Advanced Synthesis of p-Bromophenylboronic Acid via In-Situ Quenching for Scalable Pharma Production
The pharmaceutical and fine chemical industries rely heavily on robust supply chains for critical building blocks, and p-bromophenylboronic acid stands out as a pivotal intermediate for Suzuki-Miyaura cross-coupling reactions. Patent CN102731542A introduces a transformative preparation method that addresses long-standing inefficiencies in synthesizing this valuable compound. By shifting from sequential addition protocols to a novel "in-situ quenching" strategy, this technology enables manufacturers to achieve superior purity and yield while operating at significantly more manageable temperatures. For R&D directors and procurement specialists, understanding this shift is crucial, as it represents a move away from fragile, low-yield processes toward a rugged, industrially viable pathway that ensures consistent quality for complex API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
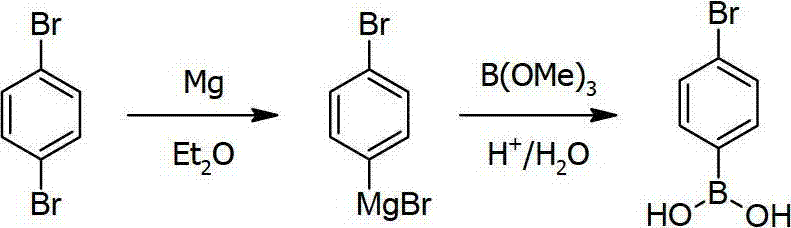
Historically, the synthesis of p-bromophenylboronic acid has been plagued by two primary methodologies, both of which suffer from inherent structural flaws that limit their commercial viability. The traditional Grignard reagent method involves reacting magnesium with paradibromobenzene, followed by the addition of boric acid esters. However, controlling the stoichiometry to prevent double metallation is notoriously difficult; even with precise magnesium control, thermodynamic equilibrium favors the formation of bis-Grignard species, leading to substantial by-product generation and depressed yields. Similarly, the conventional n-Butyl Lithium method requires deep cryogenic conditions at -78°C to mitigate side reactions, yet it still struggles with bimetallic impurities and instability upon warming. These legacy processes impose severe burdens on manufacturing infrastructure, requiring expensive cooling systems and yielding inconsistent product quality that complicates downstream purification.
The Novel Approach
In stark contrast, the patented "in-situ quenching" method fundamentally reengineers the reaction sequence to leverage kinetic advantages over thermodynamic limitations. Instead of generating the organometallic intermediate in isolation, this approach pre-mixes paradibromobenzene with the boric acid ester in the solvent system before introducing the n-Butyl Lithium. This ensures that as soon as the mono-lithiated species forms, it is immediately trapped by the adjacent borate ester, effectively suppressing the formation of unwanted di-lithiated by-products. Furthermore, this method allows the reaction to proceed at a much warmer temperature range of -40°C to -30°C, eliminating the need for extreme cryogenic cooling. This operational shift not only simplifies the engineering requirements for large-scale reactors but also dramatically enhances the overall yield and purity profile of the final product.
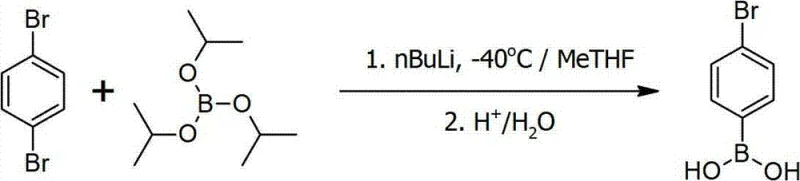
Mechanistic Insights into Lithium-Halogen Exchange and Trapping
The core innovation of this process lies in the precise manipulation of reaction kinetics during the lithium-halogen exchange. In standard protocols, the organolithium intermediate is highly reactive and prone to attacking unreacted starting material or undergoing further metallation if not instantly quenched. By introducing the borate ester into the reaction matrix prior to the addition of the base, the system creates a high local concentration of the trapping agent. As the n-Butyl Lithium exchanges with the bromine atom on the benzene ring, the resulting aryl-lithium species encounters the borate ester immediately. This rapid interception prevents the aryl-lithium from reacting with a second equivalent of n-Butyl Lithium or participating in Wurtz-type coupling side reactions. The result is a clean conversion to the boronate ester derivative, which is subsequently hydrolyzed to the free boronic acid with minimal impurity burden.
From an impurity control perspective, this mechanism offers a distinct advantage by minimizing the formation of phenylboronic acid and biphenyl derivatives that typically contaminate batches produced via older methods. The absence of these structurally similar by-products simplifies the crystallization process, allowing for the attainment of HPLC purities exceeding 99% without the need for extensive chromatographic purification. For quality assurance teams, this means a more predictable impurity profile and reduced risk of carryover contaminants in the final Active Pharmaceutical Ingredient. The stability of the intermediate at -30°C also reduces the risk of thermal decomposition events that can occur during the warming phases of deeper cryogenic processes, ensuring batch-to-batch consistency.
How to Synthesize p-Bromophenylboronic Acid Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature control to maximize the kinetic benefits described above. The process begins with the dissolution of paradibromobenzene and a trialkyl borate, such as triisopropyl borate, in a suitable ether solvent like 2-methyltetrahydrofuran, often blended with hexane to optimize solubility and safety. The mixture is cooled to the target range of -40°C before the controlled addition of n-Butyl Lithium. Following the reaction completion, a carefully managed hydrolysis step involving water and acid adjustment precipitates the product, which is then isolated via crystallization. The detailed standardized synthetic steps see the guide below.
- Mix paradibromobenzene and triisopropyl borate in an organic solvent such as 2-methyltetrahydrofuran under nitrogen protection.
- Cool the mixture to -40°C and slowly add n-Butyl Lithium hexane solution while maintaining temperature between -40°C and -30°C.
- Warm the reaction to 0°C, hydrolyze with water and acid, then crystallize the product from water to obtain high-purity p-bromophenylboronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers tangible strategic benefits that extend beyond simple chemistry. By eliminating the requirement for extreme cryogenic temperatures, the process significantly reduces energy consumption and reliance on specialized low-temperature equipment, directly translating to lower manufacturing overheads. The improved yield profile means that less raw material is wasted per kilogram of finished product, enhancing the overall material efficiency of the supply chain. Additionally, the simplified workup procedure reduces the time required for batch turnover, allowing facilities to increase throughput without expanding physical footprint. These factors combine to create a more resilient and cost-effective supply source for this critical intermediate.
- Cost Reduction in Manufacturing: The elimination of deep cryogenic cooling (-78°C) in favor of moderate low temperatures (-40°C) drastically reduces utility costs associated with refrigeration. Furthermore, the higher yield achieved through in-situ trapping minimizes the loss of expensive starting materials like paradibromobenzene and n-Butyl Lithium. The simplified purification process also reduces solvent usage and waste disposal costs, contributing to a leaner cost structure for the final API intermediate.
- Enhanced Supply Chain Reliability: Operating at less extreme temperatures reduces the risk of batch failures due to equipment malfunction or temperature excursions, ensuring more consistent delivery schedules. The use of common solvents like 2-methyltetrahydrofuran and hexane ensures that raw material sourcing remains stable and unaffected by niche supply constraints. This reliability is critical for maintaining continuous production lines for downstream pharmaceutical clients who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The process is inherently safer and easier to scale from pilot plant to commercial tonnage because it avoids the thermal hazards associated with large volumes of pyrophoric reagents at ultra-low temperatures. The reduction in by-product formation leads to a cleaner waste stream, facilitating easier compliance with environmental regulations regarding solvent discharge and hazardous waste treatment. This makes the technology suitable for long-term sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of p-bromophenylboronic acid using this advanced methodology. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their own supply chains. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industrial application.
Q: How does the in-situ quenching method improve yield compared to traditional Grignard methods?
A: Traditional Grignard methods often suffer from double metallation due to thermodynamic equilibrium, leading to significant by-product formation. The novel in-situ quenching method utilizes kinetic control where the borate ester is present before lithiation, immediately trapping the mono-lithiated intermediate and preventing over-reaction, thereby significantly boosting yield.
Q: What are the energy efficiency benefits of the new reaction temperature range?
A: Conventional n-Butyl Lithium methods typically require deep cryogenic conditions around -78°C, which are energy-intensive and difficult to maintain on a large scale. This patented process operates effectively between -40°C and -30°C, drastically reducing refrigeration load and making industrial scale-up much more feasible and cost-effective.
Q: Which solvents are preferred for this synthesis to ensure optimal solubility and safety?
A: The patent highlights ether solvents like THF and 2-methyltetrahydrofuran (MeTHF), particularly when mixed with hexane. A mixture containing 65% to 85% MeTHF is preferred as it offers a balance of solvating power for the organolithium species and improved safety profiles compared to pure ethers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Bromophenylboronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of p-bromophenylboronic acid meets the exacting standards required for GMP manufacturing. Our commitment to technical excellence ensures that you receive a product that facilitates smooth downstream processing and consistent API quality.
We invite you to contact our technical procurement team to discuss how our advanced synthesis capabilities can support your specific project needs. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our optimized process can enhance your supply chain efficiency and reduce overall project timelines.
