Revolutionizing Lipid-Lowering Drug Production: A Deep Dive into Green Ciprofibrate Synthesis
The pharmaceutical industry is currently undergoing a significant paradigm shift towards greener, more sustainable manufacturing processes, particularly in the synthesis of critical lipid-lowering agents. Patent CN103373916A represents a pivotal advancement in this domain, detailing an environment-friendly preparation method for Ciprofibrate, also known chemically as Win-35833. This specific intellectual property outlines a novel synthetic route that fundamentally alters the production landscape by utilizing 4-Vinyl phenol as a primary raw material to execute Bargellini and annulation reactions. For R&D Directors and technical decision-makers, this patent offers a compelling alternative to legacy methods that have long been plagued by safety hazards and inefficiencies. The technology described herein not only addresses the urgent need for reduced environmental footprints but also aligns perfectly with the strategic goals of a reliable pharmaceutical intermediates supplier seeking to optimize process robustness. By transitioning away from hazardous oxidants and corrosive Lewis acids, this method establishes a new benchmark for industrial feasibility and operational safety in the manufacture of high-purity lipid-lowering drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ciprofibrate has relied heavily on multi-step pathways that introduce significant operational risks and cost inefficiencies into the supply chain. Traditional routes, such as those disclosed in earlier patents like CN1514819, typically necessitate the use of Friedel-Crafts acylation followed by Baeyer-Villiger rearrangement. These conventional methodologies are inherently problematic because they require the utilization of aluminum chloride, a corrosive Lewis acid that generates substantial amounts of acidic waste and complicates post-reaction processing. Furthermore, the reliance on peroxy acids for the rearrangement step introduces severe safety hazards, including the risk of explosion and thermal runaway during large-scale production. The cumulative effect of these dangerous reagents, combined with a reaction sequence that often exceeds four distinct steps, results in a fragmented production cycle with lower overall atom economy. For procurement managers, these factors translate into higher costs for waste disposal, specialized safety equipment, and complex regulatory compliance, making the traditional approach increasingly untenable in a modern, cost-sensitive market.
The Novel Approach
In stark contrast to the cumbersome legacy protocols, the innovative method described in CN103373916A streamlines the entire synthesis into a concise two-step process that prioritizes both safety and efficiency. This novel approach leverages the Bargellini reaction to construct the core ether linkage, followed immediately by an annulation reaction to form the cyclopropane ring, effectively bypassing the need for dangerous peroxy acids entirely. The visual representation of this streamlined pathway highlights the direct conversion of 4-Vinyl phenol into the final active pharmaceutical ingredient through a series of controlled, mild transformations. 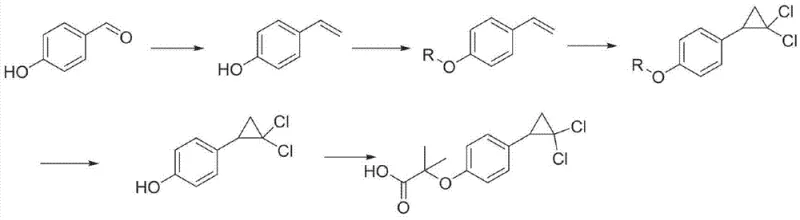 . By eliminating the intermediate isolation steps and hazardous reagents associated with the Baeyer-Villiger oxidation, this new route drastically simplifies the operational workflow. For supply chain heads, this reduction in complexity means a shorter production cycle and a more predictable manufacturing timeline, which are critical factors for ensuring the continuous availability of high-purity pharmaceutical intermediates in a global market.
. By eliminating the intermediate isolation steps and hazardous reagents associated with the Baeyer-Villiger oxidation, this new route drastically simplifies the operational workflow. For supply chain heads, this reduction in complexity means a shorter production cycle and a more predictable manufacturing timeline, which are critical factors for ensuring the continuous availability of high-purity pharmaceutical intermediates in a global market.
Mechanistic Insights into Bargellini and Phase Transfer Catalyzed Annulation
From a mechanistic perspective, the success of this synthesis relies on the precise control of carbene generation and phase transfer dynamics. In the first step, the reaction between 4-Vinyl phenol and chloroform under basic conditions generates a dichlorocarbene intermediate, which subsequently reacts to form the 2-methyl-2-(4-vinyl phenoxy) propionic acid precursor. The second step utilizes a phase-transfer catalyst, such as tetrabutyl ammonium bromide or benzyltriethylammonium chloride, to facilitate the interaction between the organic substrate and the aqueous alkaline phase. This catalytic system allows the reaction to proceed efficiently at mild temperatures ranging from -5°C to 70°C, avoiding the extreme thermal conditions that often degrade sensitive functional groups. For R&D teams focused on impurity profiles, this mild condition is paramount; it minimizes the formation of side products and polymerization of the vinyl group, ensuring a cleaner reaction mixture. The ability to fine-tune the molar ratios of alkali and chloroform further enhances the selectivity of the reaction, providing a robust framework for scaling up without compromising the chemical integrity of the final molecule.
Furthermore, the purification strategy embedded within this patent demonstrates a sophisticated understanding of crystallization thermodynamics to ensure exceptional product quality. The process involves adjusting the pH of the reaction mixture to between 2 and 3 using hydrochloric acid, which precipitates the product while leaving soluble impurities in the aqueous phase. Subsequent extraction with ethyl acetate and drying with anhydrous sodium sulfate removes residual water and organic contaminants before the final recrystallization from n-hexane. The consistency of the melting point data across multiple embodiments, specifically the sharp range of 114-115°C, serves as a definitive indicator of high crystalline purity. This level of quality control is essential for meeting the stringent specifications required by regulatory bodies for API intermediates. By integrating these rigorous purification steps directly into the standard operating procedure, the method guarantees that the final output meets the rigorous standards expected of a high-purity lipid-lowering drug intermediate, thereby reducing the burden on downstream quality assurance teams.
How to Synthesize Ciprofibrate Efficiently
Implementing this synthesis route requires a disciplined approach to reagent preparation and reaction monitoring to maximize yield and safety. The process begins with the careful dissolution of 4-Vinyl phenol in acetone, followed by the controlled addition of alkali and chloroform to initiate the Bargellini reaction under specific temperature constraints. Once the intermediate is secured, the second phase involves dissolving the acid in a suitable solvent system and introducing the phase-transfer catalyst to drive the cyclization. Detailed standardized synthetic steps see the guide below, which outlines the precise stoichiometric ratios and thermal profiles necessary for reproducibility. Adhering to these parameters ensures that the reaction proceeds with the high molar yields reported in the patent embodiments, typically exceeding 90% for individual steps. This structured approach not only facilitates technology transfer from the laboratory to the pilot plant but also provides a clear roadmap for troubleshooting potential deviations in large-scale manufacturing environments.
- Dissolve 4-Vinyl phenol in acetone, add alkali and chloroform to perform the Bargellini reaction, yielding 2-methyl-2-(4-vinyl phenoxy) propionic acid.
- Dissolve the intermediate in a solvent mixture, add aqueous alkali and a phase-transfer catalyst to initiate the annulation reaction.
- Adjust pH to 2-3 with hydrochloric acid, extract with ethyl acetate, and recrystallize from n-hexane to obtain pure Ciprofibrate.
Commercial Advantages for Procurement and Supply Chain Teams
The transition to this novel synthesis method offers profound commercial benefits that extend far beyond simple chemical efficiency, directly impacting the bottom line for procurement and supply chain operations. By replacing hazardous and expensive reagents like peroxy acids and aluminum chloride with conventional soda ash and recoverable solvents, the process fundamentally alters the cost structure of manufacturing. This shift eliminates the need for specialized containment systems required for explosive oxidants, thereby reducing capital expenditure on safety infrastructure. Additionally, the reduction in reaction steps from four or more down to just two significantly lowers labor costs and energy consumption per kilogram of product. For procurement managers, these efficiencies translate into a more competitive pricing model without sacrificing quality, allowing for better margin management in the final drug product. The use of readily available raw materials like 4-Vinyl phenol further insulates the supply chain from volatility associated with niche specialty chemicals, ensuring a stable and reliable source of input materials for continuous production.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and hazardous oxidants removes the necessity for expensive heavy metal removal steps and complex waste treatment protocols. This simplification of the downstream processing workflow leads to substantial cost savings in utilities and consumables. Furthermore, the high total yield of the process means that less raw material is wasted, optimizing the atom economy and reducing the cost of goods sold. The ability to recover and reuse organic solvents like acetone and chloroform adds another layer of financial efficiency, minimizing the recurring expense of solvent procurement. These cumulative factors create a lean manufacturing environment that is highly resilient to market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium hydroxide and chloroform, which are produced globally in vast quantities, ensures that the supply chain is not vulnerable to the bottlenecks often seen with specialized reagents. The mild reaction conditions, operating safely between -10°C and 70°C, reduce the risk of unplanned shutdowns due to thermal excursions or safety incidents. This operational stability is crucial for maintaining consistent delivery schedules to downstream API manufacturers. By adopting a process that is inherently safer and easier to control, supply chain heads can guarantee greater continuity of supply, mitigating the risks of production delays that could impact the availability of life-saving lipid-lowering medications in the global market.
- Scalability and Environmental Compliance: The design of this synthesis route is intrinsically scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without significant re-engineering. The generation of harmless byproducts like sodium chloride and potassium chloride simplifies wastewater treatment, ensuring compliance with increasingly stringent environmental regulations. This eco-friendly profile enhances the corporate sustainability metrics of the manufacturer, appealing to partners who prioritize green chemistry principles. The ease of post-processing, characterized by simple filtration and crystallization steps, allows for rapid scale-up of batch sizes, enabling manufacturers to respond quickly to surges in market demand. This scalability ensures that the production capacity can grow in tandem with the commercial success of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical advantages of this method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The insights provided here bridge the gap between theoretical patent claims and real-world industrial application, ensuring that stakeholders have a comprehensive understanding of the value proposition.
Q: Why is the new Ciprofibrate synthesis method considered more environmentally friendly than traditional routes?
A: Traditional methods rely on hazardous peroxy acids for Baeyer-Villiger rearrangement and corrosive aluminum chloride for Friedel-Crafts acylation. The new patent CN103373916A eliminates these dangerous reagents, using conventional soda acid and recoverable organic solvents instead, resulting in harmless salt byproducts and significantly reduced environmental impact.
Q: How does this two-step process improve overall production yield compared to prior art?
A: By condensing the synthesis into only two main reaction steps (Bargellini and annulation) rather than the four or more steps required in previous methods, the cumulative loss of material is drastically minimized. The patent data demonstrates a total molar yield approaching 80%, which is a substantial improvement over the 56% to 63% yields typical of older multi-step pathways.
Q: What specific quality indicators confirm the high purity of the synthesized Ciprofibrate?
A: The consistency of the melting point across multiple embodiments serves as a primary indicator of purity. The synthesized product consistently exhibits a sharp melting point range of 114-115°C. A narrow melting point range indicates a homogeneous crystal lattice with minimal impurity inclusion, confirming the effectiveness of the purification and reaction control mechanisms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ciprofibrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CN103373916A process are translated into tangible supply chain solutions. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which verify that every batch of Ciprofibrate meets the highest international standards. We understand that for R&D Directors and Procurement Managers, consistency and reliability are non-negotiable, and our infrastructure is designed to deliver exactly that. By leveraging our technical expertise, we can help you navigate the complexities of green chemistry implementation while maintaining the highest levels of product integrity and safety.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out to us to request specific COA data and route feasibility assessments that demonstrate the practical benefits of partnering with a reliable pharmaceutical intermediates supplier. Together, we can drive the next generation of efficient, sustainable, and high-quality lipid-lowering drug production, ensuring that your supply chain remains robust and competitive in an ever-changing industry landscape.
