Advanced Manufacturing of 2-Haloimidazoles: A Strategic Breakthrough for Global Pharma Supply Chains
The global demand for high-purity heterocyclic building blocks continues to surge, driven by the relentless pace of drug discovery in oncology and antiviral therapeutics. Within this critical landscape, Patent CN101652350B introduces a transformative methodology for the production of 2-halogenated imidazole compounds, addressing long-standing inefficiencies in synthetic organic chemistry. This intellectual property outlines a robust, one-step protocol that converts readily available 2,4-dihaloimidazole precursors into valuable 2-haloimidazole derivatives using inexpensive iodine compounds and common organic solvents. For R&D directors and procurement strategists, this innovation represents a pivotal shift away from hazardous reagents and complex multi-step sequences toward a streamlined, cost-effective manufacturing paradigm. By leveraging simple halogen exchange mechanics, this technology not only enhances reaction yields but also drastically simplifies the downstream purification processes required for pharmaceutical-grade intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-haloimidazoles has been plagued by significant operational hazards and economic inefficiencies that burden both laboratory research and industrial scale-up. Traditional Route A, as depicted in the prior art, relies heavily on the use of n-butyllithium (n-BuLi), a pyrophoric reagent that demands stringent cryogenic conditions and specialized handling infrastructure to prevent catastrophic safety incidents. Furthermore, alternative pathways often necessitate the use of expensive 2-imidazolinone starting materials which suffer from inherently low reaction yields, thereby inflating the cost of goods sold. Other methods involving catalytic hydrogenation require precious metal catalysts like platinum, introducing supply chain vulnerabilities related to rare earth metal availability and necessitating rigorous heavy metal removal steps to meet regulatory purity standards.
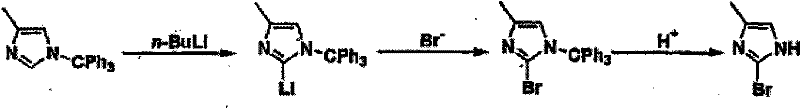
In addition to safety concerns, the regioselectivity issues inherent in older dehalogenation strategies often result in complex mixtures of by-products, such as 2-debrominated imidazoles, which are difficult and labor-intensive to separate. The reliance on sulfites or hydrogen gas in these legacy processes further complicates the workflow, requiring high-pressure equipment or generating substantial waste streams that challenge environmental compliance teams. These cumulative factors create a bottleneck in the supply of reliable pharmaceutical intermediate supplier networks, forcing manufacturers to absorb high overhead costs associated with safety protocols, waste disposal, and low-yield batch failures. Consequently, the industry has urgently required a method that bypasses these technical hurdles while maintaining the structural integrity of sensitive functional groups.
The Novel Approach
The breakthrough detailed in the patent data offers a sophisticated yet operationally simple solution by utilizing a halogen exchange mechanism mediated by iodide ions in the presence of specific organic additives. Instead of relying on dangerous organolithium reagents or expensive hydrogenation catalysts, this novel approach reacts 2,4-dihaloimidazole compounds with accessible iodine sources such as sodium iodide or potassium iodide. The reaction is facilitated by a solvent system comprising ketones, esters, or nitriles, which act not merely as diluents but as crucial participants in stabilizing the transition state and driving the equilibrium toward the desired 2-halo product. This strategic selection of reagents allows the transformation to proceed under moderate thermal conditions, typically between 60°C and 110°C, eliminating the need for cryogenic cooling or high-pressure vessels.
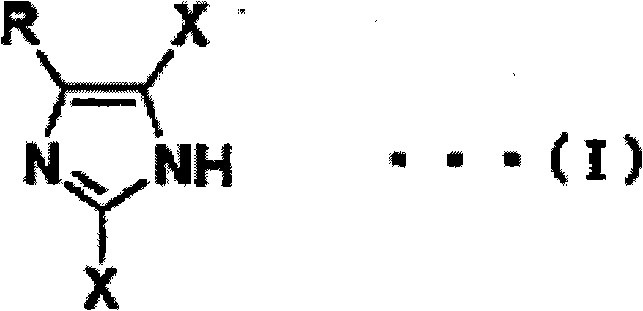
By shifting the synthetic strategy to this iodide-mediated pathway, manufacturers can achieve high conversion rates and superior purity profiles in a single operational step. The versatility of this method is evident in its compatibility with a wide range of substituents, including alkyl, aryl, nitro, and cyano groups, ensuring broad applicability across diverse drug scaffolds. This technological leap effectively decouples production efficiency from the volatility of precious metal markets and the logistical complexities of hazardous material transport. For supply chain heads, this translates to a more resilient sourcing strategy where lead times are reduced due to the simplicity of the reaction setup and the widespread availability of raw materials, ultimately securing a steady flow of critical intermediates for downstream API synthesis.
Mechanistic Insights into Iodide-Mediated Halogen Exchange
The core of this innovative process lies in the nucleophilic displacement capabilities of the iodide ion, which exhibits superior leaving group properties compared to chloride or bromide in specific solvent environments. When the 2,4-dihaloimidazole substrate interacts with the iodine compound, the iodide ion selectively targets the halogen at the 4-position (or 5-position depending on tautomerism), facilitating a halogen exchange that renders the adjacent halogen at the 2-position more susceptible to retention or specific modification. The presence of ketone or ester solvents plays a pivotal mechanistic role by coordinating with the metal cation of the iodide salt, thereby increasing the nakedness and nucleophilicity of the iodide anion. This solvation effect lowers the activation energy barrier for the substitution reaction, allowing it to proceed efficiently at elevated temperatures without the need for aggressive catalysts.
Furthermore, the reaction conditions are meticulously tuned to minimize side reactions such as complete dehalogenation or over-iodination, which are common pitfalls in less controlled environments. The patent data indicates that by controlling the stoichiometry of the iodine compound—preferably using at least 1.5 equivalents relative to the dihalo precursor—the system drives the equilibrium toward the formation of the mono-halo product with high fidelity. Impurity control is inherently built into the mechanism; the specific interaction between the iodide source and the carbonyl-containing solvent suppresses the formation of 2-debrominated by-products that plague sulfite-based reduction methods. This high degree of chemoselectivity ensures that sensitive functional groups on the imidazole ring remain intact, preserving the molecular complexity required for advanced medicinal chemistry applications.
How to Synthesize 2-Haloimidazole Efficiently
Implementing this synthesis route in a production environment requires careful attention to reagent quality and thermal management to maximize yield and purity. The process begins with the preparation of a reaction mixture containing the 2,4-dihaloimidazole starting material and a slight excess of an inorganic iodide salt, such as sodium iodide or lithium iodide. To this solid mixture, a selected organic solvent from the ketone, ester, or nitrile family is added; preferred embodiments suggest using cyclohexanone, ethyl acetate, or butyronitrile to optimize reaction kinetics. The detailed standardized synthesis steps see the guide below outline the precise heating profiles and workup procedures necessary to isolate the crystalline product.
- Mix 2,4-dihaloimidazole starting material with an excess of an iodine compound such as sodium iodide.
- Add a solvent system comprising ketones, esters, or nitriles to facilitate the halogen exchange reaction.
- Heat the mixture to 60-110°C for several hours, then isolate the product via crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iodide-mediated synthesis route offers profound advantages that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. By eliminating the dependency on pyrophoric reagents like n-butyllithium and precious metal catalysts like platinum, the process fundamentally alters the cost structure of producing 2-haloimidazole intermediates. This shift reduces the capital expenditure required for specialized safety infrastructure and high-pressure reactors, allowing facilities to utilize standard glass-lined or stainless-steel batch reactors. The simplification of the workflow also minimizes the manpower hours needed for reaction monitoring and hazard mitigation, freeing up technical resources for other value-added activities within the plant.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with commodity chemicals that are globally available at stable prices. The elimination of platinum catalysts removes a significant variable cost component and avoids the complex, costly procedures associated with scavenging trace heavy metals from the final product to meet pharmacopeial limits. Additionally, the high conversion rates observed in the patent examples mean that less raw material is wasted, improving the overall atom economy of the synthesis. This efficiency translates into substantial cost savings per kilogram of produced intermediate, providing a competitive edge in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of ubiquitous raw materials such as sodium iodide and common organic solvents, which are not subject to the same geopolitical supply constraints as rare earth metals or specialized organolithium solutions. The robustness of the reaction conditions, which tolerate moderate temperature fluctuations and do not require inert atmosphere gloveboxes for reagent handling, reduces the risk of batch failures due to minor operational deviations. This reliability ensures consistent delivery schedules for clients, mitigating the risks of production stoppages that can ripple through the entire pharmaceutical supply network. Furthermore, the simplified logistics of storing stable solid iodide salts versus pressurized hydrogen or cryogenic liquids enhances warehouse safety and reduces insurance premiums.
- Scalability and Environmental Compliance: Scaling this process from laboratory benchtop to multi-ton commercial production is straightforward due to the absence of exothermic hazards associated with organolithium additions or high-pressure hydrogenation steps. The reaction generates minimal hazardous waste, as the by-products are primarily inorganic salts that can be easily separated and disposed of according to standard environmental protocols. This green chemistry profile aligns with increasingly stringent global environmental regulations, reducing the regulatory burden on manufacturing sites. The ability to run the reaction in common solvents also facilitates easier solvent recovery and recycling, further enhancing the sustainability metrics of the manufacturing process and appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy techniques. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the iodide-mediated method superior to n-BuLi lithiation?
A: The iodide-mediated method eliminates the need for cryogenic conditions and pyrophoric n-butyllithium, significantly improving operational safety and reducing equipment costs compared to traditional lithiation routes.
Q: What represents the primary cost driver in this new synthesis route?
A: The primary cost drivers are the inexpensive inorganic iodide salts and common organic solvents like cyclohexanone, replacing expensive platinum catalysts or specialized imidazolinone precursors.
Q: Can this process be scaled for industrial production?
A: Yes, the process operates at moderate temperatures (60-110°C) without high-pressure hydrogenation, making it highly suitable for large-scale batch reactors in standard chemical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Haloimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this iodide-mediated process are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-haloimidazole intermediate meets the exacting standards required for global pharmaceutical registration. We understand that consistency is key, and our process engineering teams are dedicated to optimizing every parameter to deliver a product that is both cost-effective and chemically robust.
We invite forward-thinking procurement leaders and R&D directors to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our implementation of Patent CN101652350B can drive efficiency and reliability in your supply chain. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
