Advanced Synthesis of Isoquinoline Alkaloid Metabolites for Commercial Scale-up
Advanced Synthesis of Isoquinoline Alkaloid Metabolites for Commercial Scale-up
The pharmaceutical industry is constantly seeking novel derivatives of established bioactive compounds to overcome limitations in bioavailability and therapeutic efficacy. Patent CN1190425C introduces a significant breakthrough in the field of isoquinoline alkaloids, specifically focusing on the synthesis of berberine metabolites and their analogues. This technology addresses the critical issue where oral administration of berberine results in extremely low plasma concentrations, limiting its clinical potential despite its broad pharmacological profile. By chemically modifying the core structure through specific sulfation processes, this patent outlines a method to produce compounds M1 through M6, which demonstrate promising effects on cardiovascular diseases, tumors, and bacterial infections. For R&D directors and procurement specialists, understanding the synthetic pathway of these metabolites is crucial for developing next-generation therapeutic agents that leverage the known safety profile of berberine while enhancing its pharmacokinetic properties.
The core innovation lies in the strategic modification of the isoquinoline backbone, transforming naturally occurring alkaloids into more potent sulfate esters. This approach not only expands the chemical space available for drug discovery but also provides a robust manufacturing route for high-purity intermediates. The patent details the isolation of three new compounds from human urine metabolites and the subsequent chemical synthesis of these structures along with three additional analogues. This dual approach of biological identification followed by chemical replication ensures that the synthesized products are biologically relevant. For supply chain leaders, this represents an opportunity to secure a reliable isoquinoline alkaloids supplier capable of delivering complex, value-added intermediates that are otherwise difficult to source from natural extraction alone due to low abundance in plant materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the utilization of berberine and related isoquinoline alkaloids has been constrained by their poor pharmacokinetic profiles and the challenges associated with extracting sufficient quantities from natural sources. Conventional methods rely heavily on the extraction of berberine from plants like Coptis chinensis, which is subject to agricultural variability, seasonal fluctuations, and geographical limitations. Furthermore, the direct use of berberine hydrochloride is limited by its low solubility and rapid metabolism, leading to sub-therapeutic concentrations in the bloodstream even at effective oral doses. The inability to consistently produce specific metabolites like M1, M2, and M3 through extraction means that researchers have historically been unable to fully explore the therapeutic potential of these endogenous compounds. This reliance on natural variation creates significant supply chain risks and hinders the development of standardized pharmaceutical formulations that require precise impurity profiles and consistent potency.
The Novel Approach
The novel approach detailed in the patent overcomes these barriers by employing a semi-synthetic strategy that converts readily available precursors into high-value metabolites. Instead of relying on the unpredictable yield of natural metabolites, the process utilizes chlorosulfonic acid to introduce sulfate groups at specific positions on the isoquinoline ring system. This chemical transformation is highly controlled, allowing for the production of specific isomers such as M1, M2, and M3 with defined substitution patterns. The method also extends to the synthesis of analogues M4, M5, and M6, providing a broader library of compounds for structure-activity relationship studies. By shifting from extraction to synthesis, manufacturers can achieve cost reduction in pharmaceutical intermediates manufacturing through improved yield consistency and reduced dependency on raw plant material. This synthetic route ensures a stable supply of high-purity isoquinoline alkaloids, enabling continuous clinical development and commercial production without the bottlenecks associated with agricultural sourcing.
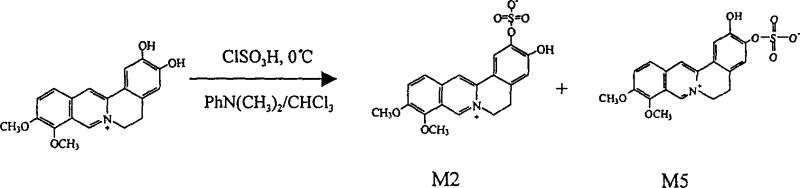
The chemical transformation is exemplified by the reaction of demethyleneberberine with chlorosulfonic acid in a mixed solvent system. This reaction proceeds under mild conditions, typically at 0°C, which minimizes the degradation of the sensitive isoquinoline core. The use of N,N-dimethylaniline and chloroform as solvents facilitates the solubility of the alkaloid salts and ensures efficient contact with the sulfating agent. The result is the formation of sulfate esters that are more water-soluble and potentially more bioactive than the parent compound. This level of control over the chemical structure is essential for meeting the stringent regulatory requirements of modern drug development, where the identity and purity of every impurity must be characterized. The ability to synthesize these metabolites on demand allows for the rapid generation of material for preclinical and clinical studies, accelerating the timeline for bringing new therapies to market.
Mechanistic Insights into Chlorosulfonic Acid Esterification
The mechanism of this synthesis involves a nucleophilic attack by the hydroxyl groups on the isoquinoline ring onto the sulfur atom of the chlorosulfonic acid. This reaction is highly sensitive to temperature and solvent polarity, which is why the patent specifies cooling the reaction mixture to 0°C in an ice-water bath. Maintaining this low temperature is critical to prevent over-sulfation or decomposition of the alkaloid skeleton, which could lead to complex impurity profiles that are difficult to separate. The reaction proceeds through the formation of a reactive sulfonyl intermediate, which is then quenched by the addition of ammonia water. This quenching step neutralizes excess acid and converts the acidic sulfate esters into their ammonium or free acid forms, which can then be precipitated by adjusting the pH. The precision required in this step highlights the need for advanced process control capabilities in a manufacturing setting to ensure batch-to-batch consistency.
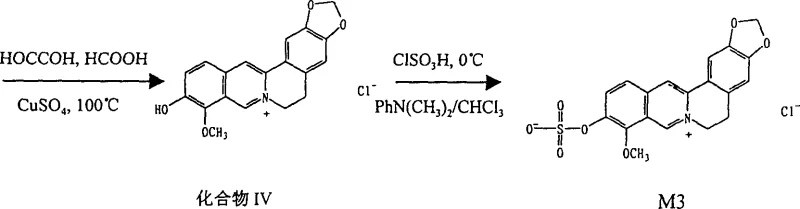
Furthermore, the synthesis of precursors like Compound III and Compound IV involves condensation reactions followed by cyclization, demonstrating the versatility of the synthetic route. For instance, the formation of Compound III from piperethylamine and o-vanillin involves a condensation followed by reduction with potassium borohydride. This step builds the core carbon framework necessary for the subsequent cyclization to the isoquinoline system. The subsequent reaction with glyoxal in the presence of formic acid and copper sulfate closes the ring to form the tetracyclic structure. Understanding these mechanistic details is vital for R&D teams aiming to optimize the process for commercial scale-up of complex pharmaceutical intermediates. Each step offers opportunities for yield improvement and waste reduction, contributing to a more sustainable and cost-effective manufacturing process that aligns with green chemistry principles.
Impurity control is another critical aspect of this mechanism. The use of specific solvents and recrystallization techniques ensures that byproducts such as unreacted starting materials or over-sulfated species are removed. The patent describes purification via macroporous resin column chromatography and Sephadex LH-20, which are effective for separating polar metabolites. In a commercial setting, these chromatographic steps might be replaced or supplemented by crystallization processes to reduce costs. The ability to control the impurity profile is paramount for regulatory approval, as it ensures the safety and efficacy of the final drug product. The detailed characterization data provided in the patent, including NMR spectra, serves as a benchmark for quality control, allowing manufacturers to verify the identity and purity of the synthesized metabolites against established standards.
How to Synthesize Isoquinoline Alkaloids Efficiently
The synthesis of these bioactive isoquinoline alkaloids requires a systematic approach that balances reaction efficiency with product purity. The process begins with the selection of high-quality starting materials, such as berberine sulfate or jatrorrhizine hydrochloride, which are readily available from natural sources. The reaction conditions must be strictly monitored, particularly the temperature during the addition of chlorosulfonic acid, to ensure the formation of the desired sulfate esters without degrading the sensitive alkaloid structure. Following the reaction, the workup procedure involves careful pH adjustment to precipitate the product, followed by recrystallization from methanol and water to achieve high purity. Detailed standardized synthesis steps are essential for reproducibility and are outlined in the technical guide below for process engineers.
- Prepare the reaction vessel with mixed solvent of N,N-dimethylaniline and chloroform, cooling to 0°C.
- Add chlorosulfonic acid in portions while maintaining strict temperature control to ensure regioselective sulfation.
- Terminate the reaction with ammonia water, adjust pH to precipitate crystals, and purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of production from agricultural constraints. By synthesizing berberine metabolites chemically, companies can mitigate the risks associated with crop failures, price volatility of raw herbal materials, and seasonal supply disruptions. This shift ensures a more predictable and stable supply of critical intermediates, which is essential for maintaining continuous manufacturing operations in the pharmaceutical sector. The ability to produce these compounds on demand allows for better inventory management and reduces the need for large safety stocks of expensive natural extracts, thereby freeing up working capital and improving overall financial efficiency.
- Cost Reduction in Manufacturing: The synthetic pathway described eliminates the need for extensive extraction and purification processes typically associated with isolating minor metabolites from plant materials. By starting with abundant precursors like berberine and converting them through efficient chemical steps, the overall cost of goods sold can be significantly optimized. The use of common reagents such as chlorosulfonic acid and standard solvents like chloroform and methanol further contributes to cost efficiency, as these materials are widely available and inexpensive on a global scale. Additionally, the high yields reported in the patent examples suggest that the process is atom-economical, minimizing waste generation and reducing the costs associated with waste disposal and environmental compliance.
- Enhanced Supply Chain Reliability: Relying on a chemical synthesis route enhances supply chain reliability by diversifying the source of raw materials. Instead of depending on a single geographical region for plant extraction, manufacturers can source chemical precursors from multiple suppliers worldwide. This diversification reduces the risk of supply interruptions caused by geopolitical issues or natural disasters. Furthermore, the synthetic process is scalable, meaning that production capacity can be increased rapidly to meet surges in demand without the long lead times required to cultivate additional plant material. This flexibility is crucial for responding to market dynamics and ensuring that clinical trials and commercial launches are not delayed due to material shortages.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in the fine chemical industry, such as stirred tank reactors and crystallization vessels. This compatibility with existing infrastructure reduces the capital expenditure required for technology transfer and scale-up. From an environmental perspective, the process offers advantages in terms of waste management. The use of recyclable solvents and the potential for recovering byproducts aligns with modern sustainability goals. The elimination of heavy metal catalysts in the key sulfation step simplifies the purification process and reduces the environmental burden associated with metal removal and disposal, making it a more attractive option for companies committed to green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these isoquinoline alkaloid derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to provide clarity on the feasibility, safety, and strategic value of integrating this technology into your existing product portfolio. Understanding these details is essential for making informed decisions about process adoption and partnership opportunities.
Q: What are the primary advantages of sulfated berberine metabolites over the parent compound?
A: Sulfated metabolites exhibit improved water solubility and potentially enhanced bioavailability compared to the parent berberine, addressing the limitation of low plasma concentration observed in oral administration.
Q: Is the chlorosulfonic acid esterification process scalable for industrial production?
A: Yes, the process utilizes standard reagents and temperature controls (0°C to 100°C) that are compatible with existing stainless steel reactor infrastructure, facilitating commercial scale-up.
Q: How is product purity ensured during the isolation of these alkaloid derivatives?
A: Purity is achieved through a combination of pH-controlled precipitation and recrystallization from methanol/water mixed solvents, which effectively removes unreacted starting materials and byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline Alkaloids Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes described in patent CN1190425C for the development of advanced cardiovascular and oncology therapeutics. As a leading CDMO partner, we possess the technical expertise and infrastructure required to translate these laboratory-scale methods into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering high-purity isoquinoline alkaloids that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. By leveraging our process chemistry expertise, we can help you achieve significant cost savings and enhance the scalability of your production. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us partner with you to bring these innovative therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
