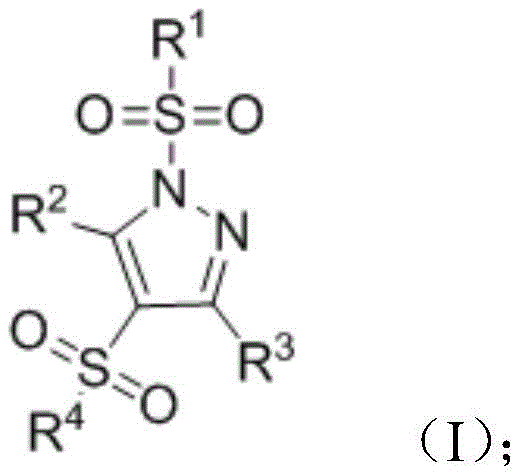
The pharmaceutical industry continuously seeks robust synthetic methodologies that can deliver complex heterocyclic scaffolds with high efficiency and minimal environmental impact. A significant breakthrough in this domain is documented in patent CN112961109B, which discloses a novel preparation method for 1,4-bissulfonylated fully substituted pyrazole compounds. This technology represents a paradigm shift in constructing densely functionalized pyrazole cores, which are critical structural motifs in numerous bioactive molecules, particularly non-steroidal anti-inflammatory drugs (NSAIDs). By leveraging a multi-component reaction strategy mediated by elemental iodine and a mild base, this process enables the direct assembly of 1,3,4,5-tetrasubstituted pyrazoles in a single operational step. For R&D directors and procurement specialists evaluating new supply chains, this patent offers a compelling route to access high-purity pharmaceutical intermediates with reduced process complexity and enhanced cost-efficiency compared to traditional multi-step sequences.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fully substituted pyrazoles bearing distinct sulfonyl groups at the 1 and 4 positions has been a formidable challenge for organic chemists. Conventional strategies often rely on sequential functionalization, where the pyrazole ring is constructed first, followed by laborious protection and deprotection steps to introduce the second sulfonyl moiety selectively. These traditional routes frequently necessitate the use of harsh reaction conditions, such as strong acids or elevated temperatures, which can lead to the decomposition of sensitive functional groups and the formation of difficult-to-remove impurities. Furthermore, the requirement for transition metal catalysts in some cross-coupling approaches introduces the risk of heavy metal contamination, necessitating expensive purification protocols to meet stringent regulatory standards for API intermediates. The cumulative effect of these inefficiencies is a prolonged production timeline and significantly inflated manufacturing costs, creating bottlenecks for supply chain managers aiming to scale production for commercial markets.
The Novel Approach
In stark contrast, the methodology outlined in CN112961109B employs a convergent one-pot strategy that dramatically simplifies the synthetic landscape. By reacting sulfonyl hydrazides, 1,3-dicarbonyl compounds, and sodium sulfinates simultaneously in the presence of iodine and a phosphate base, the process achieves the construction of the pyrazole ring and the installation of both sulfonyl groups in a single operation. This approach operates under remarkably mild conditions, typically at room temperature (20-40°C), which preserves the integrity of diverse substituents and minimizes energy consumption. The use of inexpensive and readily available reagents like sodium sulfinate and elemental iodine eliminates the dependency on precious metal catalysts, thereby reducing raw material costs and simplifying waste treatment. For a reliable pharmaceutical intermediates supplier, this translates into a more resilient and economically viable manufacturing process capable of delivering consistent quality at scale.
Mechanistic Insights into Iodine-Mediated Oxidative Coupling
The success of this transformation hinges on the unique role of elemental iodine as a mild oxidant and mediator in the reaction cycle. Mechanistically, the reaction is believed to proceed through the generation of sulfonyl radicals from the sodium sulfinate precursors, facilitated by the oxidative properties of iodine. These reactive species then engage with the hydrazone intermediates formed in situ from the condensation of sulfonyl hydrazides and 1,3-dicarbonyl compounds. The subsequent cyclization and oxidation steps lead to the aromatization of the pyrazole ring, locking in the 1,4-bissulfonylation pattern with high regioselectivity. This radical-mediated pathway avoids the need for stoichiometric amounts of toxic oxidants or aggressive Lewis acids, aligning with green chemistry principles. Understanding this mechanism is crucial for process chemists, as it highlights the tolerance of the system towards various electronic environments on the aromatic rings, allowing for the synthesis of a broad library of derivatives without compromising yield or purity.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring the quality of the final product. Traditional high-temperature syntheses often promote side reactions such as over-oxidation or hydrolysis of the sulfonyl groups, leading to complex impurity profiles that are challenging to characterize and remove. In this iodine-mediated protocol, the reaction temperature is maintained near ambient levels, significantly suppressing thermal degradation pathways. Moreover, the choice of dipotassium hydrogen phosphate trihydrate as the base provides a buffered environment that prevents the acid-catalyzed decomposition of sensitive intermediates. This inherent stability simplifies the downstream processing, often allowing for the isolation of the target compound through simple filtration and washing, rather than requiring resource-intensive chromatographic purification. Such operational simplicity is a key driver for cost reduction in API manufacturing, as it reduces solvent usage and processing time.
How to Synthesize 1,4-Bissulfonylated Pyrazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize efficiency and safety. The general procedure involves charging a reaction vessel with the sulfonyl hydrazide, 1,3-dicarbonyl compound, and sodium sulfinate in a molar ratio optimized for complete conversion, typically using acetonitrile as the solvent of choice due to its excellent solubility profile and stability under oxidative conditions. Elemental iodine and the phosphate base are then added, and the mixture is stirred at room temperature for a period ranging from 15 to 22 hours. The detailed standardized synthesis steps see the guide below.
- Combine sulfonyl hydrazide, 1,3-dicarbonyl compound, and sodium sulfinate in acetonitrile with elemental iodine and a phosphate base.
- Stir the reaction mixture at room temperature (20-40°C) for 15 to 22 hours to ensure complete conversion.
- Quench with saturated sodium thiosulfate, filter the precipitated solid, and wash with water and petroleum ether to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic advantages beyond mere technical novelty. The primary benefit lies in the substantial simplification of the supply chain for raw materials. Unlike processes requiring specialized organometallic reagents or custom-synthesized building blocks, this method utilizes commodity chemicals such as sodium sulfinates and simple ketones, which are available from multiple global vendors. This diversification of the supply base mitigates the risk of shortages and price volatility, ensuring a steady flow of materials for continuous production. Additionally, the elimination of transition metal catalysts removes the need for costly metal scavenging resins and extensive testing for residual metals, directly lowering the cost of goods sold (COGS) and accelerating the release of batches for further processing.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the convergence of multiple synthetic steps into a single pot, which drastically reduces labor hours, energy consumption, and solvent waste. By operating at room temperature, the process eliminates the need for heating or cooling infrastructure, leading to significant utility savings. Furthermore, the high atom economy of the multi-component reaction ensures that a larger proportion of the starting mass is incorporated into the final product, minimizing waste disposal costs. These factors combine to create a lean manufacturing model that enhances profit margins while maintaining competitive pricing for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch-to-batch consistency, a critical factor for maintaining supply continuity in the pharmaceutical sector. The tolerance of the method to moisture and air, as indicated by the ability to carry out the reaction without strict inert atmosphere requirements in some embodiments, simplifies operational logistics and reduces the risk of batch failures due to environmental excursions. This reliability allows supply chain planners to optimize inventory levels and reduce safety stock requirements, freeing up working capital. The straightforward workup procedure, often involving simple filtration, also shortens the production cycle time, enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the absence of exothermic hazards associated with strong oxidants or cryogenic reagents. The use of iodine, which can be recovered and recycled, aligns with sustainability goals and reduces the environmental footprint of the manufacturing process. The aqueous workup generates waste streams that are easier to treat compared to those containing heavy metals or chlorinated solvents. This environmental compatibility not only ensures compliance with increasingly stringent regulatory frameworks but also enhances the corporate social responsibility profile of the manufacturing partner, making it a preferred choice for multinational corporations seeking sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazole synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing portfolios.
Q: What are the typical reaction yields for this pyrazole synthesis?
A: According to patent CN112961109B, the optimized reaction conditions typically provide yields around 80%, with some specific derivatives achieving up to 90% isolated yield after simple filtration.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes mild room temperature conditions and inexpensive reagents like iodine and sodium sulfinate, making it highly scalable without requiring cryogenic cooling or high-pressure equipment.
Q: What is the primary biological application of these compounds?
A: These 1,4-bissulfonylated pyrazoles exhibit significant anti-inflammatory activity, serving as potent analogs to Celecoxib, making them valuable intermediates for NSAID development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Bissulfonylated Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN112961109B for the production of advanced anti-inflammatory intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this mild, one-pot synthesis are realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,4-bissulfonylated pyrazole meets the exacting standards required for downstream API synthesis, providing our clients with peace of mind and regulatory confidence.
We invite you to collaborate with us to optimize your supply chain for these valuable heterocyclic building blocks. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our process development capabilities, we can help you navigate the transition from conventional methods to this more efficient technology. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
