Advanced Manufacturing of Ethyl Chromane-4-Formate: A Technical Breakthrough for Pharmaceutical Intermediates
Advanced Manufacturing of Ethyl Chromane-4-Formate: A Technical Breakthrough for Pharmaceutical Intermediates
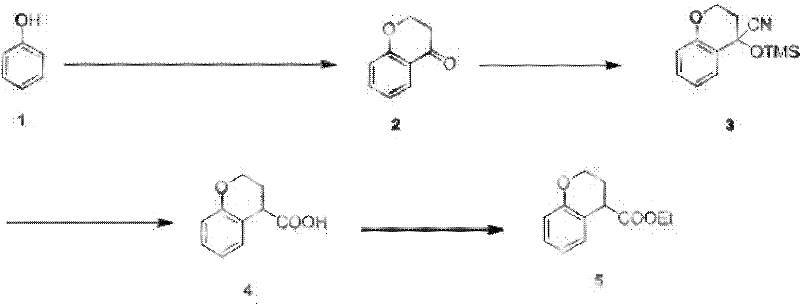
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective pathways for synthesizing complex heterocyclic intermediates. A pivotal development in this domain is detailed in patent CN102603693A, which outlines a superior preparation process for ethyl chromane-4-formate, a critical building block in medicinal chemistry. This patent addresses long-standing inefficiencies in traditional synthetic routes by introducing a modified Friedel-Crafts acylation strategy that replaces prohibitively expensive catalysts with commodity chemicals. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a tangible opportunity to optimize supply chains and reduce the cost of goods sold (COGS) without compromising on purity or structural integrity. The methodology described offers a streamlined approach that enhances both economic viability and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chroman derivatives has relied heavily on the use of trifluoromethanesulfonic acid (TfOH) as a catalyst for the initial Friedel-Crafts acylation step. While effective in driving the reaction, TfOH is an extremely costly reagent that significantly inflates the raw material budget for large-scale manufacturing. Furthermore, the handling of such strong acids requires specialized equipment and rigorous safety protocols, adding layers of complexity and overhead to the production process. Traditional methods often suffer from lower overall yields, typically hovering around 30%, which necessitates larger batch sizes to meet demand, thereby increasing waste generation and energy consumption. These factors combine to create a bottleneck for reliable pharmaceutical intermediate supplier operations, where margin compression is a constant threat.
The Novel Approach
The innovative route presented in the patent data fundamentally alters the economic landscape of this synthesis by substituting trifluoromethanesulfonic acid with anhydrous aluminum chloride (AlCl3). This Lewis acid is not only vastly more affordable but also widely available, ensuring supply chain continuity even during market fluctuations. The process initiates with the reaction of phenol and 3-chloropropionyl chloride, followed by cyclization using sodium hydroxide to yield the key chroman-4-one intermediate. Subsequent steps involve a zinc iodide-catalyzed addition of trimethylsilyl cyanide, followed by hydrolysis and final esterification. This strategic replacement results in a dramatic improvement in overall yield, reaching up to 47%, while simultaneously simplifying the purification workflow. By leveraging common reagents and standard reaction conditions, this approach exemplifies cost reduction in pharma manufacturing through intelligent process design rather than mere scale.
Mechanistic Insights into AlCl3-Catalyzed Cyclization and Functionalization
The core of this technological advancement lies in the mechanistic efficiency of the aluminum chloride-catalyzed Friedel-Crafts acylation. In this step, anhydrous AlCl3 acts as a potent Lewis acid, activating the carbonyl group of 3-chloropropionyl chloride to facilitate electrophilic aromatic substitution on the phenol ring. The reaction is meticulously controlled at temperatures between 35-45°C, a range that balances reaction kinetics with the suppression of side reactions such as polyacylation or resin formation. Following the acylation, the intramolecular cyclization is triggered by treatment with 10% NaOH, closing the pyran ring to form the chroman-4-one scaffold. This two-stage sequence within the first step ensures high conversion rates, with the patent reporting isolated yields of 80% for the crude ketone after recrystallization, demonstrating exceptional selectivity.
Subsequent functionalization relies on precise control of nucleophilic additions and hydrolytic cleavage. The addition of trimethylsilyl cyanide (TMSCN) in the presence of zinc iodide generates a cyanohydrin silyl ether intermediate with high stereochemical fidelity. The hydrolysis step is particularly noteworthy, utilizing stannous chloride dihydrate in concentrated hydrochloric acid and acetic acid at 100°C. This specific reagent combination effectively converts the nitrile group to a carboxylic acid while preserving the sensitive chroman ring structure, avoiding the harsh conditions often required for nitrile hydrolysis that might lead to ring opening or degradation. The final esterification with ethanol and sulfuric acid proceeds under reflux to yield the target ethyl chromane-4-formate with HPLC purity exceeding 98%, validating the robustness of the impurity control mechanisms embedded in this route.
How to Synthesize Ethyl Chromane-4-Formate Efficiently
Implementing this synthesis requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the reaction vessel under a nitrogen atmosphere to prevent moisture interference with the Lewis acid catalyst. Solvents such as nitrobenzene or dichloromethane are employed to ensure solubility and heat transfer during the exothermic acylation phase. Careful monitoring via HPLC is essential to determine the endpoint of each stage, particularly the hydrolysis which requires extended heating times of up to 18 hours. The workup procedures involve standard liquid-liquid extractions using solvents like MTBE and ethyl acetate, followed by pH adjustments to isolate the acidic intermediates. For a comprehensive guide on the exact molar ratios, addition rates, and workup specifics, refer to the standardized protocol below.
- Perform Friedel-Crafts acylation and cyclization using phenol and 3-chloropropionyl chloride with anhydrous aluminum chloride at 40-45°C.
- Conduct an addition reaction with trimethylsilyl cyanide catalyzed by zinc iodide to form the cyanohydrin intermediate.
- Hydrolyze the nitrile group using stannous chloride dihydrate and concentrated hydrochloric acid to obtain the carboxylic acid.
- Complete the synthesis via esterification with ethanol and concentrated sulfuric acid under reflux conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers immediate and tangible benefits that extend beyond simple raw material pricing. The elimination of trifluoromethanesulfonic acid removes a significant cost driver and a potential supply risk, as this reagent can be subject to volatile pricing and logistical constraints. By shifting to anhydrous aluminum chloride, a commodity chemical produced in massive volumes globally, manufacturers can secure long-term supply contracts with greater stability. This change also simplifies waste management protocols, as the effluent profile becomes less aggressive and easier to treat compared to processes involving fluorinated superacids. Consequently, the total cost of ownership for producing high-purity pharmaceutical intermediates is substantially lowered, enhancing the competitiveness of the final API in the global market.
- Cost Reduction in Manufacturing: The substitution of expensive catalysts with inexpensive alternatives directly impacts the bottom line by reducing the bill of materials. Furthermore, the improved yield from 30% to nearly 50% means that less raw material is required to produce the same amount of product, effectively doubling the efficiency of the input resources. This efficiency gain translates into significant cost savings per kilogram of finished good, allowing for more aggressive pricing strategies or improved margins. Additionally, the use of standard solvents and reagents reduces the need for specialized storage and handling infrastructure, further lowering capital and operational expenditures associated with the manufacturing facility.
- Enhanced Supply Chain Reliability: Relying on widely available reagents like aluminum chloride, zinc iodide, and stannous chloride mitigates the risk of production stoppages due to raw material shortages. These chemicals are staples in the chemical industry with multiple qualified suppliers, ensuring that the supply chain remains resilient against geopolitical or logistical disruptions. The robustness of the reaction conditions, which tolerate standard industrial equipment rather than requiring exotic alloys for corrosion resistance, also means that production can be easily transferred between different manufacturing sites if necessary. This flexibility is crucial for maintaining continuous supply to downstream customers who depend on just-in-time delivery models for their own production schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as reflux, filtration, and crystallization that are easily replicated from pilot plant to commercial scale. The avoidance of hazardous fluorinated acids simplifies environmental compliance and reduces the burden on wastewater treatment systems, aligning with increasingly stringent global regulations on industrial effluents. The high purity of the final product reduces the need for extensive downstream purification, minimizing solvent usage and energy consumption during the isolation phases. This green chemistry approach not only satisfies regulatory requirements but also appeals to end-users who are prioritizing sustainable sourcing in their supply chain audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of ethyl chromane-4-formate using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the source documentation, providing clarity on yield expectations, reagent roles, and scalability factors. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific project requirements. The answers reflect the practical realities of running this chemistry in a GMP-compliant environment.
Q: What is the primary cost advantage of this new synthesis route?
A: The primary advantage is the substitution of expensive trifluoromethanesulfonic acid with inexpensive anhydrous aluminum chloride in the initial Friedel-Crafts step, drastically reducing raw material costs while maintaining high yields.
Q: How does this process improve overall yield compared to traditional methods?
A: By optimizing reaction conditions such as temperature control at 40-45°C and utilizing specific catalysts like zinc iodide and stannous chloride, the process achieves an overall yield of approximately 47%, significantly higher than the traditional 30%.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes standard unit operations such as reflux, extraction, and crystallization with common solvents like nitrobenzene and ethyl acetate, making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl Chromane-4-Formate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-yielding synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the pathway described in CN102603693A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. We are equipped with state-of-the-art reactors capable of handling the specific thermal and corrosive demands of Friedel-Crafts chemistry, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of intermediates at every stage, guaranteeing that the final ethyl chromane-4-formate delivered to your facility is of the highest quality.
We invite you to collaborate with us to leverage this cost-effective technology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. We encourage you to contact us today to discuss your needs,索取 specific COA data for our reference standards, and review our detailed route feasibility assessments. Let us help you secure a reliable supply of this vital intermediate while driving down your overall manufacturing costs through our proven expertise in process optimization and scale-up.
