Advanced Manufacturing of Canagliflozin Peroxide: A Breakthrough in Impurity Control and Process Efficiency
The pharmaceutical industry's relentless pursuit of purity and safety is exemplified in the rigorous control of impurities for SGLT2 inhibitors like Canagliflozin. Patent CN110790755A introduces a transformative methodology for the preparation of Canagliflozin Peroxide, a critical genotoxic impurity that must be strictly monitored to ensure the safety profile of the final drug product. This impurity, formed during the long-term stability of Canagliflozin tablets, poses significant regulatory challenges, with originator companies enforcing limits as low as 220ppm. The structural complexity of this peroxide derivative necessitates a synthesis route that is not only efficient but also capable of producing high-purity reference standards for quality control laboratories globally.
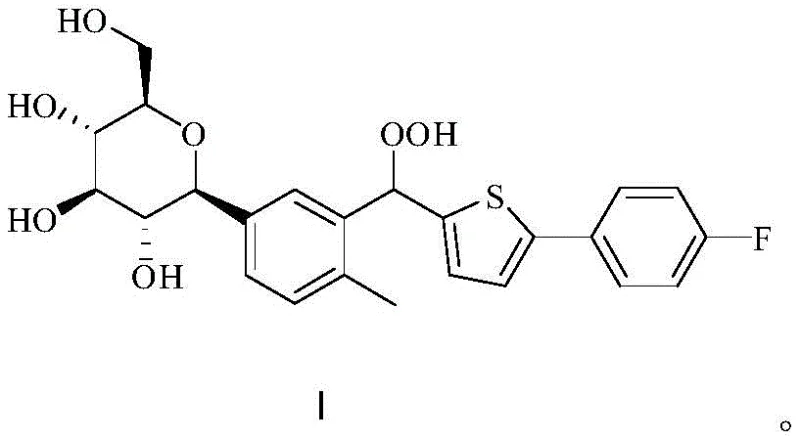
The significance of this patent lies in its ability to address the scarcity of reliable reference materials. By providing a robust synthetic pathway, it enables analytical teams to accurately quantify this trace impurity, thereby safeguarding the therapeutic integrity of diabetes medications. The method described moves away from the precarious balance of traditional oxidation reactions, offering a stable and reproducible protocol that aligns with the stringent demands of modern Good Manufacturing Practice (GMP) environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Canagliflozin Peroxide has been plagued by inefficiencies that render it unsuitable for commercial-scale production or rapid laboratory preparation. One prominent literature method relies on a prolonged reaction time of 6 to 7 days at moderate temperatures, yet it fails to deliver viable yields, often resulting in less than 1% recovery of the target compound. This abysmal performance is primarily attributed to the inherent instability of the peroxide bond, which leads to significant decomposition during the solvent removal phase, particularly when heating is applied. Furthermore, alternative multi-step routes involving the use of expensive and hazardous oxidants like DDQ introduce additional purification burdens, requiring column chromatography after every single step to remove toxic byproducts.
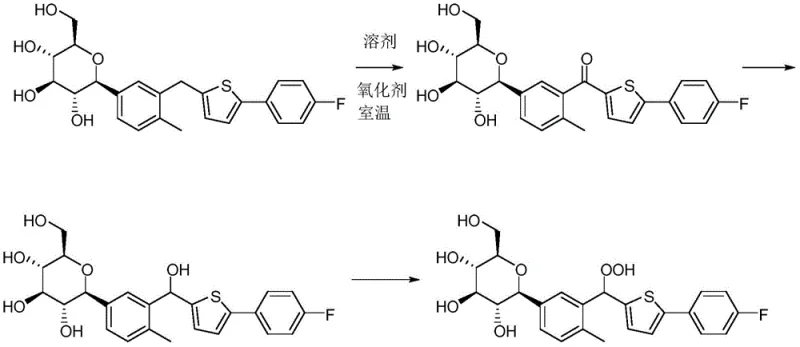
These conventional approaches create a bottleneck for supply chains, as the low conversion rates and complex workup procedures drastically increase the cost of goods sold. The reliance on distillation for solvent recovery is particularly detrimental, as the thermal stress exacerbates the degradation of the sensitive peroxide functionality. Consequently, procurement teams face difficulties in sourcing high-purity standards, and R&D departments struggle with inconsistent batch quality, hindering the timely release of finished drug products.
The Novel Approach
In stark contrast, the methodology disclosed in CN110790755A revolutionizes the production landscape by employing a clever "one-pot" strategy that maximizes atom economy and minimizes waste. Instead of discarding the alcohol byproduct generated during the initial oxidation, the process ingeniously recycles it back into the target peroxide through a secondary acid-catalyzed reaction. This dual-stage approach ensures that the reaction mixture, which initially contains a substantial amount of the alcohol intermediate, is effectively converted into the desired product, thereby boosting the overall yield to impressive levels ranging from 30% to 35%. By eliminating the need for intermediate isolation, the process significantly reduces operational time and solvent consumption.
Mechanistic Insights into Acid-Catalyzed Peroxidation
The core innovation of this synthesis lies in the precise manipulation of reaction conditions to favor peroxide formation over decomposition. The first stage involves heating Canagliflozin with hydrogen peroxide in a mixed solvent system at 50-70°C, which initiates the oxidation at the benzylic position. However, rather than stopping here, the process leverages the presence of the resulting Canagliflozin Alcohol (Formula III) as a reservoir for further conversion. In the second stage, the addition of a strong acid catalyst, such as sulfuric acid or trifluoroacetic acid, activates the alcohol towards nucleophilic attack by excess hydrogen peroxide. This mechanism effectively transforms what would traditionally be a waste byproduct into valuable product, driving the equilibrium towards the formation of Formula I.
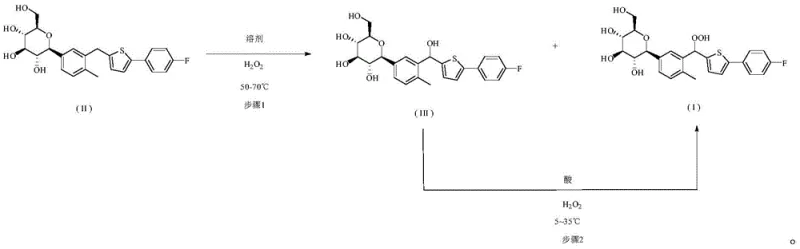
Furthermore, the patent highlights the critical importance of temperature control during the workup phase to preserve the integrity of the peroxide bond. Traditional methods often fail because they attempt to concentrate the product via distillation, which provides the activation energy for decomposition. This new protocol avoids thermal stress entirely by inducing crystallization through the addition of water and cooling the mixture to 0 to -10°C. This gentle precipitation technique ensures that the final isolated solid retains its chemical structure without degradation, resulting in HPLC purities exceeding 98%. Such mechanistic understanding allows for the scalable production of high-purity materials essential for regulatory compliance.
How to Synthesize Canagliflozin Peroxide Efficiently
The synthesis of this critical impurity standard requires careful adherence to the two-stage oxidative protocol to ensure maximum yield and purity. The process begins with the suspension of the starting material in a compatible organic solvent system, followed by the controlled addition of oxidants under specific thermal conditions. Detailed operational parameters, including solvent ratios and acid concentrations, are critical for success. For the complete standardized synthesis procedure and specific reagent quantities, please refer to the step-by-step guide below.
- React Canagliflozin with hydrogen peroxide in an organic solvent-water mixture at 50-70°C to generate a crude mixture containing the target peroxide and the alcohol byproduct.
- Without isolating intermediates, add additional hydrogen peroxide and a strong acid catalyst to the mixture at 5-35°C to convert the alcohol byproduct into the target peroxide.
- Quench the reaction with water and induce crystallization by cooling to 0 to -10°C, avoiding thermal decomposition associated with distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend beyond mere technical feasibility. The elimination of expensive and specialized oxidants like DDQ directly translates to a reduction in raw material costs, making the production of this impurity standard economically viable for large-scale operations. Additionally, the simplification of the workflow by removing multiple column chromatography steps significantly decreases the labor hours and solvent volumes required per batch. This streamlining of the manufacturing process enhances the overall throughput, allowing suppliers to respond more rapidly to market demands for quality control materials.
- Cost Reduction in Manufacturing: The replacement of costly reagents with commodity chemicals like hydrogen peroxide and sulfuric acid creates a substantial decrease in the bill of materials. By avoiding the need for complex purification after every reaction step, the process reduces the consumption of silica gel and organic solvents, which are significant cost drivers in fine chemical synthesis. This economic efficiency allows for more competitive pricing structures without compromising on the quality or purity of the final product.
- Enhanced Supply Chain Reliability: The drastic reduction in reaction time from several days to a manageable timeframe improves the agility of the supply chain. Manufacturers can produce batches on demand with shorter lead times, reducing the risk of stockouts for critical reference standards. The robustness of the method against decomposition ensures consistent batch-to-batch quality, minimizing the need for re-work or rejection of out-of-specification materials, thereby stabilizing the supply flow to pharmaceutical clients.
- Scalability and Environmental Compliance: The avoidance of thermal distillation for solvent removal not only protects the product but also aligns with green chemistry principles by reducing energy consumption. The simplified workup involving aqueous quenching and crystallization is inherently easier to scale from laboratory to pilot plant than multi-step chromatographic separations. This scalability ensures that the supply of Canagliflozin Peroxide can meet the growing global demand for SGLT2 inhibitor testing without encountering bottlenecks associated with complex processing equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Canagliflozin Peroxide. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is vital for quality assurance teams and procurement specialists evaluating potential suppliers.
Q: Why is Canagliflozin Peroxide considered a critical impurity?
A: Canagliflozin Peroxide is identified as a genotoxic impurity in EMA review reports, with strict content limits (e.g., 220ppm) imposed by originator companies to ensure patient safety and drug efficacy.
Q: How does the new patented method improve yield compared to literature methods?
A: Unlike conventional methods that suffer from yields as low as 1% due to product decomposition during solvent removal, this novel approach achieves yields up to 35% by converting unstable byproducts directly into the target molecule.
Q: Does this synthesis require expensive oxidants like DDQ?
A: No, the process eliminates the need for complex and costly oxidants such as 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ), relying instead on hydrogen peroxide and common strong acids, which significantly lowers material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Canagliflozin Peroxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurity standards play in the development and safety assessment of life-saving medications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our rigorous QC labs are equipped to verify the identity and potency of complex molecules like Canagliflozin Peroxide, guaranteeing that every batch delivered supports your regulatory filings and quality control protocols with absolute confidence.
We invite you to collaborate with our technical procurement team to explore how our advanced synthesis capabilities can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient manufacturing processes can reduce your overall expenditure. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project's unique requirements, ensuring a seamless partnership in the development of next-generation pharmaceutical products.
