Advanced Synthesis of 5-Chloro-3-alkylsulfanyl-pyridine-2-carboxylic Acid Amides for Agrochemical Intermediates
Introduction to Next-Generation Agrochemical Intermediates
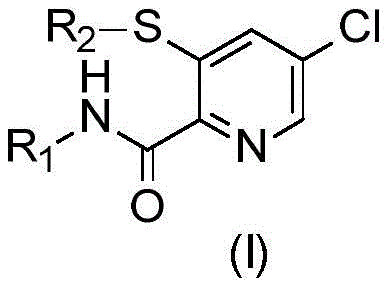
The global agrochemical industry continuously demands more efficient and sustainable pathways for synthesizing complex heterocyclic building blocks. Patent CN115210220A, published on October 18, 2022, introduces a groundbreaking process for the preparation of 5-chloro-3-alkylsulfanyl-pyridine-2-carboxylic acid amides and formates. These compounds, represented by the general formula (I), serve as critical intermediates in the manufacture of biologically active agents, particularly herbicides and insecticides. The invention addresses long-standing challenges in regioselective functionalization of the pyridine ring, offering a robust alternative to legacy synthetic routes.
Traditionally, accessing these specific substitution patterns required convoluted multi-step sequences involving expensive halogenated precursors. The disclosed technology leverages a direct nucleophilic aromatic substitution strategy that achieves exceptional ortho-selectivity. By utilizing readily available 3,5-dichloropyridine-2-carboxamides as starting materials, the process eliminates the need for pre-functionalized bromo- or iodo-derivatives. This strategic shift not only simplifies the synthetic tree but also aligns with green chemistry principles by reducing waste and energy consumption. For R&D teams and procurement specialists alike, this represents a significant opportunity to optimize supply chains for high-value agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
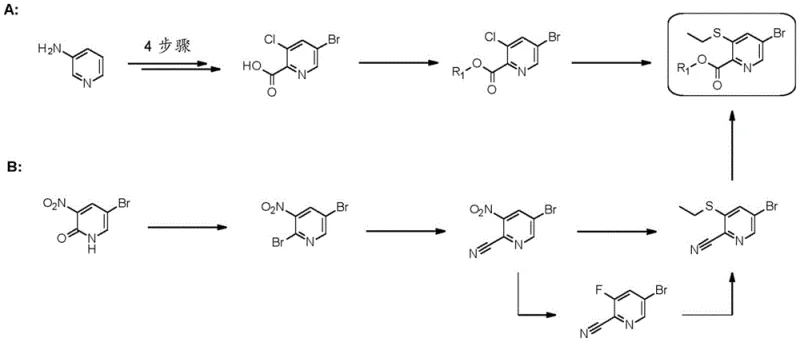
Prior art methods for synthesizing 5-halo-pyridine-2-carboxylic acids with 3-alkylsulfanyl substituents have been fraught with inefficiencies. As illustrated in existing literature such as WO 2016/005263 and related patents, conventional routes often rely on 5-bromo or 5-iodo derivatives. These pathways typically involve four or more reaction steps starting from simple pyridines, necessitating harsh conditions and generating substantial quantities of hazardous waste. For instance, the synthesis of 5-bromo derivatives often requires protection-deprotection strategies and multiple purification stages, which drastically erode overall yield and increase the cost of goods sold (COGS).
Furthermore, the reliance on bromine or iodine atoms at the 5-position introduces significant cost volatility due to the fluctuating market prices of these halogens. The synthetic complexity also poses risks for commercial scale-up, where reproducibility and impurity control become paramount. The formation of regioisomers is a persistent issue in these older methodologies, often requiring chromatographic purification that is impractical on a metric-ton scale. Consequently, there has been a pressing need for a more direct, cost-effective, and scalable solution to access these valuable scaffolds.
The Novel Approach
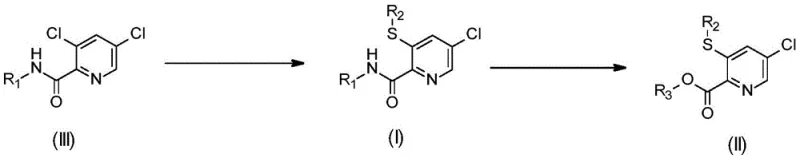
The present invention overturns these limitations by proposing a direct displacement strategy starting from 3,5-dichloropyridine-2-carboxamides. This approach capitalizes on the differential reactivity of the chlorine atoms at the 3- and 5-positions of the pyridine ring. By carefully tuning the reaction environment, specifically the solvent properties, the process achieves highly selective substitution at the 3-position while retaining the chlorine at the 5-position. This single-step transformation converts cheap, commercially abundant dichloro-starting materials directly into the target mono-thiolated products.
This novel pathway effectively bypasses the need for expensive halogen exchange reactions or multi-step functional group manipulations. The versatility of the method allows for the introduction of various alkylsulfanyl groups (R2) and amide substituents (R1), providing a modular platform for generating diverse libraries of agrochemical intermediates. Moreover, the resulting amides can be easily hydrolyzed to the corresponding carboxylic acids (Formula II), further expanding the utility of this platform. This streamlined logic offers a compelling value proposition for manufacturers seeking to reduce lead times and enhance the economic viability of their production processes.
Mechanistic Insights into Solvent-Controlled Regioselectivity
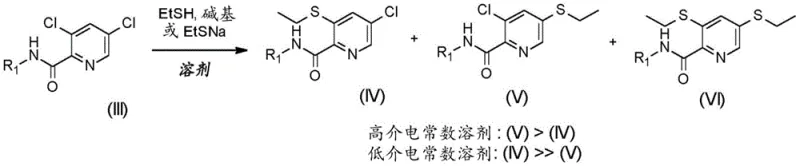
The cornerstone of this invention lies in the profound influence of solvent dielectric constant on reaction selectivity. The patent data reveals a surprising and counter-intuitive phenomenon: the regioselectivity of the thiolation reaction is not merely a function of the nucleophile or temperature, but is dominantly controlled by the polarity of the reaction medium. In solvents with a high relative dielectric constant, such as DMF (dielectric constant ~36.7), the reaction favors attack at the 5-position (para to the nitrogen), leading to the undesired isomer (V). Conversely, in solvents with a low relative dielectric constant (less than 15), such as tetrahydrofuran (THF), 2-methyltetrahydrofuran (MeTHF), or toluene, the reaction exhibits high ortho-selectivity, preferentially displacing the chlorine at the 3-position to yield the desired compound (I).
This mechanistic understanding allows for precise impurity control. By strictly adhering to low dielectric solvents, the formation of the bis-thiolated byproduct (VI) and the wrong regioisomer (V) is minimized. The data indicates that in MeTHF, the selectivity for the desired ortho-isomer can reach upwards of 98%, whereas in polar aprotic solvents like NMP or DMF, the selectivity drops significantly, producing complex mixtures that are difficult to separate. This insight transforms the process from a trial-and-error exercise into a predictable, engineering-controlled operation. It empowers process chemists to design robust manufacturing protocols that consistently deliver high-purity intermediates without the need for extensive downstream purification.
How to Synthesize 5-Chloro-3-ethylsulfanyl-pyridine-2-carboxamide Efficiently
The practical implementation of this technology involves a straightforward procedure accessible to most chemical manufacturing facilities. The process begins with the suspension of 3,5-dichloropyridine-2-carboxamide in a selected low-dielectric solvent such as MeTHF. A base, typically an alkali metal carbonate or hydroxide, is employed to generate the active thiolate species in situ or is added as a pre-formed salt like sodium ethanethiolate. The reaction proceeds smoothly at ambient temperatures, although mild heating can be applied to drive conversion to completion if necessary. The simplicity of the workup, involving aqueous extraction and crystallization, underscores the industrial feasibility of this method.
- React 3,5-dichloropyridine-2-carboxamide with a thiol compound (e.g., sodium ethanethiolate) in a suitable solvent with a dielectric constant of less than 15.
- Maintain reaction temperature between 0°C and 100°C, preferably ambient temperature, to ensure high ortho-selectivity.
- Optionally hydrolyze the resulting amide under basic or acidic conditions to yield the corresponding 5-chloro-3-alkylsulfanyl-pyridine-2-carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical elegance. The shift from multi-step bromo-derivative routes to a direct chloro-displacement methodology fundamentally alters the cost structure of producing these key agrochemical intermediates. By eliminating several synthetic steps, the process reduces the consumption of raw materials, solvents, and energy, leading to a leaner and more cost-efficient manufacturing footprint. This efficiency translates directly into improved margins and greater competitiveness in the global marketplace.
- Cost Reduction in Manufacturing: The primary driver of cost savings is the drastic reduction in step count. Traditional routes often involve four or more steps with cumulative yield losses, whereas this new method achieves the target in a single transformation. Furthermore, the starting material, 3,5-dichloropyridine-2-carboxamide, is significantly cheaper and more readily available than the specialized bromo- or iodo-precursors required by legacy methods. The elimination of expensive halogens like iodine and the avoidance of chromatographic purification steps further contribute to substantial cost reductions in agrochemical manufacturing.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like 3,5-dichloropyridine derivatives mitigates supply risk. Unlike specialized halogenated intermediates which may have limited suppliers and long lead times, the starting materials for this process are produced on a large scale by multiple vendors globally. This diversification of the supply base ensures continuity of supply even during market disruptions. Additionally, the robustness of the reaction conditions means that production schedules are less likely to be impacted by sensitive operational parameters, enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing common industrial solvents like MeTHF and THF which are well-understood in large-scale operations. The high selectivity achieved minimizes the generation of hazardous waste and byproducts, simplifying waste treatment and disposal. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The ability to produce high-purity intermediates with minimal waste makes this technology an attractive option for companies aiming to reduce their environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific product pipelines. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is solvent selection critical in this thiolation process?
A: The patent demonstrates that solvent dielectric constant dictates regioselectivity. Low dielectric solvents (e.g., MeTHF, Toluene) favor the desired ortho-substitution at the 3-position, whereas high dielectric solvents (e.g., DMF) lead to undesired para-substitution at the 5-position.
Q: What are the advantages of this method over traditional bromo-derivative routes?
A: Traditional routes often require multi-step synthesis starting from expensive or less available bromo-precursors. This novel method utilizes readily available 3,5-dichloropyridine-2-carboxamides, significantly reducing step count, waste generation, and overall production costs.
Q: Can this process be scaled for commercial manufacturing?
A: Yes, the process operates under mild conditions (ambient to moderate temperatures) and uses common industrial solvents like MeTHF or THF. The workup involves standard extraction and crystallization techniques, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-3-alkylsulfanyl-pyridine-2-carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis methods described in CN115210220A for the agrochemical sector. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly translate these laboratory-scale innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this more efficient route is seamless and risk-free. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this advanced chemistry for your next-generation agrochemical products. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior supply chain efficiency and product quality.
