Advanced Ullmann Coupling Strategy for High-Purity Sulfonamide Pharmaceutical Intermediates
Advanced Ullmann Coupling Strategy for High-Purity Sulfonamide Pharmaceutical Intermediates
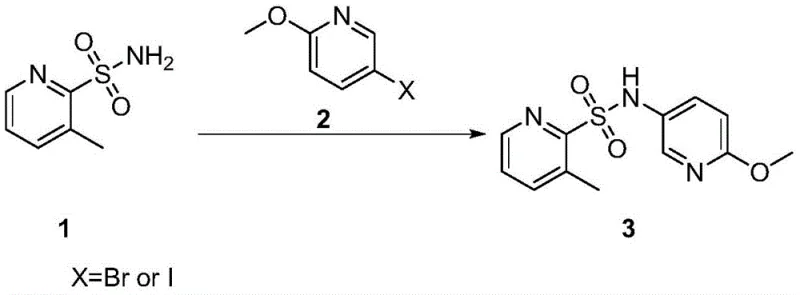
The pharmaceutical and agrochemical industries are constantly seeking more efficient, environmentally benign, and cost-effective synthetic routes for critical heterocyclic intermediates. A significant breakthrough in this domain is documented in patent CN114149364A, which discloses a novel preparation method for N-(6-methoxypyridine-3-yl)-3-methylpyridine-2-sulfonamide. This specific compound serves as a vital building block in the synthesis of various bioactive molecules, including potential antibacterial and anticancer agents. The patented methodology represents a paradigm shift from traditional, hazardous synthetic pathways to a streamlined, copper-catalyzed coupling process. By leveraging the robustness of the Ullmann-type reaction, this innovation addresses long-standing challenges related to precursor stability, odor control, and overall process safety. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for securing a reliable supply chain of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonamide derivatives has predominantly relied on the nucleophilic substitution reaction between sulfonyl chlorides and amine compounds under basic conditions. While chemically straightforward in theory, this conventional approach suffers from severe practical drawbacks when applied to complex heterocyclic systems. The primary bottleneck lies in the preparation of the requisite sulfonyl chloride starting material, which often necessitates the use of volatile, intensely malodorous, and toxic thiol intermediates such as 3-methylpyridine-2-thiol. These thiols pose significant occupational health hazards and require specialized containment infrastructure to prevent environmental contamination. Furthermore, the resulting sulfonyl chloride intermediates are frequently unstable under standard storage conditions, leading to decomposition and variable batch quality. This instability complicates inventory management and increases the risk of supply chain disruptions for downstream manufacturers who rely on consistent raw material specifications.
The Novel Approach
In stark contrast to the legacy methods, the innovative process described in patent CN114149364A utilizes a direct coupling strategy that bypasses the need for unstable sulfonyl chlorides entirely. This novel approach involves the reaction of a stable sulfonamide (Compound 1) with a halogenated pyridine derivative (Compound 2) in the presence of a copper catalyst and an inorganic base. The reaction proceeds smoothly under relatively mild thermal conditions, typically ranging from 120°C to 150°C, utilizing common organic solvents such as toluene, DMF, or NMP. By eliminating the generation of foul-smelling thiols and unstable acid chlorides, this method drastically simplifies the operational workflow and enhances the safety profile of the manufacturing facility. The result is a cleaner reaction profile with fewer byproducts, facilitating easier purification and yielding a final product with superior purity and stable quality characteristics suitable for stringent medical applications.
Mechanistic Insights into Cu-Catalyzed Ullmann Coupling
The core of this synthetic advancement lies in the efficient execution of the Ullmann condensation, a powerful tool for forming carbon-nitrogen bonds between aryl halides and nitrogen nucleophiles. In this specific transformation, the copper catalyst, whether cuprous iodide (CuI) or cuprous oxide (Cu2O), plays a pivotal role in activating the carbon-halogen bond of the 5-halo-2-methoxypyridine substrate. The mechanistic cycle likely involves the oxidative addition of the aryl halide to the copper center, followed by the coordination and deprotonation of the sulfonamide nitrogen by the base. This generates a reactive copper-amido species which subsequently undergoes reductive elimination to forge the desired C-N bond, regenerating the active copper catalyst for the next cycle. The choice of base, such as cesium carbonate or potassium carbonate, is critical not only for neutralizing the acidic byproduct but also for facilitating the formation of the nucleophilic nitrogen species necessary for the coupling event.
From an impurity control perspective, this catalytic system offers distinct advantages over non-catalytic thermal methods. The presence of the copper catalyst lowers the activation energy of the reaction, allowing it to proceed at moderate temperatures (120-150°C) rather than the extreme heat often required for uncatalyzed variants. This moderation of thermal stress minimizes the degradation of sensitive functional groups, such as the methoxy moiety on the pyridine ring, thereby preserving the structural integrity of the molecule. Additionally, the use of a slight molar excess of the halopyridine (1:1.5 to 1:3 ratio) ensures complete consumption of the sulfonamide starting material, preventing the carryover of unreacted amine into the final product stream. This precise control over stoichiometry and reaction kinetics results in a crude product profile that is significantly cleaner, reducing the burden on downstream purification processes like silica gel chromatography.
How to Synthesize N-(6-methoxypyridine-3-yl)-3-methylpyridine-2-sulfonamide Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins by dissolving the stable sulfonamide and the halopyridine in a dry, polar aprotic solvent, ensuring an inert atmosphere to prevent catalyst oxidation. Following the addition of the copper catalyst and base, the mixture is heated, with microwave irradiation offering a rapid alternative to conventional oil bath heating for accelerated throughput. The standardized protocol outlined below details the specific stoichiometric ratios and workup procedures necessary to achieve the reported yields of approximately 60% with high purity.
- Dissolve 3-methylpyridine-2-sulfonamide and 5-halo-2-methoxypyridine in a polar aprotic solvent such as DMF or NMP under nitrogen atmosphere.
- Add a copper catalyst (CuI or Cu2O) and an inorganic base (Cs2CO3, K2CO3, or Na2CO3) to the reaction mixture.
- Heat the mixture to 120-150°C using oil bath or microwave irradiation, then isolate the product via extraction and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling strategic benefits that extend beyond mere chemical elegance. The elimination of hazardous and unstable intermediates translates directly into reduced regulatory burdens and lower insurance costs associated with handling dangerous chemicals. By sourcing stable, commodity-grade starting materials like 3-methylpyridine-2-sulfonamide and halo-methoxypyridines, manufacturers can mitigate the risks of supply volatility that often plague specialty chemical markets. This stability ensures a continuous flow of materials, preventing production stoppages and enabling more accurate forecasting for downstream API manufacturing schedules.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the synthetic sequence and the avoidance of expensive, specialized reagents. By removing the multi-step preparation of sulfonyl chlorides and the associated waste treatment costs for malodorous thiols, the overall cost of goods sold is significantly decreased. Furthermore, the use of inexpensive inorganic bases and recoverable copper catalysts contributes to a leaner cost structure, allowing for competitive pricing in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on robust, shelf-stable raw materials fundamentally strengthens the supply chain resilience. Unlike traditional routes that depend on freshly prepared, unstable intermediates which degrade rapidly, this method utilizes starting materials that can be stockpiled safely for extended periods. This capability allows suppliers to maintain strategic inventory buffers, ensuring that they can meet sudden spikes in demand from pharmaceutical clients without facing the lead time delays associated with complex precursor synthesis.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this process is inherently greener and more scalable. The absence of toxic thiol emissions simplifies废气 (waste gas) treatment requirements, while the use of standard solvents facilitates easier recycling and recovery. The reaction conditions are mild enough to be safely scaled from gram-scale laboratory optimization to multi-ton industrial reactors using standard stainless steel equipment, ensuring that the technology can grow alongside the commercial needs of the drug development pipeline.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this sulfonamide intermediate. These insights are derived directly from the experimental data and technical specifications provided in the underlying patent literature, ensuring accuracy and relevance for industry professionals.
Q: Why is the Ullmann coupling method preferred over traditional sulfonyl chloride routes?
A: The Ullmann coupling method avoids the use of unstable and malodorous thiol precursors required for sulfonyl chloride synthesis, significantly improving operational safety and environmental compliance while maintaining high product purity.
Q: What are the critical reaction parameters for optimizing yield in this synthesis?
A: Critical parameters include maintaining a reaction temperature between 120-150°C, utilizing a molar excess of the halopyridine substrate (1:1.5 to 1:3 ratio), and selecting appropriate copper catalysts like cuprous iodide to ensure efficient C-N bond formation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available raw materials and standard heating methods (oil bath or microwave), making it highly adaptable for scale-up from laboratory grams to multi-ton commercial production without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(6-methoxypyridine-3-yl)-3-methylpyridine-2-sulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the Ullmann coupling methodology described in CN114149364A and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project never faces capacity bottlenecks. Our state-of-the-art facilities are designed to handle copper-catalyzed reactions with precision, supported by rigorous QC labs that enforce stringent purity specifications to meet the exacting demands of the pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for this valuable intermediate. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall procurement expenses. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us partner with you to accelerate your drug development timeline.
