Advanced Hinokitiol Manufacturing: Optimizing Yield and Scalability for Global Supply Chains
Advanced Hinokitiol Manufacturing: Optimizing Yield and Scalability for Global Supply Chains
The global demand for high-purity hinokitiol, also known as chamaecypariol, has surged due to its potent antimicrobial and anti-inflammatory properties in pharmaceutical and personal care applications. Patent CN101602656A introduces a transformative synthetic methodology that addresses the longstanding inefficiencies of traditional production routes. This innovation centers on a novel ring expansion strategy that utilizes a specific non-aqueous solvent system, fundamentally altering the economic and operational landscape for manufacturers. By shifting away from aqueous reaction media to a carefully balanced toluene-acetate mixture, the process achieves a dramatic enhancement in conversion efficiency while simultaneously simplifying downstream purification. For R&D directors and procurement specialists, this represents a critical opportunity to secure a reliable hinokitiol supplier capable of delivering consistent quality at a reduced cost basis. The technical breakthrough lies not merely in the chemical transformation itself, but in the precise optimization of reaction conditions that allow for industrial scalability without compromising purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of hinokitiol has been plagued by significant technical bottlenecks that hinder cost-effective mass production. Prior art methods, such as those utilizing aqueous acetone systems or harsh organometallic reagents like ethylmagnesium bromide, suffer from inherently low yields and complex workup procedures. For instance, conventional aqueous ring expansion processes often struggle to exceed yields of approximately 30%, primarily due to side reactions and the formation of irretrievable byproducts that contaminate the final stream. Furthermore, the reliance on strict anhydrous conditions for Grignard-type reagents necessitates expensive solvent drying protocols and specialized equipment, driving up capital expenditure. The generation of substantial volumes of industrial wastewater containing organic residues poses a severe environmental compliance burden, requiring costly treatment infrastructure. These factors collectively render many traditional pathways economically unviable for large-scale cost reduction in fine chemical manufacturing, forcing buyers to rely on limited suppliers with inflated pricing structures.
The Novel Approach
The methodology disclosed in the patent data offers a robust solution by replacing problematic aqueous environments with a tailored organic solvent system. The core innovation involves the use of toluene in conjunction with sodium hydroxide and acetate during the critical ring expansion step. This specific combination creates a reaction environment that stabilizes the transition states involved in the rearrangement of the ketene adduct, thereby minimizing decomposition pathways. Unlike previous methods that required complex extraction steps to separate the product from water-miscible solvents, this new approach leverages toluene's immiscibility with water to facilitate direct phase separation after acidification. The result is a streamlined workflow that drastically reduces processing time and solvent consumption. By achieving yields that substantially surpass prior art benchmarks, this route provides a compelling value proposition for supply chain managers seeking to mitigate risk and ensure continuity of supply for high-value pharmaceutical intermediates.
Mechanistic Insights into Toluene-Mediated Ring Expansion
The success of this synthetic route hinges on the precise control of isomerization and the subsequent cycloaddition kinetics. The process begins with the generation of cyclopentadienyl potassium, which is alkylated to form sec-propyl cyclopentadiene. A crucial mechanistic feature is the spontaneous isomerization of the 5-isomer to the 1-isomer upon standing at room temperature, ensuring that the subsequent reaction with dichloroacetyl chloride proceeds with high regioselectivity. This selectivity is vital because only the 1-isomer leads to the desired hinokitiol skeleton upon ring expansion. The addition of dichloroacetyl chloride forms a ketene adduct, which serves as the precursor for the seven-membered ring formation. The presence of triethylamine acts as a proton scavenger, driving the equilibrium forward and preventing the accumulation of acidic byproducts that could degrade the sensitive intermediate. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for optimal throughput.
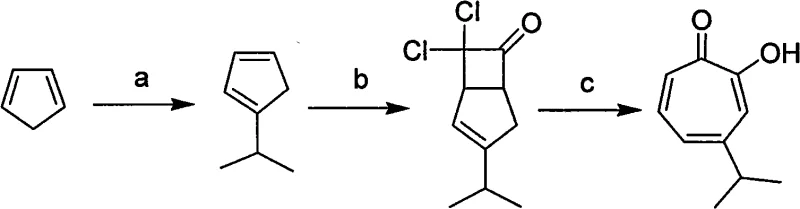
The pivotal ring expansion step is where the novel solvent system exerts its most profound influence. In traditional aqueous systems, the hydrolysis of the ketene adduct competes with the desired rearrangement, leading to significant material loss. However, in the toluene-acetate system, the local concentration of hydroxide ions is modulated by the organic medium, favoring the intramolecular rearrangement over hydrolysis. The molar ratio of ketene adduct to sodium hydroxide to acetate to toluene is maintained within a narrow window (approximately 1:3-4:10-14:10-14) to maximize this effect. This specific stoichiometry ensures that the base is available to deprotonate the intermediate without causing excessive degradation. Furthermore, the use of toluene facilitates the dissolution of the organic intermediate while keeping the inorganic salts in a separate phase or suspension, creating a biphasic-like environment that enhances mass transfer. This mechanistic understanding underscores why the process delivers high-purity hinokitiol with minimal impurity profiles, meeting the stringent requirements of regulatory bodies for active ingredients.
How to Synthesize Hinokitiol Efficiently
Implementing this synthesis requires strict adherence to the specified reagent ratios and temperature controls to replicate the high yields observed in the patent examples. The process is designed to be scalable, moving seamlessly from laboratory benchtop to pilot plant operations with minimal re-optimization. Operators must ensure that the isomerization of the cyclopentadiene derivative is allowed to proceed to completion before introducing the dichloroacetyl chloride, as this dictates the final isomeric purity. The subsequent ring expansion must be conducted under reflux conditions to provide the necessary activation energy for the rearrangement. Detailed standard operating procedures regarding the addition rates of reagents and the specific workup protocols are essential for maintaining batch-to-batch consistency. The following guide outlines the standardized synthesis steps derived from the patented technology.
- Generate cyclopentadienyl potassium by reacting cyclopentadiene with potassium hydroxide in DMSO, followed by alkylation with isopropyl bromide to form 1-sec-propyl cyclopentadiene.
- React the 1-sec-propyl cyclopentadiene with dichloroacetyl chloride and triethylamine in n-hexane to form the ketene adduct intermediate.
- Perform ring expansion by refluxing the ketene adduct in a specific molar ratio mixture of sodium hydroxide, acetate, and toluene, followed by acidification and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible operational benefits and risk mitigation. The primary advantage lies in the substantial improvement in overall process yield, which effectively lowers the cost of goods sold (COGS) per kilogram of finished product. By minimizing the loss of raw materials to side reactions and waste streams, manufacturers can offer more competitive pricing without sacrificing margin. Additionally, the use of commodity chemicals such as cyclopentadiene, toluene, and sodium hydroxide ensures that the supply chain is resilient against fluctuations in the availability of exotic or specialized reagents. This reliance on widely available feedstocks reduces the risk of production stoppages due to raw material shortages, a critical factor for maintaining commercial scale-up of complex pharmaceutical intermediates. The simplified purification process also means faster turnaround times from reactor to warehouse, enhancing the agility of the supply response.
- Cost Reduction in Manufacturing: The elimination of expensive drying agents and the reduction in solvent volume requirements lead to significant operational savings. Traditional methods often require rigorous solvent purification to remove trace water, which consumes energy and time; this new method tolerates the reaction conditions more robustly, lowering utility costs. Furthermore, the higher yield means that less raw material is needed to produce the same amount of final product, directly impacting the variable cost structure. The ability to use toluene as both a reaction solvent and an extraction agent removes the need for purchasing and managing multiple distinct solvent types, streamlining inventory management and reducing procurement complexity.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for greater flexibility in manufacturing scheduling. Unlike processes that require cryogenic temperatures or inert atmosphere gloveboxes, this synthesis can be performed in standard stainless steel reactors with conventional cooling systems. This accessibility means that more contract manufacturing organizations (CMOs) have the capability to produce the material, diversifying the supplier base and reducing single-source dependency. The stability of the intermediates also allows for potential campaign manufacturing, where batches can be held at certain stages without degradation, providing a buffer against demand spikes and ensuring reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the reduction in wastewater generation is a major advantage. Aqueous waste streams containing organic residues are costly to treat and dispose of; by shifting to an organic-dominated system, the volume of hazardous waste is minimized. This aligns with increasingly strict global environmental regulations, future-proofing the manufacturing process against tighter compliance standards. The simplified workup also reduces the exposure of personnel to hazardous chemicals during the isolation phase, improving workplace safety metrics. These factors combined make the process highly attractive for long-term investment and commercial scale-up in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of hinokitiol using this advanced methodology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding specification alignment and process validation. The answers reflect the consensus on the technical superiority of the toluene-based system over legacy aqueous methods.
Q: What is the primary advantage of the toluene-based solvent system in hinokitiol synthesis?
A: The use of a non-aqueous toluene-acetate system significantly increases reaction yield (up to 81% compared to ~31% in aqueous methods) and simplifies product isolation by utilizing toluene as both a reaction medium and extraction solvent.
Q: How does this method address environmental concerns in fine chemical manufacturing?
A: By eliminating the need for large volumes of water and avoiding difficult-to-separate mixed solvent systems, this process reduces industrial wastewater generation and lowers the energy consumption required for solvent recovery.
Q: Is the 1-sec-propyl cyclopentadiene isomer stable during the process?
A: Yes, the process includes a controlled isomerization step where the 5-isomer converts to the thermodynamically more stable 1-isomer at room temperature, ensuring high regioselectivity before the ketene addition step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hinokitiol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis are fully realized in practice. We operate state-of-the-art facilities equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling via HPLC and GC-MS. Our commitment to quality ensures that every batch of hinokitiol meets the exacting standards required for pharmaceutical and high-end cosmetic applications, providing our partners with absolute confidence in the material's performance and safety profile.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your organization. We encourage you to request specific COA data and route feasibility assessments to validate the compatibility of our material with your downstream processes. Together, we can establish a sustainable and efficient supply partnership that drives innovation and value in the global marketplace.
