Advanced Synthesis of Novel Benzofuran Azanaphthalene Dione Derivatives for Oncology Applications
Advanced Synthesis of Novel Benzofuran Azanaphthalene Dione Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking next-generation scaffolds that can overcome the pharmacokinetic limitations of legacy anticancer agents. Patent CN108484617B introduces a groundbreaking class of novel benzofuran azanaphthalene dione derivatives designed specifically to address the poor water solubility and low bioavailability associated with traditional DQ-93 compounds. By strategically introducing nitrogen atoms into the naphthalene ring system, this innovation creates a brand-new parent nucleus framework that exhibits significantly improved physicochemical properties. This structural modification is not merely a theoretical exercise but a practical solution for developing more effective cytotoxic drugs that can interfere with tumor DNA synthesis more efficiently. For R&D teams focused on oncology, this represents a critical evolution in scaffold design, offering a reliable pathway to candidates with better therapeutic indices.
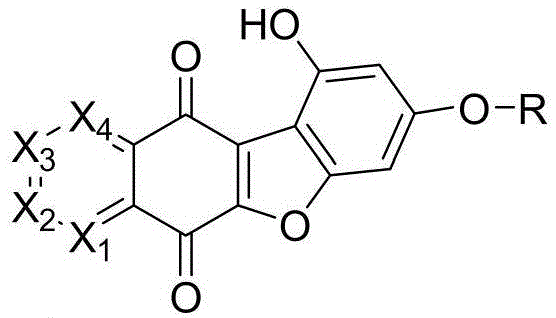
As a reliable pharmaceutical intermediate supplier, we recognize that the transition from concept to commercial reality requires robust synthetic methodologies. The patent details a comprehensive preparation method that allows for the systematic variation of substituents at the X1 through X4 positions, as well as the R side chain, enabling extensive Structure-Activity Relationship (SAR) studies. This flexibility is paramount for medicinal chemists aiming to optimize potency while maintaining favorable ADMET profiles. The ability to tune the electronic and steric environment of the core heterocycle provides a distinct competitive advantage in the race to identify viable clinical candidates. Furthermore, the described synthesis avoids overly exotic reagents, suggesting a pathway that is amenable to cost reduction in API manufacturing through standard process engineering.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of antitumor agents based on the '2-phenyl substituted naphthalene' pharmacophore has been hindered by inherent physicochemical drawbacks. Traditional compounds like DQ-93, while demonstrating significant inhibitory effects on leukemia and small cell lung cancer cell lines, suffer from relatively low solubility in aqueous solutions. This limitation directly translates to poor bioavailability in vivo, necessitating higher dosages that can increase the risk of off-target toxicity and complicate formulation development. Conventional synthetic routes often struggle to introduce polar functionalities without compromising the stability of the quinone moiety, leading to complex purification challenges and lower overall yields. Additionally, the reliance on harsh reaction conditions in older methodologies can generate difficult-to-remove impurities, posing significant hurdles for regulatory approval and commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally reimagines the core architecture by replacing carbon atoms with nitrogen within the naphthalene system to form an aza-naphthalene dione structure. This bioisosteric replacement dramatically alters the electron density and hydrogen bonding capability of the molecule, resulting in markedly improved water solubility and lipid-water distribution properties. The new approach utilizes a modular three-step synthesis that begins with the oxidation of a hydroxy-methoxy precursor, followed by selective demethylation and final alkylation. This sequence allows for precise control over the substitution pattern, ensuring that the resulting derivatives maintain the crucial cytotoxic activity while overcoming the solubility barriers of their predecessors. By focusing on this optimized parent nucleus, the methodology opens up new avenues for creating diverse libraries of compounds with enhanced therapeutic potential.
Mechanistic Insights into Oxidative Quinone Formation and Functionalization
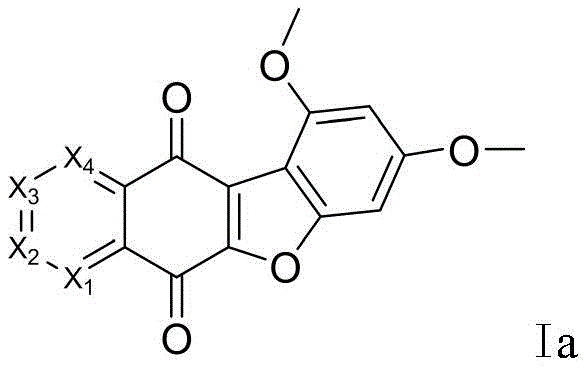
The cornerstone of this synthetic route is the efficient construction of the quinone system, which is achieved through a carefully controlled oxidation process. In the first critical step, the precursor compound I is dissolved in a DMF/methanol solvent system and treated with an oxidant such as potassium nitrosopersulfate. The reaction is maintained at a pH of 6 using a saturated KH2PO4 buffer and heated to a moderate temperature range of 45-50°C. This specific set of conditions is vital for preventing over-oxidation or degradation of the sensitive benzofuran ring while ensuring complete conversion to the desired quinone intermediate Ia. The choice of oxidant is particularly significant; comparative data indicates that potassium nitrosopersulfate outperforms other agents like silver bromide, delivering yields around 78% compared to 76%, which may seem marginal but is crucial for process consistency at scale.
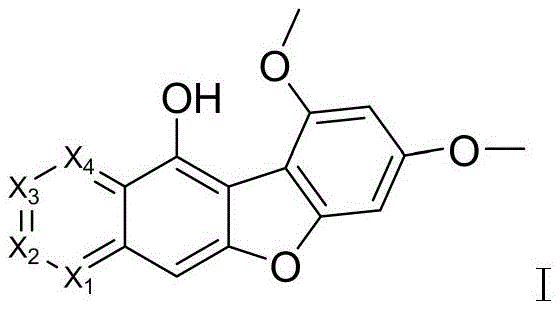
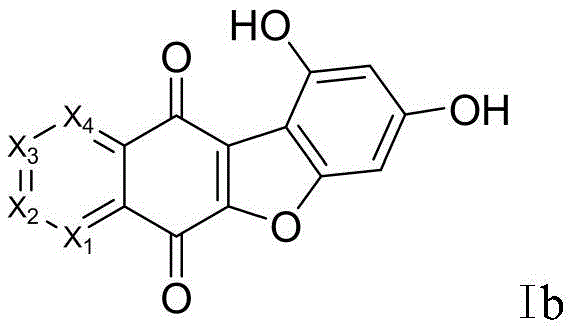
Following the formation of the quinone core, the synthesis proceeds with a demethylation step to reveal the reactive phenolic hydroxyl groups necessary for subsequent functionalization. This transformation is executed using a Lewis acid, preferably boron tribromide (BBr3), in dichloromethane under strictly controlled low-temperature conditions initially, before warming to room temperature. The use of BBr3 is mechanistically superior to protonic acids for cleaving aryl methyl ethers in this context, as evidenced by the high yields of up to 92% reported in the examples. This step generates the dihydroxy intermediate Ib, which serves as the pivotal branching point for introducing various solubilizing side chains. The efficiency of this demethylation is a key factor in the overall economic viability of the process, minimizing material loss and reducing the burden on downstream purification systems.
The final stage of the synthesis involves the alkylation of the dihydroxy intermediate Ib to install the amino-alkoxy side chain, which is essential for modulating the drug's solubility and bioavailability. This reaction is conducted in chloroform in the presence of a suitable base, such as sodium carbonate, and an amino-alkyl halide reagent. The mixture is heated to reflux to drive the nucleophilic substitution to completion, typically requiring 1 to 4 hours. The selection of the base and the specific alkylating agent allows for further diversification of the final product library, enabling the creation of derivatives with tailored pharmacokinetic profiles. Comparative examples highlight that using sodium carbonate provides better yields (around 61%) compared to sodium bicarbonate, underscoring the importance of optimizing even the seemingly minor parameters of the reaction environment to maximize output.
How to Synthesize 8-(2-Diethylaminoethoxy)-6-hydroxybenzofuran Derivatives Efficiently
Executing this synthesis requires strict adherence to the optimized parameters regarding temperature, pH control, and reagent stoichiometry to ensure high purity and yield. The process begins with the dissolution of the starting material in a specific ratio of DMF to methanol, followed by the buffered addition of the oxidant to maintain the critical pH level throughout the reaction. Once the quinone intermediate is isolated and purified via column chromatography, it is immediately subjected to the demethylation conditions using BBr3, taking care to quench the reaction properly to avoid side reactions. Finally, the alkylation step demands careful selection of the base and solvent to facilitate the nucleophilic attack without degrading the sensitive quinone system. For detailed operational protocols and safety guidelines, please refer to the standardized synthesis steps provided below.
- Oxidize the hydroxy-methoxy precursor (Compound I) using potassium nitrosopersulfate in a DMF/methanol system at 45-50°C to form the quinone intermediate (Ia).
- Perform demethylation on intermediate Ia using boron tribromide (BBr3) in dichloromethane under cooling conditions to yield the dihydroxy compound (Ib).
- Conduct alkylation of compound Ib with amino-alkyl halides in chloroform using sodium carbonate as a base under reflux to obtain the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond mere technical novelty. The streamlined three-step sequence minimizes the number of unit operations required, which inherently reduces the consumption of solvents and energy, leading to significant cost reduction in pharmaceutical intermediate manufacturing. By avoiding the use of expensive transition metal catalysts that require complex removal steps, the process simplifies the purification workflow and lowers the overall cost of goods sold. Furthermore, the high yields observed in the demethylation step (up to 92%) ensure that valuable starting materials are utilized efficiently, reducing waste generation and enhancing the sustainability profile of the production line. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of costly noble metal catalysts and the use of readily available oxidants like potassium nitrosopersulfate drastically lower raw material expenses. The high efficiency of the demethylation step minimizes material loss, ensuring that the overall process mass intensity is optimized for large-scale production. Additionally, the simplified workup procedures reduce the consumption of auxiliary chemicals and decrease the time required for batch turnover. These cumulative efficiencies translate into a more competitive pricing structure for the final active pharmaceutical ingredients derived from these intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as DMF, methanol, dichloromethane, and sodium carbonate ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized reagents. The robustness of the reaction conditions, particularly the tolerance for moderate temperatures and standard atmospheric pressure, facilitates easier technology transfer between manufacturing sites. This flexibility allows for diversified sourcing strategies and reduces the risk of production delays due to equipment limitations or regional supply disruptions. Consequently, partners can rely on a consistent and uninterrupted flow of high-quality intermediates to support their clinical and commercial timelines.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing reaction conditions that are easily adaptable from laboratory benchtop to multi-ton industrial reactors. The avoidance of highly toxic heavy metals simplifies wastewater treatment and waste disposal, aligning with increasingly stringent environmental regulations. The high atom economy of the key steps reduces the volume of hazardous waste generated per kilogram of product, supporting corporate sustainability goals. This environmentally conscious approach not only mitigates regulatory risks but also enhances the brand reputation of companies adopting these greener manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel derivatives. Understanding the nuances of the synthetic route and the properties of the final compounds is essential for making informed decisions about their integration into drug development pipelines. The answers provided are based on the specific experimental data and technical disclosures found within the patent literature, ensuring accuracy and relevance for industry professionals. We encourage stakeholders to review these insights to fully appreciate the value proposition of this technology.
Q: What is the primary advantage of the aza-naphthalene structure over traditional naphthalene derivatives?
A: The introduction of nitrogen atoms into the naphthalene ring significantly improves water solubility and lipid-water distribution coefficients, addressing the low bioavailability issues found in traditional DQ-93 compounds.
Q: Which oxidant provides the highest yield in the quinone formation step?
A: According to the experimental data in patent CN108484617B, potassium nitrosopersulfate yields approximately 78%, which is superior to alternatives like silver bromide which yielded 76% in comparative examples.
Q: How is the demethylation step optimized for industrial scale-up?
A: The process utilizes boron tribromide (BBr3) as a Lewis acid in dichloromethane, achieving high conversion rates (up to 92% yield) under controlled temperature conditions, ensuring reproducibility and purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran Azanaphthalene Dione Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure necessary to bring these complex molecules from patent to production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of benzofuran azanaphthalene dione derivatives meets the highest industry standards. Our commitment to quality assurance means that you can trust our intermediates to perform consistently in your downstream synthesis and biological assays, accelerating your path to clinical success.
We invite you to collaborate with us to leverage this advanced technology for your oncology drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project goals. Let us be your partner in transforming innovative chemical structures into life-saving medicines.
