Revolutionizing Miticidal Phenol Production: A Safer Two-Step Synthetic Strategy for Global Agrochemical Supply Chains
The global demand for effective acaricides continues to drive innovation in agrochemical intermediate manufacturing, particularly for compounds like Miticidal Phenol (Acefenol). A recent technological breakthrough detailed in patent CN114605265B introduces a transformative synthetic pathway that addresses long-standing safety and efficiency challenges in producing this critical active ingredient. Unlike traditional methods that rely on hazardous direct nitration, this novel approach utilizes a strategic two-step sequence involving halogenation followed by nitration. This shift not only mitigates the severe safety risks associated with mixed acid operations but also significantly enhances the overall yield and purity profile of the final product. For R&D directors and supply chain leaders, understanding this mechanistic pivot is essential for securing a reliable agrochemical intermediate supplier capable of delivering high-quality materials consistently.
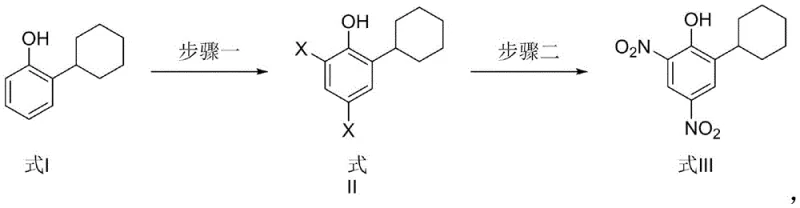
This patent specifically outlines a method starting from 2-cyclohexylphenol, a commercially accessible raw material, which undergoes controlled halogenation to form a di-halo intermediate before being converted into the target dinitrophenol. The significance of this development lies in its ability to bypass the inherent limitations of electrophilic aromatic substitution on deactivated rings. By decoupling the introduction of substituents into two distinct, manageable phases, the process offers a robust framework for commercial scale-up of complex phenolic derivatives. This report analyzes the technical merits of this invention, providing a comprehensive evaluation of its potential to redefine cost structures and supply reliability in the pesticide manufacturing sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Miticidal Phenol has been dominated by direct nitration protocols, a method fraught with significant operational and environmental drawbacks. The conventional reliance on concentrated nitric acid or mixed acid systems creates an inherently unstable reaction environment, posing severe safety risks such as thermal runaway and potential explosions during large-scale manufacturing. Furthermore, the electronic nature of the phenol ring presents a chemical challenge; once the first nitro group is introduced, the ring becomes strongly deactivated towards further electrophilic attack. This deactivation effect drastically reduces the reaction rate for the second nitration step, leading to incomplete reactions and the accumulation of mono-nitrated impurities that are difficult to separate. Consequently, manufacturers face low overall yields and substantial waste generation in the form of spent acid and nitrogen oxides, driving up both disposal costs and the carbon footprint of the production facility.
The Novel Approach
In stark contrast, the methodology disclosed in CN114605265B employs a clever retrosynthetic strategy that circumvents these electronic deactivation issues entirely. By first introducing halogen atoms at the 4 and 6 positions of the 2-cyclohexylphenol ring, the synthesis establishes the necessary substitution pattern under much milder conditions. Halogenation is generally more facile and controllable than direct dinitration, allowing for high conversion rates without the need for aggressive oxidizing acids. Subsequently, the halogen atoms serve as leaving groups in a nucleophilic substitution or displacement reaction with nitro donors, effectively installing the nitro groups without the kinetic barriers associated with direct nitration of a deactivated ring. This two-step sequence not only improves the safety profile by eliminating mixed acids but also ensures a cleaner reaction profile with fewer by-products, thereby simplifying downstream purification and enhancing the final product quality.
Mechanistic Insights into Halogenation-Nitration Sequence
The core of this innovative synthesis lies in the precise control of electrophilic and nucleophilic events on the aromatic ring. In the first step, the phenol substrate reacts with a halogen donor, such as N-bromosuccinimide (NBS) or copper bromide, in a polar aprotic solvent like acetonitrile or tetrahydrofuran. The hydroxyl group acts as a strong ortho/para director, guiding the halogenation specifically to the 4 and 6 positions relative to the hydroxyl group, while the 2-position is blocked by the cyclohexyl group. This regioselectivity is crucial for ensuring that the subsequent nitration occurs at the correct sites. The reaction temperature is maintained between 40°C and 60°C, a range that provides sufficient energy for the reaction to proceed efficiently while preventing side reactions or over-halogenation. The use of 2 to 8 equivalents of the halogen donor ensures complete conversion to the di-halo intermediate, which can be isolated as a high-purity solid through simple precipitation techniques.
The second step involves the displacement of the halogen atoms with nitro groups using nitro donors such as sodium nitrite or alkyl nitrites in acetic acid. This transformation is mechanistically distinct from classical electrophilic nitration. Under these conditions, the nitro species acts as a nucleophile or participates in a radical-mediated pathway to displace the halogen, a process facilitated by the electron-withdrawing nature of the developing transition state. The reaction is conducted at moderate temperatures of 50°C to 60°C, which are significantly lower than those required for traditional nitration, further enhancing process safety. The choice of solvent, preferably acetic acid, plays a dual role in solubilizing the intermediate and stabilizing the reactive nitro species. This mechanism effectively turns a thermodynamic disadvantage (deactivated ring) into a kinetic advantage by utilizing a leaving group strategy, resulting in yields that consistently exceed 96% in pilot examples, demonstrating exceptional process robustness.
How to Synthesize Miticidal Phenol Efficiently
The practical implementation of this synthesis route is designed for ease of operation and scalability, making it highly attractive for industrial adoption. The process begins with the dissolution of 2-cyclohexylphenol in a suitable organic solvent, followed by the controlled addition of the halogenating agent. Detailed standard operating procedures for temperature ramping, quenching, and extraction are critical to maintaining the high purity specifications required for agrochemical applications. The isolation of the di-halo intermediate serves as a key quality control checkpoint, ensuring that only the correct regioisomer proceeds to the final nitration step. Following the isolation, the intermediate is subjected to the nitration conditions described previously, with careful monitoring via thin-layer chromatography to determine the endpoint.
- Dissolve 2-cyclohexylphenol in an organic solvent like acetonitrile or THF, add a halogen donor (e.g., NBS or CuBr2), and react at 40-60°C to form a di-halo intermediate.
- Quench the reaction with water, extract with ethyl acetate or dichloromethane, and purify the di-halo intermediate by precipitation with a poor solvent.
- Dissolve the di-halo intermediate in acetic acid, add a nitro donor (e.g., sodium nitrite or tert-butyl nitrite), react at 50-60°C to substitute halogens with nitro groups, yielding Miticidal Phenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this halogenation-nitration pathway offers compelling economic and logistical benefits that extend beyond mere technical feasibility. The elimination of hazardous mixed acids translates directly into reduced regulatory compliance costs and lower insurance premiums for manufacturing facilities. Moreover, the ability to use a variety of commercially available halogen donors and nitro sources provides significant flexibility in sourcing strategies, allowing companies to mitigate supply chain disruptions by switching reagents without revalidating the entire process. The high yields reported in the patent examples imply a substantial reduction in raw material consumption per kilogram of finished product, which is a primary driver for cost reduction in pesticide manufacturing. Additionally, the simplified purification steps reduce solvent usage and waste treatment volumes, aligning with increasingly stringent environmental regulations globally.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive and hazardous nitrating agents with more economical halogen donors and mild nitro sources. The high yield of each step minimizes the loss of valuable starting materials, effectively lowering the cost of goods sold (COGS). Furthermore, the avoidance of complex waste acid treatment systems reduces capital expenditure on environmental control infrastructure. The use of common solvents like ethyl acetate and acetic acid, which are easily recovered and recycled, further contributes to long-term operational savings and sustainability goals.
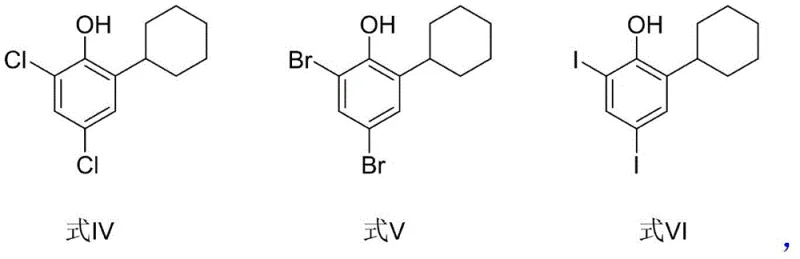
- Enhanced Supply Chain Reliability: By utilizing 2-cyclohexylphenol as a starting material, which is readily available from multiple global suppliers, the risk of raw material shortages is minimized. The flexibility to use different halogen donors (Cl, Br, I) means that if one specific reagent faces supply constraints, the process can be adapted to use an alternative without compromising the final output. This adaptability ensures continuous production schedules and reliable delivery timelines for downstream formulators. The robustness of the reaction conditions also reduces the likelihood of batch failures, ensuring a steady flow of high-purity intermediates to the market.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of explosive mixed acids make this process inherently safer for scale-up from pilot plant to multi-ton commercial production. The reduced generation of toxic nitrogen oxide gases and spent acid simplifies the environmental permitting process and lowers the burden on waste management teams. This green chemistry approach not only meets current regulatory standards but also future-proofs the manufacturing site against tightening environmental laws. The solid-state isolation of intermediates facilitates easy handling and transport within the facility, streamlining the overall production workflow.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or licensing agreements.
Q: Why is the halogenation-nitration route safer than direct nitration for Miticidal Phenol?
A: Direct nitration typically requires concentrated nitric acid or mixed acids, which pose high risks of explosion and generate significant acidic waste. The patented method uses milder nitro donors like alkyl nitrites or sodium nitrite in acetic acid, significantly reducing operational hazards and environmental pollution.
Q: What represents the key yield improvement in this new synthesis method?
A: Traditional direct nitration suffers from low yields because nitro groups are deactivating, making subsequent nitration difficult. By introducing halogens first, which are easier to install, and then displacing them with nitro groups, the process avoids the deactivation issue, achieving yields exceeding 96% in each step according to patent examples.
Q: Which halogen donors are compatible with this synthesis protocol?
A: The process is versatile and supports various halogen donors including N-bromosuccinimide (NBS), N-chlorosuccinimide (NCS), copper bromide, and copper iodide. This flexibility allows manufacturers to select reagents based on cost and availability without compromising the final product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Miticidal Phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN114605265B to maintain competitiveness in the global agrochemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity Miticidal Phenol that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in process optimization allows us to maximize the benefits of this novel halogenation-nitration strategy, offering our partners a superior product with a consistent quality profile.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term growth and sustainability objectives. Let us be your trusted partner in navigating the complexities of modern agrochemical synthesis.
