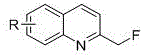
Scalable Synthesis of 2-Fluoromethylquinoline Derivatives for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust methodologies for introducing fluorine atoms into heterocyclic scaffolds, driven by the profound impact of fluorination on metabolic stability and binding affinity. Patent CN116283759A discloses a groundbreaking preparation method for 2-monofluoromethylquinoline and its derivatives, addressing a critical gap in current synthetic capabilities. This innovation leverages potassium bis(trimethylsilyl)amide (KHMDS) as a key mediator to facilitate the direct fluorination of 2-methylquinoline substrates using Selectfluor. Unlike conventional approaches that struggle with selectivity and cost, this technique offers a streamlined pathway to access valuable bioisosteres of methyl and hydroxymethyl groups. The method operates under relatively mild thermal conditions in benzene solutions, ensuring good substrate solubility and wide applicability across diverse chemical spaces. For R&D directors and procurement specialists, this represents a significant advancement in accessing high-purity pharmaceutical intermediates with enhanced process controllability and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of monofluoromethylated heterocycles has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Prior art, such as the work by Pang et al., relied heavily on a two-step sequence involving initial iodination of the C-H bond followed by substitution with silver difluoride (AgF2). This reliance on silver-based reagents introduces prohibitive costs due to the high market price of silver salts, making the process economically unfeasible for industrial applications beyond small-scale laboratory synthesis. Furthermore, the handling of silver compounds often necessitates specialized waste treatment protocols to recover precious metals and manage heavy metal contamination, adding layers of complexity to the manufacturing workflow. The harsh conditions often associated with these traditional halogenation-substitution sequences can also lead to poor functional group tolerance, limiting the scope of compatible substrates and resulting in lower overall yields due to side reactions and decomposition of sensitive molecular architectures.
The Novel Approach
The methodology outlined in CN116283759A fundamentally reshapes the synthetic landscape by replacing expensive transition metal mediators with a more accessible base-promoted system. By utilizing potassium bis(trimethylsilyl)amide in conjunction with Selectfluor, the process achieves direct fluorination without the need for prior halogenation steps, thereby shortening the synthetic route and improving atom economy. This novel approach operates effectively in common organic solvents like benzene, providing excellent solubility for the quinoline substrates and facilitating homogeneous reaction conditions that enhance reproducibility. The elimination of silver reagents not only drastically reduces raw material costs but also simplifies the downstream purification process, as there is no need for extensive heavy metal scavenging or recovery operations. This shift towards base-mediated fluorination represents a paradigm change in cost reduction in pharmaceutical intermediate manufacturing, offering a greener and more efficient alternative that aligns with modern sustainability goals while maintaining high reaction yields and selectivity.
Mechanistic Insights into KHMDS-Mediated Fluorination
The core of this innovative synthesis lies in the unique reactivity profile of potassium bis(trimethylsilyl)amide (KHMDS) as a non-nucleophilic strong base. In the reaction mixture, KHMDS serves to deprotonate the methyl group at the 2-position of the quinoline ring, generating a stabilized carbanion or radical anion species depending on the specific mechanistic pathway involved. This activation step is crucial as it renders the otherwise inert methyl C-H bond susceptible to electrophilic attack by the fluorinating agent, Selectfluor. The use of KHMDS ensures that the deprotonation occurs selectively at the desired position without interfering with other sensitive functional groups present on the quinoline scaffold, such as halogens or alkoxy substituents. The subsequent interaction between the activated quinoline species and Selectfluor facilitates the transfer of a fluorine atom, effectively converting the methyl group into a monofluoromethyl moiety. This mechanism avoids the formation of difluoromethyl or trifluoromethyl byproducts, which are common issues in radical fluorination chemistry, thereby ensuring high specificity for the target monofluoromethyl structure.
Controlling the impurity profile is a paramount concern for any process intended for pharmaceutical production, and this method demonstrates superior control over side reaction pathways. The mild reaction temperature range of 70-90°C, optimally at 80°C, prevents thermal degradation of the quinoline core and minimizes the formation of polymeric byproducts that often arise from aggressive radical conditions. Furthermore, the stoichiometry of the reagents is tightly controlled, with a molar ratio of substrate to Selectfluor and KHMDS maintained around 1:1.1, which prevents excess reagent accumulation that could lead to over-fluorination or decomposition. The workup procedure involving ethyl acetate extraction and silica gel column chromatography using a hexane/ethyl acetate system effectively separates the desired product from any unreacted starting materials or minor side products. This rigorous control over reaction parameters and purification ensures that the final 2-fluoromethylquinoline derivatives meet stringent purity specifications required for downstream drug development, reducing the burden on quality control laboratories and accelerating the timeline for clinical candidate selection.
How to Synthesize 2-Fluoromethylquinoline Efficiently
Implementing this synthesis requires careful attention to inert atmosphere techniques and precise reagent dosing to maximize yield and safety. The process begins with the preparation of the KHMDS solution in benzene, which must be handled within a glove box to prevent moisture ingress that could deactivate the base. Once the base solution is ready, the 2-methylquinoline substrate and Selectfluor are introduced, and the mixture is sealed in a Schlenk tube to maintain anhydrous conditions throughout the heating phase. The reaction is then subjected to thermal energy at 80°C for a duration of 36 hours, allowing sufficient time for the fluorination to reach completion. Following the reaction, the mixture is cooled to room temperature and subjected to a standard aqueous workup involving ethyl acetate extraction and brine washing to remove inorganic salts. The crude product is then dried over anhydrous sodium sulfate and concentrated before undergoing final purification via silica gel column chromatography.
- Prepare a solution of potassium bis(trimethylsilyl)amide in benzene under inert atmosphere conditions within a glove box.
- Add 2-methylquinoline derivatives and Selectfluor to the base solution, maintaining a molar ratio of approximately 1: 1.1.
- Seal the reaction vessel and heat at 80°C for 36 hours, followed by extraction, washing, and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this KHMDS-mediated fluorination route offers substantial strategic benefits that extend beyond simple chemical transformation. The primary advantage lies in the drastic simplification of the supply chain for raw materials, as the process eliminates the dependency on volatile and expensive precious metal catalysts like silver. This shift mitigates the risk of supply disruptions caused by geopolitical instability or mining constraints associated with rare metals, ensuring a more reliable and predictable sourcing strategy for critical intermediates. Additionally, the use of common organic solvents and commercially available fluorinating agents reduces the complexity of inventory management and lowers the overall cost of goods sold. The robustness of the reaction conditions also translates to higher batch consistency, reducing the frequency of failed batches and the associated costs of reprocessing or disposal. This reliability is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of silver-based reagents represents a direct and significant reduction in raw material expenditures, as silver salts constitute a major cost driver in traditional fluorination methods. By substituting these with cost-effective bases like KHMDS and Selectfluor, the overall production cost per kilogram of the intermediate is substantially lowered, improving profit margins for manufacturers. Furthermore, the simplified workup procedure reduces the consumption of specialized scavenging resins and solvents required for metal removal, leading to additional savings in operational expenses. The high yield and selectivity of the reaction minimize waste generation, further contributing to cost efficiency by maximizing the output from each batch of starting materials. These cumulative savings make the process highly competitive for large-scale commercial production of fluorinated pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly strengthened by the use of widely available commodity chemicals rather than specialized precious metal catalysts that may have long lead times or limited suppliers. The stability of the reagents and the simplicity of the reaction setup allow for flexible manufacturing scheduling, enabling producers to respond quickly to fluctuations in market demand without being constrained by complex catalyst procurement cycles. This agility is particularly valuable in the fast-paced pharmaceutical sector where timelines for drug development are critical. Moreover, the reduced regulatory burden associated with heavy metal residues simplifies the compliance landscape, facilitating smoother audits and faster approval processes for new drug applications. This reliability ensures that downstream partners can depend on a steady flow of high-quality intermediates to support their own production pipelines.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor equipment and conditions that are easily transferable from laboratory to pilot and commercial scales. The absence of toxic heavy metals simplifies waste treatment protocols, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The green nature of the process, characterized by fewer side products and efficient atom economy, reduces the environmental footprint of the manufacturing operation. This compliance with eco-friendly standards enhances the brand reputation of the manufacturer and opens up opportunities in markets with strict environmental criteria. The ability to scale up without significant process redesign ensures that supply can grow in tandem with the commercial success of the final drug product, securing long-term partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing frameworks.
Q: Why is the KHMDS/Selectfluor method superior to traditional silver-mediated fluorination?
A: Traditional methods often rely on expensive silver salts like AgF2, which impose severe cost constraints and limit industrial scalability. The novel KHMDS-mediated approach utilizes cost-effective reagents and mild conditions, significantly enhancing economic viability for large-scale manufacturing.
Q: What is the substrate scope for this fluorination reaction?
A: The method demonstrates excellent versatility, accommodating various substituents on the quinoline ring including alkyl, halogen (chloro, bromo, iodo), alkoxy, and aromatic groups, ensuring broad applicability for diverse drug discovery programs.
Q: How does this process impact supply chain reliability for fluorinated intermediates?
A: By eliminating the dependency on scarce precious metal catalysts and utilizing common organic solvents like benzene, the process mitigates supply chain bottlenecks. This ensures consistent production continuity and reduces lead times for critical pharmaceutical building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoromethylquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this KHMDS-mediated synthesis route for producing high-value fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of base-mediated fluorination, including rigorous inert atmosphere controls and advanced purification capabilities. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-fluoromethylquinoline meets the highest international standards. Our commitment to technical excellence allows us to optimize this patented process further, tailoring it to your specific volume and quality needs while maintaining cost efficiency and supply security.
We invite you to collaborate with us to leverage this advanced technology for your next drug development program. By engaging our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this silver-free methodology for your specific portfolio. We encourage you to reach out for specific COA data and route feasibility assessments to validate the performance of our manufactured intermediates against your internal benchmarks. Let us help you secure a sustainable and cost-effective supply chain for your critical fluorinated building blocks, driving your projects forward with confidence and speed.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
