Advanced Nickel-Catalyzed Synthesis of O-Nitrobenzonitrile Intermediates for Commercial Herbicide Production
Advanced Nickel-Catalyzed Synthesis of O-Nitrobenzonitrile Intermediates for Commercial Herbicide Production
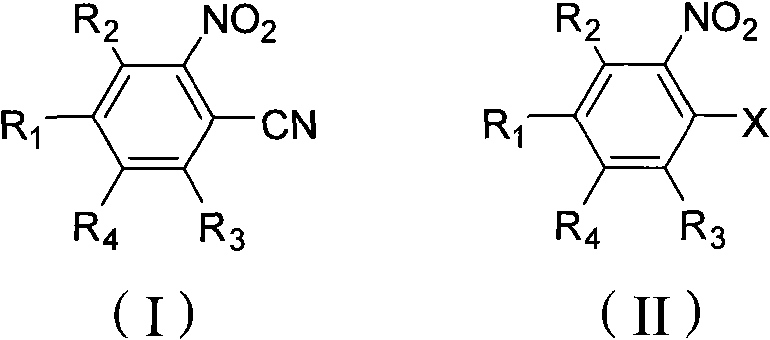
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key agrochemical intermediates, particularly within the o-nitrobenzonitrile series. A pivotal advancement in this domain is detailed in patent CN101585783B, which introduces a robust preparation method utilizing a novel nickel bromide catalyst system. This technology addresses long-standing challenges in the production of compounds like 2-nitro-4-trifluoromethylbenzonitrile, a crucial building block for next-generation HPPD inhibitor herbicides. By shifting away from traditional copper-centric methodologies, this process offers a pathway to significantly enhanced reaction kinetics and product purity. For R&D directors and procurement strategists, understanding this shift is vital for securing a competitive edge in the agrochemical supply chain. The method not only improves yield metrics but also simplifies the downstream processing required to meet stringent pharmaceutical and agricultural grade specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-nitrobenzonitrile derivatives has relied heavily on Rosenmund-von Braun type reactions using cuprous cyanide. As referenced in prior art such as US4886936 and US6635780, these conventional approaches present distinct economic and technical bottlenecks. One established route utilizes 3-nitro-4-bromobenzotrifluoride as a starting material; while effective, this halogenated precursor is prohibitively expensive and difficult to source in bulk quantities, severely impacting cost structures. Alternative methods employing cheaper chloro-substituted precursors often require complex additive systems involving lithium bromide or cuprous bromide to activate the less reactive carbon-chlorine bond. Even with these activators, traditional copper-catalyzed processes frequently struggle to achieve simultaneous high conversion and high selectivity, often resulting in significant formation of by-products and requiring extensive purification steps that erode overall process efficiency.
The Novel Approach
The methodology disclosed in the subject patent represents a paradigm shift by introducing nickelous bromide as the primary catalyst for the cyanation of chloro-substituted nitrobenzenes. This innovative catalytic system enables the direct use of cost-effective 3-nitro-4-chlorobenzotrifluoride, bypassing the need for expensive brominated starting materials entirely. The reaction operates under relatively mild thermal conditions, typically between 140°C and 180°C, yet achieves exceptional performance metrics with conversion rates and selectivity consistently exceeding 95%. Furthermore, the nickel catalyst forms a distinct solid-liquid reaction system with the cyanide source and solvent, which facilitates straightforward separation of the catalyst post-reaction. This operational simplicity translates directly into reduced processing time and lower energy consumption, making the novel approach vastly superior for industrial scale-up compared to legacy copper-based protocols.
Mechanistic Insights into Nickel-Bromide Catalyzed Cyanation
The efficacy of the nickel bromide catalyst lies in its unique ability to facilitate nucleophilic aromatic substitution on the electron-deficient nitrobenzene ring. Unlike copper catalysts which often require harsh conditions or specific ligand environments to activate the aryl-chloride bond, the nickel species interacts dynamically with the cyanide ion to form a highly reactive intermediate complex. This complex effectively lowers the activation energy required for the displacement of the chlorine atom, allowing the reaction to proceed rapidly even with the less reactive chloro-substrate. The presence of the nitro group at the ortho position further activates the ring towards nucleophilic attack, but the nickel catalyst ensures that this reactivity is channeled specifically towards the desired cyanation rather than non-specific decomposition or polymerization. This precise control over the reaction trajectory is what enables the observed high selectivity, minimizing the generation of tars and other difficult-to-remove impurities that typically plague high-temperature cyanation reactions.
From an impurity control perspective, the mechanistic pathway offered by the nickel system significantly reduces the formation of homocoupling by-products and hydrolysis derivatives. In traditional copper-mediated processes, the presence of moisture or oxygen can lead to the formation of carboxylic acids or biaryl impurities, complicating the purification landscape. The nickel bromide system, operating within polar aprotic solvents like N-Methyl pyrrolidone or N,N-Dimethylacetamide, maintains a stable catalytic cycle that is less susceptible to these deactivation pathways. Moreover, the catalyst's ability to be separated as a solid residue post-reaction means that metal contamination in the final organic product is inherently minimized. This is a critical quality attribute for agrochemical intermediates, where residual heavy metal levels are strictly regulated. The result is a cleaner crude product that requires less aggressive downstream purification, preserving yield and reducing solvent waste.
How to Synthesize 2-Nitro-4-trifluoromethylbenzonitrile Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing pilot and production facilities. The process begins with the careful charging of reactants under an inert atmosphere to prevent oxidative degradation of the catalyst or cyanide source. Operators introduce the polar solvent, the chloro-nitro substrate, the nickelous bromide catalyst, and the cyanide source, typically cuprous cyanide or sodium cyanide, into the reactor. The mixture is then heated to the optimized temperature window, where the exothermic nature of the reaction is carefully managed to maintain steady conversion. Following the reaction period, the workup procedure leverages the differential solubility of the product and the catalyst, allowing for efficient isolation through extraction and crystallization. Detailed standardized synthetic steps for this high-efficiency protocol are outlined in the guide below.
- Charge a reaction vessel with polar aprotic solvent such as N-Methyl pyrrolidone, the substrate 3-nitro-4-chlorobenzotrifluoride, nickelous bromide catalyst, and cuprous cyanide under nitrogen protection.
- Heat the reaction mixture to a temperature range of 140°C to 180°C and maintain stirring for 3 to 6 hours to ensure complete conversion.
- Cool the mixture, extract with ether and water, dry the organic phase, and purify via decolorization and precipitation to isolate the high-purity nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this nickel-catalyzed technology offers tangible strategic benefits beyond mere technical performance. The primary advantage stems from the drastic reduction in raw material costs achieved by substituting expensive brominated precursors with widely available chlorinated analogues. This switch eliminates a major cost driver in the bill of materials, allowing for more competitive pricing structures in the final herbicide active ingredient. Additionally, the high selectivity of the reaction means that less raw material is wasted on by-product formation, effectively increasing the atom economy of the process. This efficiency gain reduces the volume of waste streams requiring treatment, aligning with increasingly stringent environmental compliance standards and lowering the total cost of ownership for the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of costly brominated starting materials and the use of a highly efficient catalyst system significantly lowers the direct material costs associated with producing o-nitrobenzonitrile intermediates. By achieving conversion rates above 95%, the process minimizes the loss of valuable substrates, ensuring that nearly every kilogram of input material contributes to the final product yield. Furthermore, the simplified workup procedure reduces the consumption of solvents and energy required for purification, leading to substantial operational expenditure savings. These cumulative efficiencies allow manufacturers to offer high-purity intermediates at a more attractive price point without compromising on quality margins.
- Enhanced Supply Chain Reliability: Relying on chlorinated starting materials rather than specialized brominated derivatives mitigates supply risk, as chloro-nitro compounds are commodity chemicals with robust global availability. This stability ensures consistent production schedules and reduces the likelihood of delays caused by raw material shortages. The robustness of the nickel catalyst system also contributes to reliability, as it tolerates minor variations in reaction conditions better than sensitive copper-ligand systems. Consequently, production batches are more consistent, reducing the incidence of out-of-specification results that can disrupt downstream formulation timelines and inventory planning.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to perform effectively across various batch sizes without loss of efficiency. The ability to separate the solid catalyst easily simplifies the scale-up engineering, removing the need for complex filtration or distillation units often required to remove soluble metal catalysts. From an environmental standpoint, the high selectivity reduces the generation of hazardous organic waste, and the potential for catalyst recycling further diminishes the environmental footprint. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals, making the technology a future-proof choice for long-term manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed cyanation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear picture of the method's capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the consensus on performance metrics and operational parameters observed during the development of this process.
Q: Why is the Nickel Bromide catalyst superior to traditional Copper systems for this synthesis?
A: The Nickel Bromide catalyst system described in patent CN101585783B achieves significantly higher reaction conversion rates and selectivity, often exceeding 95%, compared to traditional copper-based methods which may suffer from lower efficiency or require expensive brominated starting materials.
Q: What are the primary applications of the o-nitrobenzonitrile series produced by this method?
A: These compounds serve as critical intermediates in the manufacture of benzoylisoxazole class herbicides, such as Isoxaflutole and Pyrasulfotole, which function as HPPD inhibitors in modern agriculture.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability, featuring simplified technological operations, short reaction times, and a catalyst system that allows for easy separation and potential recycling, making it ideal for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-4-trifluoromethylbenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of essential agrochemical intermediates. Our technical team has extensively analyzed the nickel-catalyzed methodology described in CN101585783B and validated its potential for large-scale application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of o-nitrobenzonitrile intermediate meets the exacting standards required for herbicide synthesis. We are committed to delivering consistency and quality that empowers our partners to innovate without supply chain constraints.
We invite you to collaborate with us to leverage this advanced technology for your specific production needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing setup, highlighting exactly where this new catalytic system can drive value. Please contact our technical procurement team today to request specific COA data for our o-nitrobenzonitrile series and to discuss detailed route feasibility assessments. Together, we can optimize your supply chain and secure a competitive advantage in the global agrochemical market.
