Advanced Enzymatic Transesterification for High-Purity L-Ascorbyl Aryl Acetates
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign, and cost-effective methods for synthesizing complex bioactive intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN102827893A, which discloses a novel ester exchange method for synthesizing aryl acetic (propionic) acid L-ascorbic acid esters. This technology represents a paradigm shift from traditional direct esterification to a sophisticated enzymatic transesterification process, specifically designed to overcome the thermodynamic and kinetic limitations inherent in previous methodologies. By utilizing biocatalysts such as Novozym 435 in a non-aqueous phase, the process facilitates the reaction between aryl acetic acid low-carbon alcohol esters and L-ascorbic acid. The core innovation lies in the continuous removal of the generated low-carbon alcohol under reduced pressure, which effectively drives the chemical equilibrium towards the synthesis direction without compromising the catalytic activity of the enzyme. This approach not only ensures that the reaction proceeds to near completion but also integrates a Simulated Moving Bed Chromatography (SMBC) separation system to achieve unprecedented levels of purity and raw material utilization.
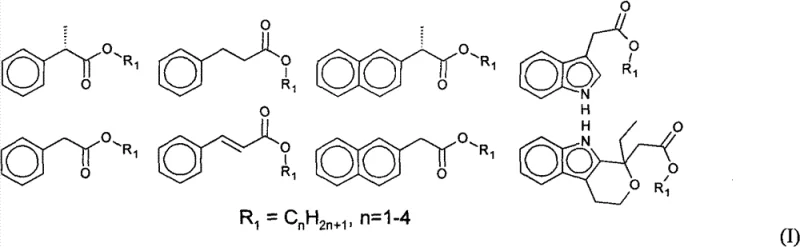
For procurement managers and supply chain directors, the implications of this technology extend far beyond mere chemical curiosity. The ability to synthesize high-purity pharmaceutical intermediates with conversion rates and yields approaching 100% translates directly into substantial cost reduction in pharmaceutical intermediate manufacturing. Traditional methods often suffer from incomplete reactions and difficult purification steps that generate significant waste, particularly high-salt wastewater from washing procedures. In contrast, this patented process eliminates the generation of water as a byproduct, thereby eradicating the need for salt washing and achieving zero discharge of wastewater and waste residue. This environmental compliance is increasingly critical for maintaining a reliable supply chain in regions with stringent environmental regulations. Furthermore, the integration of SMBC allows for the complete separation of reaction products from excess raw materials, solving the critical issue of recycling unreacted substrates. This closed-loop capability ensures that valuable starting materials like L-ascorbic acid and aryl acid esters are not lost but rather recovered and reused, significantly enhancing the overall economic viability of the production process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of aryl acetic (propionic) acid L-ascorbic acid esters has relied on the direct esterification reaction between aryl acids and L-ascorbic acid. While conceptually straightforward, this conventional approach is plagued by several fundamental thermodynamic and operational challenges that hinder its industrial scalability. The primary issue is that direct esterification is a reversible reaction that produces water as a stoichiometric byproduct. To drive the equilibrium towards the desired ester product, water must be continuously removed from the reaction system. However, in the context of biocatalysis, the removal of water is exceptionally problematic because many lipases and other biological catalysts require a certain level of hydration to maintain their active conformation; excessive dehydration can irreversibly deactivate the enzyme. Conversely, retaining water limits the conversion rate, often resulting in a mixture of product and unreacted starting materials that is difficult to separate. Additionally, traditional downstream processing typically involves washing the filtrate with saturated aqueous salt solutions to isolate the product. This step generates large volumes of high-salt wastewater containing residual L-ascorbic acid, representing both a significant environmental burden and a loss of valuable raw materials. These inefficiencies collectively result in lower yields, higher production costs, and a larger environmental footprint, making conventional methods less attractive for modern, sustainable manufacturing.
The Novel Approach
The novel approach described in the patent fundamentally reengineers the synthesis pathway by substituting the aryl acid substrate with an aryl acid low-carbon alcohol ester. This strategic substitution changes the reaction byproduct from water to a low-carbon alcohol, such as methanol or ethanol. Unlike water, these alcohols can be easily and continuously removed from the reaction system via vacuum distillation at moderate temperatures (20-70°C) and pressures (0.0001-0.09 MPa) without adversely affecting the stability or activity of the biocatalyst. This continuous removal of the alcohol byproduct effectively shifts the molecular equilibrium towards the formation of the ester product, driving the reaction to near-total completion. Furthermore, the process incorporates a Simulated Moving Bed Chromatography (SMBC) system for downstream purification. This advanced separation technology allows for the thorough isolation of the target L-ascorbyl ester from any remaining unreacted L-ascorbic acid and aryl acid ester. Crucially, the separated unreacted materials can be recycled back into the reactor, creating a highly efficient, circular production loop. This methodology not only solves the water removal problem inherent in traditional biocatalysis but also eliminates the generation of saline wastewater, realizing a truly green and economically superior manufacturing process.
Mechanistic Insights into Enzymatic Transesterification and SMBC Separation
The core of this technological advancement lies in the precise orchestration of enzymatic catalysis and thermodynamic control. The reaction utilizes lipases, such as the commercially available Novozym 435, which exhibit excellent catalytic activity towards the substrate aryl acetic acid low-carbon alcohol esters. Mechanistically, the lipase facilitates the nucleophilic attack of the hydroxyl group of L-ascorbic acid on the carbonyl carbon of the ester substrate. This transesterification reaction releases a molecule of low-carbon alcohol. The patent highlights a critical discovery: the generated low-carbon alcohol does not inhibit the catalytic activity of the lipase, unlike the potential inhibitory effects of water accumulation or harsh chemical dehydrating agents. By maintaining the reaction system under vacuum tightness, the volatile alcohol byproduct is蒸出 (distilled off) as vapor. According to Le Chatelier's principle, the continuous removal of a product (the alcohol) forces the equilibrium position to shift to the right, favoring the synthesis of the L-ascorbyl ester. This dynamic equilibrium shift is the key to achieving conversion rates that approach 100%, a feat rarely accomplished in reversible enzymatic reactions without complex engineering.
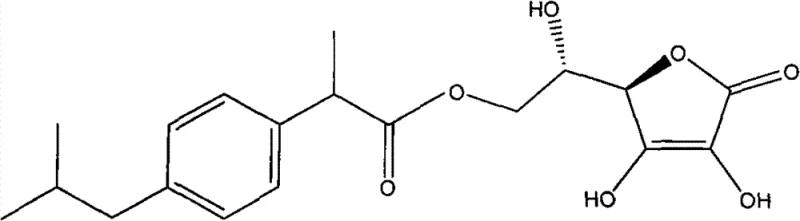
Following the reaction, the control of impurities and the recovery of materials are managed through the Simulated Moving Bed Chromatography (SMBC) system. In traditional batch chromatography, separation efficiency is often limited by peak broadening and dilution. SMBC, however, operates as a continuous counter-current separation process, mimicking the movement of the stationary phase against the flow of the mobile phase. In this specific application, the solid mixture obtained after solvent removal—containing the product and unreacted L-ascorbic acid—is loaded onto the SMBC system using a suitable sorbent material (e.g., silica gel, polymeric amide) and eluent (e.g., mixtures of methanol and dichloromethane). The system effectively separates the L-ascorbyl ester component from the L-ascorbic acid component based on their differential adsorption affinities. This high-resolution separation ensures that the final product achieves a purity of greater than 98%, meeting the stringent requirements for pharmaceutical intermediates. Moreover, the isolated L-ascorbic acid and unreacted aryl acid ester fractions are collected, the solvents are reclaimed, and the materials are fed back into the synthesis reactor. This mechanism of impurity control and material recycling is what enables the process to claim zero waste discharge and maximized atom economy.
How to Synthesize L-Ascorbyl Aryl Acetates Efficiently
The synthesis of these high-value intermediates requires careful attention to reaction parameters and separation techniques to fully realize the benefits of the patented process. The procedure generally involves dissolving L-ascorbic acid and the specific aryl acid ester (such as ibuprofen methyl ester or naproxen methyl ester) in a suitable organic solvent like tert-butanol or tertiary amyl alcohol. The biocatalyst is then added, and the mixture is heated to a temperature range of 30-70°C. Crucially, the system must be sealed and connected to a vacuum pump to maintain a pressure between 0.0001 and 0.09 MPa during the reaction phase to facilitate the removal of the alcohol byproduct. After the reaction period (typically around 24 hours), the enzyme and unreacted solids are filtered off while hot. The filtrate is then concentrated, and the residue is subjected to the SMBC separation process to isolate the pure ester. For a detailed, step-by-step protocol including specific solvent ratios and equipment settings, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining aryl acetic acid low-carbon alcohol ester, L-ascorbic acid, and a lipase catalyst such as Novozym 435 in an organic solvent.
- Conduct the reaction under reduced pressure (0.0001-0.09 MPa) at 20-70°C to continuously remove the generated low-carbon alcohol byproduct, shifting the equilibrium towards ester formation.
- Purify the crude product using a Simulated Moving Bed Chromatography (SMBC) system to separate the target ester from unreacted L-ascorbic acid, allowing for the recycling of raw materials.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and supply chain continuity, the adoption of this enzymatic transesterification technology offers transformative advantages over legacy chemical synthesis routes. The primary value proposition is the drastic simplification of the production workflow, which inherently leads to significant cost optimization. By eliminating the need for water removal agents and the subsequent salt-washing steps, the process removes entire unit operations from the manufacturing line. This reduction in processing steps not only lowers energy consumption and labor costs but also minimizes the risk of product loss during multiple transfer and washing stages. Furthermore, the ability to recycle unreacted raw materials means that the effective consumption of expensive starting materials like L-ascorbic acid and chiral aryl acids is minimized. This high atom economy translates directly into a lower cost of goods sold (COGS), providing a competitive edge in pricing for the final active pharmaceutical ingredients (APIs) derived from these intermediates.
- Cost Reduction in Manufacturing: The elimination of wastewater treatment costs is a major financial benefit. Traditional methods generate high-salt wastewater that requires expensive treatment before discharge to meet environmental standards. This new process achieves zero discharge of wastewater and waste residue, effectively removing the regulatory and financial burden of effluent management. Additionally, the high conversion rate reduces the quantity of raw materials required per kilogram of product, further driving down variable costs.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process ensures consistent batch-to-batch quality, which is critical for pharmaceutical supply chains. The use of commercially available biocatalysts like Novozym 435 ensures that the supply of critical reagents is stable and not subject to the volatility of custom synthetic catalysts. Moreover, the recycling capability acts as a buffer against raw material price fluctuations; by recovering and reusing excess reagents, the manufacturing process becomes less sensitive to short-term spikes in the cost of L-ascorbic acid or aryl acid esters.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind. The use of SMBC is a well-established technology in the sugar and petrochemical industries and is readily scalable for fine chemical production. The environmental profile of the process aligns perfectly with global trends towards green chemistry and sustainable manufacturing. Facilities adopting this technology can easily comply with increasingly strict environmental regulations regarding solvent use and waste discharge, ensuring long-term operational continuity without the risk of shutdowns due to non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing production pipelines.
Q: Why is enzymatic transesterification preferred over direct esterification for L-ascorbyl esters?
A: Direct esterification produces water as a byproduct, which is difficult to remove without deactivating the biocatalyst and often requires salt washing that generates wastewater. Transesterification produces low-carbon alcohol, which is easier to remove under vacuum without harming the enzyme, leading to higher conversion rates and zero wastewater discharge.
Q: How does the SMBC separation system contribute to cost efficiency?
A: The Simulated Moving Bed Chromatography (SMBC) system ensures complete separation of the product from excess reactants. This allows unreacted L-ascorbic acid and aryl acetic acid esters to be recovered and recycled into the next batch, drastically reducing raw material consumption and waste.
Q: What purity levels can be achieved with this patented process?
A: Through the combination of optimized enzymatic conditions and advanced SMBC purification, the process achieves product purity levels exceeding 98%, with raw material conversion and product yields approaching 100%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Ascorbyl Esters Supplier
The technological potential of enzymatic transesterification for synthesizing aryl acetic acid L-ascorbyl esters is immense, offering a pathway to high-purity intermediates with minimal environmental impact. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of L-Ascorbyl Esters meets the highest international standards. We understand the critical nature of these intermediates in the formulation of next-generation anti-inflammatory and analgesic drugs, and we are committed to delivering products that support your drug development timelines.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your budget. Please contact our technical procurement team today to request specific COA data for our available esters and to discuss route feasibility assessments for your target molecules. Let us be your partner in achieving efficient, sustainable, and cost-effective pharmaceutical manufacturing.
