Scalable Synthesis of 3-Aminomethyltetrahydrofuran for Advanced Agrochemical Manufacturing
Scalable Synthesis of 3-Aminomethyltetrahydrofuran for Advanced Agrochemical Manufacturing
The global demand for third-generation neonicotinoid insecticides, particularly dinotefuran, has necessitated the development of robust and economically viable synthetic routes for key intermediates like 3-aminomethyltetrahydrofuran. Patent CN107417648B discloses a groundbreaking four-step synthesis method that fundamentally shifts the production paradigm from hazardous, high-cost legacy processes to a streamlined, industrially friendly protocol. By utilizing maleic acid diesters as the primary feedstock, this novel approach leverages Michael addition, metal borohydride reduction, dehydration cyclization, and catalytic hydrogenation to achieve superior yields while mitigating environmental impact. For R&D directors and procurement strategists in the agrochemical sector, this technology represents a critical opportunity to optimize supply chains and enhance the purity profiles of final API intermediates without compromising on safety or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aminomethyltetrahydrofuran has been plagued by significant technical and economic bottlenecks that hinder efficient commercial manufacturing. One prominent legacy route involves the use of malic acid as a starting material, which undergoes catalytic hydrogenation to form 1,2,4-butanetriol, followed by cyclization and chlorination. 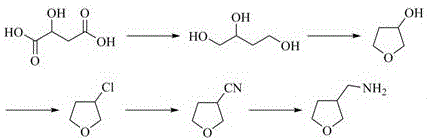 This pathway is critically flawed due to the high cost of malic acid and the necessity of using virulent sodium cyanide for nucleophilic substitution, which introduces severe occupational health hazards and complex waste treatment requirements. Another common method relies on 2,3-dihydrofuran reacting with trichloroacetyl chloride, a process that demands the use of expensive and dangerous reducing agents like lithium aluminum hydride (LiAlH4). These conventional methodologies not only suffer from low overall yields and complex operational procedures but also generate substantial hazardous waste, making them increasingly untenable in the face of modern environmental regulations and cost-reduction mandates.
This pathway is critically flawed due to the high cost of malic acid and the necessity of using virulent sodium cyanide for nucleophilic substitution, which introduces severe occupational health hazards and complex waste treatment requirements. Another common method relies on 2,3-dihydrofuran reacting with trichloroacetyl chloride, a process that demands the use of expensive and dangerous reducing agents like lithium aluminum hydride (LiAlH4). These conventional methodologies not only suffer from low overall yields and complex operational procedures but also generate substantial hazardous waste, making them increasingly untenable in the face of modern environmental regulations and cost-reduction mandates.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the innovative method described in patent CN107417648B offers a streamlined, four-step trajectory that maximizes atom economy and operational safety. The process initiates with a Michael addition between readily available maleic acid diesters and nitromethane, bypassing the need for exotic or prohibitively expensive starting materials. 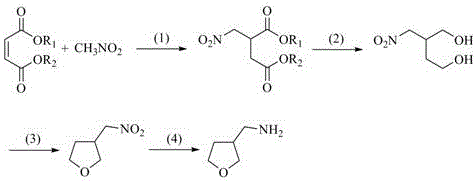 This is followed by a controlled reduction using metal borohydrides, which are significantly safer and easier to handle than lithium aluminum hydride. The subsequent dehydration cyclization and final catalytic hydrogenation steps proceed under mild conditions, eliminating the need for highly corrosive reagents like phosphorus oxychloride or explosive azides. By replacing toxic cyanide substitutions and harsh reduction conditions with this elegant sequence, the novel approach drastically simplifies the purification workflow, reduces the impurity burden on downstream processing, and establishes a foundation for consistent, high-quality production suitable for reliable agrochemical intermediate supplier networks.
This is followed by a controlled reduction using metal borohydrides, which are significantly safer and easier to handle than lithium aluminum hydride. The subsequent dehydration cyclization and final catalytic hydrogenation steps proceed under mild conditions, eliminating the need for highly corrosive reagents like phosphorus oxychloride or explosive azides. By replacing toxic cyanide substitutions and harsh reduction conditions with this elegant sequence, the novel approach drastically simplifies the purification workflow, reduces the impurity burden on downstream processing, and establishes a foundation for consistent, high-quality production suitable for reliable agrochemical intermediate supplier networks.
Mechanistic Insights into Maleic Diester-Based Cyclization
The core chemical ingenuity of this synthesis lies in the strategic manipulation of the carbon backbone through a sequence of well-defined mechanistic transformations. The initial Michael addition exploits the electron-deficient nature of the maleic acid diester double bond, allowing the nitromethane anion, generated under mild alkaline catalysis, to attack and form the crucial 2-nitromethyl-1,4-succinic acid diester skeleton. This step is pivotal as it installs both the nitro functionality required for the final amine and the ester groups that serve as handles for subsequent reduction. Following this, the metal borohydride reduction selectively converts the ester moieties into primary alcohols without affecting the nitro group, yielding 2-nitromethyl-1,4-butanediol. This chemoselectivity is essential for maintaining the integrity of the nitrogen-containing functional group while preparing the molecule for ring closure.
The cyclization mechanism involves an acid-catalyzed dehydration where the hydroxyl groups of the butanediol intermediate undergo intramolecular nucleophilic attack to form the tetrahydrofuran ring, expelling water in the process. The use of catalysts like p-toluenesulfonic acid facilitates this transformation at moderate temperatures, ensuring high conversion rates while minimizing side reactions such as polymerization or ether formation. Finally, the catalytic hydrogenation step utilizes standard heterogeneous catalysts like Pd/C or Raney nickel to reduce the nitro group to the primary amine. This final reduction is clean and efficient, avoiding the formation of hydroxylamine byproducts that often complicate purification in other synthetic routes. The cumulative effect of these mechanistic choices is a process that inherently controls the impurity profile, delivering a high-purity intermediate that meets the stringent specifications required for pharmaceutical and agrochemical applications.
How to Synthesize 3-Aminomethyltetrahydrofuran Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety at every stage. The process begins with the preparation of the nitro-succinate intermediate, followed by careful reduction and cyclization steps that demand specific temperature and pH controls. The detailed standardized synthesis steps, including exact molar ratios, solvent choices, and workup procedures validated by experimental examples, are outlined below to ensure reproducibility and quality control.
- Perform Michael addition of nitromethane to maleic acid diester under alkaline conditions to form 2-nitromethyl-1,4-succinic acid diester.
- Reduce the diester intermediate using metal borohydride in an alcohol-ether solvent system to generate 2-nitromethyl-1,4-butanediol.
- Execute dehydration cyclization of the butanediol derivative using an acid catalyst like p-toluenesulfonic acid to form the tetrahydrofuran ring.
- Complete the synthesis via catalytic hydrogenation reduction of the nitro group to obtain the final 3-aminomethyltetrahydrofuran product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this maleic diester-based methodology offers profound strategic advantages that extend far beyond simple chemical efficiency. The elimination of hazardous reagents like sodium cyanide and lithium aluminum hydride removes significant regulatory burdens and insurance costs associated with handling extremely toxic or pyrophoric materials. This shift not only safeguards the workforce but also streamlines the logistics of raw material sourcing, as maleic acid diesters and nitromethane are commodity chemicals with stable, global supply chains. Consequently, manufacturers can achieve substantial cost savings by avoiding the premium pricing of specialized reagents and the expensive waste disposal fees mandated for cyanide-containing effluents.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of high-cost starting materials like malic acid with inexpensive maleic acid diesters, which are produced on a massive industrial scale. Furthermore, the replacement of lithium aluminum hydride with metal borohydrides significantly lowers reagent costs, as borohydrides are not only cheaper but also allow for simpler quenching and workup procedures that reduce labor and utility consumption. By removing the need for complex protection-deprotection strategies or harsh chlorination steps found in older routes, the overall process mass intensity is improved, leading to a drastic reduction in solvent usage and energy requirements per kilogram of product.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks ensures that production is not vulnerable to the supply shocks often associated with niche or highly regulated chemicals. The robustness of the four-step sequence means that yield losses are minimized, providing a more predictable output volume for long-term contracts. Additionally, the mild reaction conditions reduce equipment wear and tear, decreasing maintenance downtime and ensuring continuous operation capabilities that are critical for meeting tight delivery schedules in the agrochemical sector.
- Scalability and Environmental Compliance: The absence of heavy metals and toxic cyanides simplifies the environmental compliance landscape, facilitating easier permitting for plant expansion and scale-up. The process generates less hazardous waste, aligning with green chemistry principles and corporate sustainability goals. This environmental friendliness translates directly into operational agility, allowing manufacturers to scale from pilot batches to multi-ton commercial production without encountering the regulatory bottlenecks that typically plague processes involving highly toxic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on safety, scalability, and product quality for potential partners.
Q: Why is the maleic diester route safer than traditional cyanide-based methods?
A: Traditional methods often utilize sodium cyanide for nucleophilic substitution, posing severe toxicity risks. The patented maleic diester route eliminates cyanide entirely, relying instead on safer Michael addition and borohydride reduction steps.
Q: What are the cost advantages of using maleic acid diesters over malic acid?
A: Maleic acid diesters are significantly more abundant and cost-effective compared to malic acid. Furthermore, the new process avoids expensive reagents like lithium aluminum hydride, drastically reducing raw material expenditure.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process features mild reaction conditions, simple operational steps, and high yields across all four stages, making it highly adaptable for commercial scale-up from pilot plants to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminomethyltetrahydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemicals depends on the reliability and purity of key intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-aminomethyltetrahydrofuran meets the exacting standards required for dinotefuran synthesis, thereby securing your downstream manufacturing processes against quality fluctuations.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized process can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us partner with you to drive efficiency and innovation in your agrochemical manufacturing operations.
