Advanced Room Temperature Synthesis of N-Dithiocarbamate Indoles for Commercial Scale-Up
Introduction to Novel N-Dithiocarbamate Indole Synthesis
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds that serve as core structures for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN109384702B, which discloses a highly efficient preparation method for N-dithiocarbamate indole compounds. This technology addresses a long-standing challenge in organic synthesis by enabling the chemoselective formation of nitrogen-sulfur (N-S) bonds directly on the indole nitrogen atom, rather than the traditionally targeted carbon positions. The method utilizes readily available indole derivatives and thiuram compounds as substrates, promoted by potassium tert-butoxide under mild conditions. This innovation not only expands the chemical space available for drug discovery but also offers a robust platform for the commercial production of high-purity pharmaceutical intermediates. The ability to synthesize these compounds at room temperature with short reaction times represents a paradigm shift from conventional high-energy processes, aligning perfectly with modern green chemistry principles and cost-effective manufacturing strategies required by global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioindole compounds has been predominantly focused on introducing sulfur functionalities at the C-3 position of the indole ring. Established methodologies, such as those reported by the Drozd, Knochel, and Beier research groups, typically rely on Fischer indole synthesis, Grignard reagents, or iodine-mediated coupling with carbon disulfide. These conventional routes often suffer from significant limitations, including harsh reaction conditions, the requirement for stoichiometric amounts of expensive or hazardous reagents, and limited functional group tolerance. Furthermore, the exclusive formation of C-3 substituted products restricts the structural diversity available to medicinal chemists, potentially overlooking valuable N-substituted analogs that could possess superior pharmacological profiles. The reliance on transition metals or strong electrophiles in these traditional methods also introduces challenges in downstream purification, particularly regarding the removal of trace metal impurities which is critical for API manufacturing compliance.
The Novel Approach
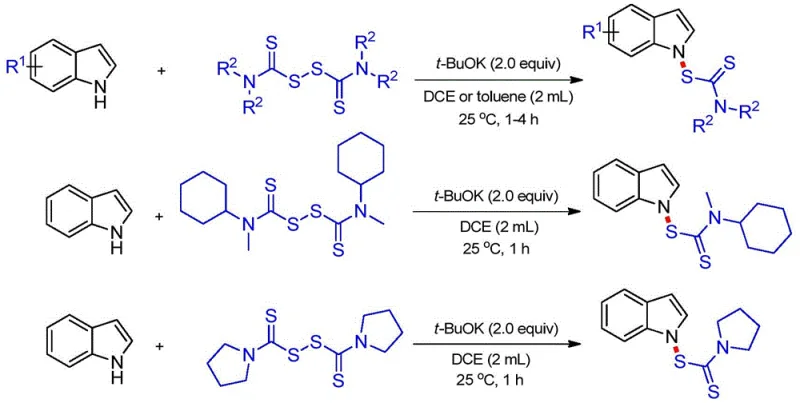
In stark contrast, the novel approach described in the patent data leverages a direct nucleophilic substitution strategy to forge the N-S bond with exceptional chemoselectivity. By employing thiuram compounds as the sulfur source and potassium tert-butoxide as a promoter, the reaction proceeds smoothly at room temperature (25 °C) in solvents like DCE or toluene. This method bypasses the need for pre-functionalization of the indole ring or the use of sensitive organometallic reagents. The reaction mechanism involves the deprotonation of the indole nitrogen to generate a nucleophilic anion, which then attacks the disulfide bond of the thiuram reagent. This pathway is not only operationally simple but also highly versatile, accommodating a wide range of substituents on both the indole and the thiuram moieties. The result is a streamlined process that delivers high yields, such as the 93% yield observed in model reactions, while minimizing waste and energy consumption, thereby offering a superior alternative for industrial scale-up.
Mechanistic Insights into t-BuOK Promoted N-S Bond Formation
The success of this synthesis hinges on the precise interplay between the base and the substrates. Potassium tert-butoxide acts as a strong, non-nucleophilic base that quantitatively deprotonates the N-H bond of the indole ring, generating a resonance-stabilized indolyl anion. This anionic species is highly nucleophilic at the nitrogen center, allowing it to selectively attack the weak sulfur-sulfur single bond present in the thiuram disulfide structure. The cleavage of the S-S bond is the driving force of the reaction, leading to the expulsion of a dithiocarbamate anion and the formation of the stable N-S bond. Experimental data indicates that the choice of base is critical; substituting t-BuOK with other inorganic or organic bases results in a dramatic decrease in yield or complete reaction failure, suggesting that the specific basicity and steric properties of t-BuOK are essential for generating the reactive intermediate without inducing side reactions.
Furthermore, the solvent environment plays a pivotal role in stabilizing the transition state and ensuring high conversion rates. The patent data highlights that non-polar or moderately polar solvents like DCE and toluene are optimal, whereas polar aprotic solvents like 1,4-dioxane lead to incomplete conversion and lower yields (e.g., 53% yield in dioxane vs 93% in DCE). This solvent effect likely influences the solubility of the ionic intermediates and the aggregation state of the potassium cation, which can affect the nucleophilicity of the indole anion. The method also demonstrates remarkable tolerance to electronic variations on the indole ring, successfully processing substrates with electron-withdrawing groups like bromine and fluorine, as well as electron-donating groups like methyl and methoxy. This broad substrate scope confirms the robustness of the mechanistic pathway, making it a reliable tool for synthesizing diverse libraries of N-dithiocarbamate indoles for biological screening.
How to Synthesize N-Dithiocarbamate Indole Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and reaction monitoring to ensure optimal outcomes. The standard procedure involves mixing the indole substrate and the thiuram compound in a molar ratio that favors complete conversion, typically with a slight excess of the thiuram reagent. The addition of exactly 2.0 equivalents of potassium tert-butoxide is crucial to drive the deprotonation equilibrium forward without causing excessive decomposition of sensitive functional groups. Reaction progress is conveniently monitored by thin-layer chromatography (TLC), with most reactions reaching completion within 1 to 4 hours at ambient temperature. Following the reaction, a straightforward aqueous workup involving extraction with ethyl acetate and drying over anhydrous sodium sulfate allows for the isolation of the crude product, which can be further purified by column chromatography to achieve analytical purity suitable for pharmaceutical applications.
- Combine indole compound, thiuram compound, and potassium tert-butoxide (2.0 equiv) in a reaction vessel with DCE or toluene solvent.
- Stir the reaction mixture at room temperature (25 °C) for 1 to 4 hours, monitoring progress via TLC.
- Quench with water, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic methodology offers transformative advantages that directly impact the bottom line and operational resilience. The reliance on commodity chemicals such as indole, thiurams, and potassium tert-butoxide eliminates the dependency on scarce or geopolitically sensitive reagents, ensuring a stable and continuous supply of raw materials. The elimination of transition metal catalysts removes the need for costly and time-consuming metal scavenging steps, which are often bottlenecks in API manufacturing. Additionally, the ability to run reactions at room temperature significantly reduces energy consumption associated with heating or cooling, contributing to lower utility costs and a smaller carbon footprint. These factors collectively enhance the economic viability of producing N-dithiocarbamate indoles, making them accessible for large-scale commercial projects without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive, commercially available starting materials and avoiding the use of precious metal catalysts or specialized ligands. The high atom economy of the reaction, coupled with the high yields reported (often exceeding 90%), minimizes raw material waste and reduces the cost per kilogram of the final product. Furthermore, the simplified workup procedure reduces labor hours and solvent usage, leading to a more lean and efficient manufacturing process that maximizes return on investment for large-scale production runs.
- Enhanced Supply Chain Reliability: By operating under mild conditions with robust reagents, the risk of batch failure due to sensitive reaction parameters is drastically minimized. The short reaction times (1-4 hours) allow for faster throughput and quicker turnaround times for custom synthesis orders, enabling suppliers to respond rapidly to fluctuating market demands. The use of common solvents like toluene and DCE ensures that solvent supply chains remain resilient, avoiding disruptions often associated with specialized or regulated solvents, thus guaranteeing consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The simplicity of the reaction setup facilitates seamless scale-up from laboratory gram-scale to multi-ton commercial production without the need for complex engineering controls. The absence of toxic heavy metals simplifies waste treatment and disposal, ensuring compliance with stringent environmental regulations. This eco-friendly profile not only reduces liability but also aligns with the sustainability goals of modern pharmaceutical companies, making this technology a preferred choice for green chemistry initiatives and long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on critical process parameters. Understanding these nuances is essential for R&D teams aiming to adapt this chemistry for specific target molecules or for process engineers planning for commercial scale-up. The answers reflect the rigorous testing performed to establish the boundaries and capabilities of this novel synthetic route.
Q: Why is potassium tert-butoxide critical for this N-S bond formation?
A: According to patent CN109384702B, potassium tert-butoxide is essential for deprotonating the indole nitrogen to form the nucleophilic indole anion. Other bases, including inorganic and organic alternatives, result in significantly reduced yields or no product formation.
Q: What solvents are compatible with this synthesis method?
A: The process specifically requires DCE (1,2-dichloroethane) or toluene as solvents to achieve high yields. Polar solvents like 1,4-dioxane lead to incomplete conversion and lower isolated yields, indicating a specific solvent effect on the reaction kinetics.
Q: Does this method tolerate functional groups on the indole ring?
A: Yes, the method demonstrates broad substrate scope. It successfully accommodates electron-withdrawing groups like halogens (Br, F) and esters, as well as electron-donating groups like methyl and methoxy, without compromising the chemoselective formation of the N-S bond.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Dithiocarbamate Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of N-dithiocarbamate indole compounds in the development of next-generation therapeutics, particularly in oncology and anti-inflammatory applications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-dithiocarbamate indole intermediates meets the highest industry standards for quality and consistency. We are committed to supporting your drug development timeline with reliable supply and technical expertise.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific pipeline needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your strategic partner in bringing innovative indole-based medicines to market faster and more cost-effectively.
