Advanced Oxidation Technology for 3 7-Diketocholanic Acid: Commercial Scale-up and Purity Control
Advanced Oxidation Technology for 3 7-Diketocholanic Acid: Commercial Scale-up and Purity Control
The pharmaceutical industry continuously seeks robust synthetic routes for critical bile acid intermediates, particularly those that ensure high purity and safety profiles for final drug substances. Patent CN111410675A introduces a groundbreaking preparation method for 3 7-diketocholanic acid, a compound of significant interest due to its role as a key impurity reference standard in the synthesis of ursodeoxycholic acid and obeticholic acid. This technology addresses the long-standing challenge of controlling peroxidation byproducts that exhibit hepatotoxicity, thereby enhancing the overall safety and quality of bile acid-based therapeutics. By leveraging a bromate-bromide-acid oxidation system, this method offers a distinct advantage over traditional enzymatic or NBS-mediated pathways, providing a scalable solution for reliable pharmaceutical intermediates supplier networks globally. The strategic implementation of this synthesis route allows manufacturers to achieve superior impurity profiling, which is essential for meeting stringent regulatory requirements in major markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxidized bile acid derivatives has relied heavily on reagents such as N-bromosuccinimide (NBS) or complex enzymatic systems involving steroid dehydrogenases. While effective on a laboratory scale, these conventional methods present substantial hurdles when translated to industrial manufacturing environments. The use of NBS often generates stoichiometric amounts of succinimide byproducts, complicating the purification process and increasing waste disposal costs significantly. Furthermore, enzymatic routes, although selective, require expensive cofactors like NAD and specialized equipment for oxygen introduction, which drastically escalates the operational expenditure. These traditional pathways often suffer from inconsistent reproducibility and difficult post-treatment procedures, leading to variable yields and purity levels that fail to meet the rigorous standards required for high-purity pharmaceutical intermediates. Consequently, supply chains relying on these outdated technologies face risks of batch failures and extended lead times.
The Novel Approach
In contrast, the novel approach detailed in patent CN111410675A utilizes a cost-effective bromate-bromide-acid complex ligand system to selectively oxidize chenodeoxycholic acid. This method eliminates the need for expensive transition metal catalysts or hazardous halogenating agents, streamlining the reaction workflow into a simple, one-pot procedure. The reaction conditions are mild, typically operating at temperatures not exceeding 60°C, which reduces energy consumption and minimizes the risk of thermal degradation of the sensitive steroid backbone. The workup procedure is equally efficient, involving simple water crystallization and washing steps that avoid complex chromatographic separations. This simplicity translates directly into cost reduction in pharmaceutical intermediates manufacturing, as it lowers both raw material costs and processing time. The robustness of this chemical system ensures high process reproducibility, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Bromate-Mediated Selective Oxidation
The core of this technological advancement lies in the precise control of the oxidation state of the steroid nucleus. The reaction initiates with the dissolution of chenodeoxycholic acid in a mixed solvent system, creating a homogeneous phase for the subsequent redox reaction. 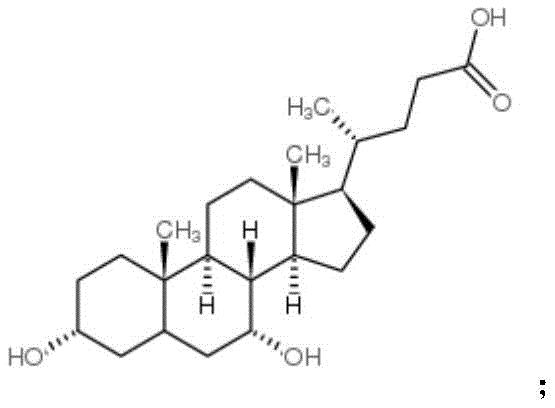 . Upon the addition of the bromide salt and acid, an active brominating species is generated in situ, which facilitates the selective oxidation of the hydroxyl groups at the C3 and C7 positions. The use of bromate as the terminal oxidant ensures that the reaction proceeds cleanly to the diketone stage without over-oxidation or ring cleavage, which are common side reactions in less controlled systems. This mechanistic pathway is critical for maintaining the integrity of the cholestane skeleton while achieving the desired functional group transformation.
. Upon the addition of the bromide salt and acid, an active brominating species is generated in situ, which facilitates the selective oxidation of the hydroxyl groups at the C3 and C7 positions. The use of bromate as the terminal oxidant ensures that the reaction proceeds cleanly to the diketone stage without over-oxidation or ring cleavage, which are common side reactions in less controlled systems. This mechanistic pathway is critical for maintaining the integrity of the cholestane skeleton while achieving the desired functional group transformation.
Controlling the impurity profile is paramount in this synthesis, as the target molecule itself is often an impurity in other bile acid syntheses. 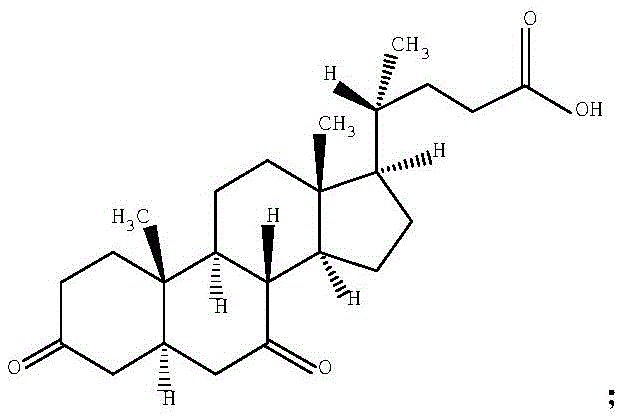 . The reaction equation demonstrates the direct conversion of the dihydroxy substrate to the diketone product.
. The reaction equation demonstrates the direct conversion of the dihydroxy substrate to the diketone product. 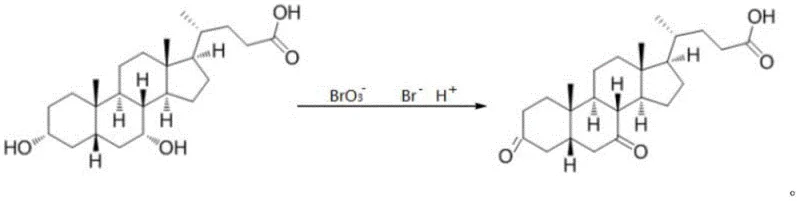 . By carefully monitoring the molar ratios of bromate to substrate, the process minimizes the formation of mono-ketone intermediates or other oxidative byproducts. The termination step using a reducing agent like sodium bisulfite effectively quenches any residual oxidizing species, preventing further degradation during the workup. This level of control ensures that the final product meets the stringent purity specifications of greater than 96%, providing a reliable reference standard for quality control laboratories.
. By carefully monitoring the molar ratios of bromate to substrate, the process minimizes the formation of mono-ketone intermediates or other oxidative byproducts. The termination step using a reducing agent like sodium bisulfite effectively quenches any residual oxidizing species, preventing further degradation during the workup. This level of control ensures that the final product meets the stringent purity specifications of greater than 96%, providing a reliable reference standard for quality control laboratories.
How to Synthesize 3 7-Diketocholanic Acid Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and reaction monitoring to maximize yield and purity. The process begins with dissolving the starting material in a specific volume of organic solvent and water, followed by the sequential addition of reagents under controlled stirring. Maintaining the reaction temperature within the specified range is crucial to prevent side reactions, and the progress should be monitored using HPLC to determine the optimal quenching point. The detailed standardized synthesis steps see the guide below, which outlines the precise molar equivalents and workup procedures required for consistent results. Adhering to these parameters ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal deviation from the laboratory data.
- Dissolve chenodeoxycholic acid in a mixed solution of organic solvent and water, then add bromide salt and acid while stirring to ensure complete dissolution.
- Add bromate to the mixture to initiate the oxidation reaction, maintaining the temperature below 60°C for 1 to 24 hours until completion.
- Terminate the reaction with a reducing agent, add water to crystallize the product, then filter, wash, and dry to obtain high-purity 3 7-diketocholanic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented method offers significant strategic benefits beyond mere technical feasibility. The shift from expensive enzymatic or NBS-based routes to a simple inorganic oxidant system drastically simplifies the sourcing of raw materials. Common reagents like sodium bromate and sulfuric acid are readily available in bulk quantities, reducing the risk of supply disruptions associated with specialized fine chemicals. This availability enhances supply chain reliability, ensuring that production schedules can be maintained without waiting for niche reagent deliveries. Furthermore, the simplified workup process reduces the demand for specialized waste treatment facilities, contributing to substantial cost savings in environmental compliance and disposal.
- Cost Reduction in Manufacturing: The elimination of expensive cofactors and transition metal catalysts directly lowers the bill of materials for each production batch. By avoiding the need for chromatographic purification, the process significantly reduces solvent consumption and labor hours associated with complex separations. This streamlined approach allows for a more competitive pricing structure without compromising on the quality of the final intermediate. The overall economic efficiency makes this route highly attractive for large-volume production where margin optimization is critical.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals rather than proprietary enzymes or sensitive reagents mitigates the risk of supply chain bottlenecks. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, ensuring consistent output across different batches. This stability is essential for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their production cycles with greater confidence and accuracy.
- Scalability and Environmental Compliance: The aqueous workup and crystallization steps are inherently scalable, translating easily from pilot plant to multi-ton commercial production without significant re-engineering. The use of less hazardous reagents simplifies the handling and storage requirements, improving workplace safety and reducing regulatory burdens. Additionally, the reduced solvent load in the waste stream facilitates easier treatment and disposal, aligning with modern green chemistry principles and environmental sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent data and are intended to clarify the operational benefits and quality standards associated with this method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the bromate oxidation system over alternative methods.
Q: Why is 3 7-diketocholanic acid considered a critical impurity in bile acid synthesis?
A: 3 7-diketocholanic acid is a peroxidation byproduct formed during the oxidation of chenodeoxycholic acid. It possesses strong hepatotoxicity and must be strictly controlled to ensure the safety and purity of final API products like ursodeoxycholic acid.
Q: What are the advantages of the bromate-bromide-acid system over traditional oxidants?
A: This system utilizes cheap and common reagents, avoids the use of expensive transition metal catalysts or hazardous NBS, and offers a simpler post-treatment process with high reproducibility suitable for large-scale manufacturing.
Q: What purity levels can be achieved with this patented preparation method?
A: The patented method consistently achieves a product purity of greater than 96% and a yield exceeding 85%, providing a reliable reference substance for quality control in pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3 7-Diketocholanic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of safe and effective bile acid therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify every batch. Our capability to implement complex oxidation chemistries like the one described in CN111410675A demonstrates our dedication to innovation and quality excellence in the pharmaceutical sector.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced process chemistry. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production requirements. We are ready to provide specific COA data and route feasibility assessments to support your project development and commercialization goals. Let us help you secure a reliable source of high-quality intermediates for your next generation of pharmaceutical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
