Advanced Manufacturing of Fluorometholone via Streamlined 9,11-Epoxy Intermediates
Advanced Manufacturing of Fluorometholone via Streamlined 9,11-Epoxy Intermediates
The pharmaceutical industry continuously seeks robust and cost-effective pathways for the production of critical glucocorticoid APIs, particularly for ophthalmic and dermatological applications. Patent CN101397325A introduces a transformative methodology for the preparation of fluorometholone and its derivatives, addressing long-standing inefficiencies in steroid synthesis. This innovation leverages 6-methyl-17-hydroxy-1,4,9-triene-pregna-3,20-diketone as a strategic starting material, enabling a concise two-step conversion involving 9,11-epoxidation and subsequent ring-opening. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for high-purity hormonal intermediates. The technology not only simplifies the operational path but also integrates seamlessly with existing production lines for related corticosteroids, offering a compelling value proposition for reliable pharmaceutical intermediates supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorometholone has been dominated by the Upjohn process, which relies on 6-methyl-fluorohydrocortisone as the primary raw material. This legacy approach is fraught with logistical and economic challenges, primarily because the starting material is derived from complex fermentation processes involving 11α,17,21-trihydroxy compounds. The synthetic sequence requires multiple discrete steps, including 21-hydroxyl sulfonylation, substitution with sodium iodide to obtain the 21-iodo compound, reduction to the 21-methyl species, and finally dehydrogenation. As illustrated in the traditional pathway below, the cumulative effect of these steps results in lower overall yields and higher production costs due to the consumption of expensive auxiliary materials and the need for rigorous purification at each stage.
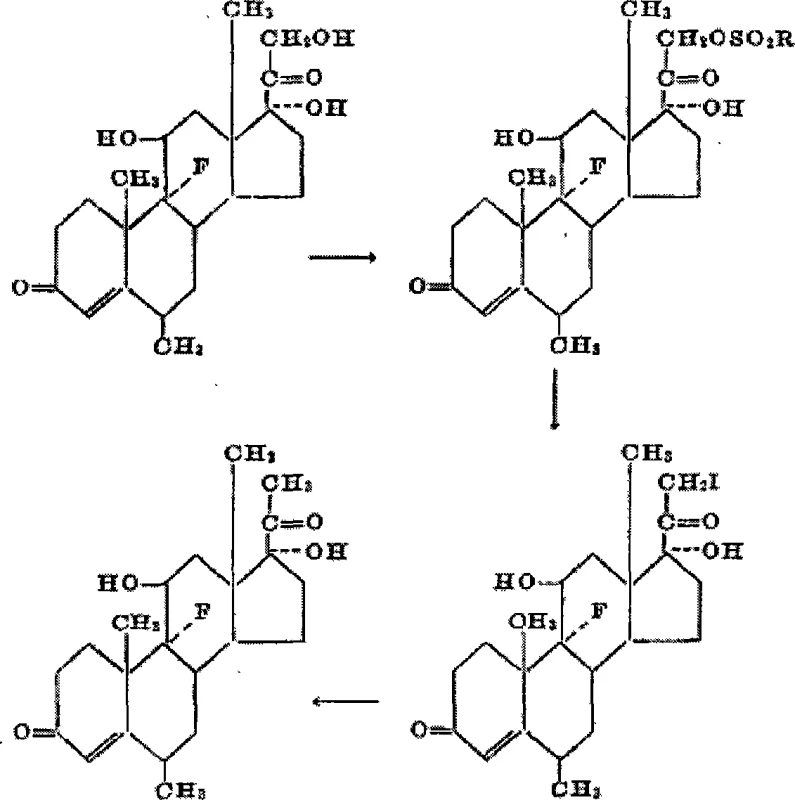
Furthermore, the conventional method suffers from significant drawbacks when attempting to synthesize fluorometholone esters, such as the 17-acetate. Direct esterification of the fluorometholone parent nucleus often leads to competitive reactions at the 11-hydroxyl position, generating substantial by-products that complicate downstream processing. This lack of regioselectivity necessitates extensive recrystallization efforts, further eroding yield and increasing the environmental footprint of the manufacturing process. For supply chain heads, these inefficiencies translate into longer lead times and reduced reliability in meeting market demand for ophthalmic steroids.
The Novel Approach
In stark contrast to the convoluted historical methods, the novel process described in CN101397325A utilizes a readily available intermediate, 6-methyl-17-hydroxy-1,4,9-triene-pregna-3,20-diketone, to achieve the target molecule in merely two chemical transformations. This streamlined approach begins with a 9,11-epoxy reaction followed by a 9,11-ring-opening reaction, effectively installing the critical 9-alpha-fluoro and 11-beta-hydroxy functionalities in a highly controlled manner. By bypassing the fermentation-dependent steps and utilizing a shared intermediate that is also used for hexamethylprednisolone production, manufacturers can achieve doubling production capabilities, significantly enhancing asset utilization and cost reduction in hormone manufacturing.
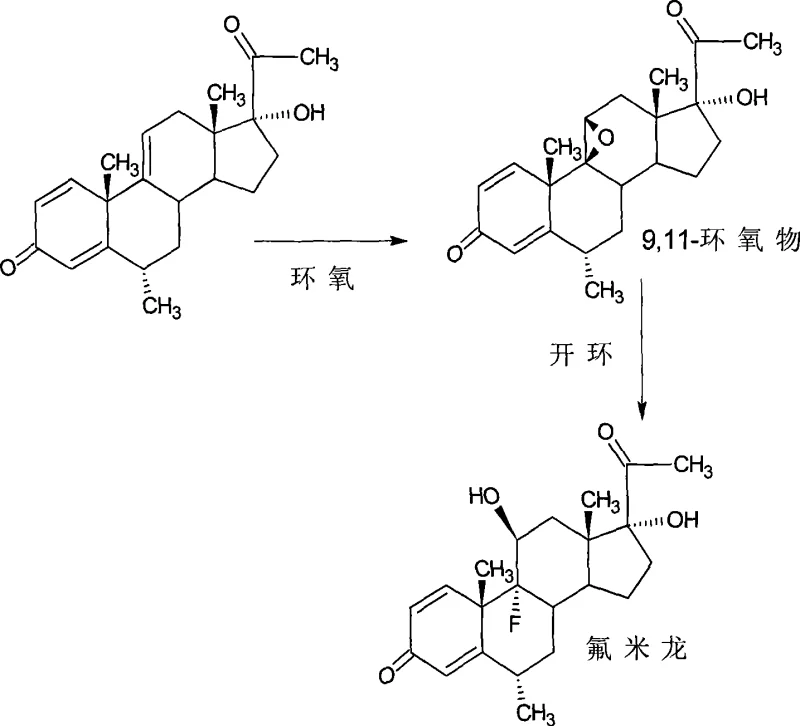
Additionally, this methodology offers a superior strategy for producing fluorometholone esters. By performing the esterification at the 17-position on the precursor compound prior to the epoxidation and ring-opening steps, the process completely avoids the issue of 11-position esterification. This strategic sequencing ensures high regioselectivity and minimizes the formation of difficult-to-remove impurities. The result is a cleaner reaction profile that facilitates easier purification and higher final purity, making it an ideal candidate for the commercial scale-up of complex steroid intermediates required by top-tier pharmaceutical companies.
Mechanistic Insights into 9,11-Epoxidation and Ring-Opening Fluorination
The core of this technological breakthrough lies in the precise control of the 9,11-double bond functionalization. The first stage involves a halogenation-epoxidation sequence where the starting triene-diketone is treated with a halide reagent, such as N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS), in the presence of an acid catalyst like perchloric acid. This reaction, conducted in solvents such as acetone or tetrahydrofuran at temperatures between 0°C and 20°C, generates a 9-halo-11-hydroxy intermediate. Subsequent treatment with a mineral base, preferably sodium hydroxide, induces cyclization to form the stable 9,11-epoxy oxide. This mechanistic pathway is highly robust, allowing for the isolation of the epoxy intermediate or a telescoped one-pot procedure, providing flexibility for different manufacturing scales.
The second critical stage is the regioselective ring-opening of the 9,11-epoxide using hydrogen fluoride. Conducted in polar aprotic solvents like dimethylformamide (DMF) or THF at low temperatures ranging from -5°C to 0°C, this step ensures the nucleophilic attack of the fluoride ion occurs specifically at the C9 position. This results in the formation of the 9-alpha-fluoro-11-beta-hydroxy configuration, which is essential for the biological activity of fluorometholone. The mild reaction conditions and the use of standard reagents minimize side reactions and degradation, ensuring a high-quality impurity profile. For R&D teams, understanding this mechanism is key to validating the process's scalability and reproducibility across different batch sizes.
How to Synthesize Fluorometholone Efficiently
The synthesis of fluorometholone via this patented route is characterized by its operational simplicity and reliance on standard chemical engineering unit operations. The process begins with the dissolution of the starting triene-diketone in a suitable organic solvent, followed by the controlled addition of the halogenating agent under cooling to manage exothermicity. After the formation of the epoxy intermediate, the reaction mixture is neutralized and worked up to isolate the solid intermediate, which serves as the substrate for the fluorination step. The detailed standardized synthesis steps, including specific reagent ratios and workup procedures, are outlined in the guide below.
- Perform 9,11-epoxidation by reacting the starting triene-diketone with N-bromosuccinimide (NBS) in acetone at 0-20°C, followed by base treatment to form the 9,11-epoxy intermediate.
- Execute the 9,11-ring-opening reaction by treating the epoxy intermediate with hydrogen fluoride (gas or solution) in DMF or THF at temperatures between -5°C and 0°C.
- Isolate the final fluorometholone product through neutralization, filtration, and recrystallization from acetone to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere technical elegance. The primary advantage is the drastic simplification of the supply chain for raw materials. By utilizing an intermediate that is already produced in-house for other corticosteroid lines, manufacturers eliminate the dependency on external fermentation sources for 6-methyl-fluorohydrocortisone. This vertical integration significantly reduces the risk of supply disruptions and stabilizes input costs, ensuring a more predictable pricing structure for long-term contracts. Furthermore, the elimination of expensive and hazardous reagents used in the older sulfonylation-iodination sequences contributes to substantial cost savings in waste management and safety compliance.
- Cost Reduction in Manufacturing: The streamlined two-step process inherently lowers the cost of goods sold (COGS) by reducing the number of unit operations and the consumption of solvents and reagents. Unlike the historical method which requires multiple isolation and purification steps, this route allows for high-yield conversions with minimal by-product formation. The avoidance of 11-esterification side reactions in the derivative synthesis further enhances cost efficiency by reducing the loss of valuable material during recrystallization and purification, leading to a more economically viable production model for high-volume API manufacturing.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents such as NBS, sodium hydroxide, and hydrogen fluoride ensures that the production process is not bottlenecked by the availability of exotic catalysts or biologically derived precursors. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without compromising quality, adds a layer of resilience to the manufacturing schedule. This reliability is crucial for maintaining continuous supply to downstream formulation partners, especially in the competitive ophthalmic drug market where consistency is paramount.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed for seamless transition from pilot plant to commercial tonnage. The solvents employed, such as acetone and THF, are widely used in the industry and can be efficiently recovered and recycled, minimizing the environmental footprint. The absence of heavy metal catalysts and the reduction in hazardous waste streams align with modern green chemistry principles, facilitating easier regulatory approval and reducing the burden on effluent treatment facilities. This makes the process highly attractive for manufacturers aiming to expand capacity while adhering to strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorometholone synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical rationale behind the new process. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production portfolios.
Q: What are the primary advantages of this new fluorometholone synthesis route over the traditional Upjohn method?
A: The new route utilizes 6-methyl-17-hydroxy-1,4,9-triene-pregna-3,20-diketone as a starting material, bypassing the complex fermentation and multi-step chemical modifications required by the historical Upjohn process. This significantly shortens the synthetic timeline, eliminates expensive auxiliary materials, and improves overall yield and cost-efficiency for industrial production.
Q: How does this process prevent unwanted esterification at the 11-position during derivative synthesis?
A: The process strategically performs the 17-esterification step on the precursor (Compound I) before the 9,11-epoxidation and ring-opening reactions. By establishing the ester group early in the sequence, the method avoids the competitive esterification at the 11-hydroxyl position that typically occurs when esterifying the final fluorometholone parent nucleus, thereby reducing by-products and simplifying purification.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the method is designed for high feasibility and strong operability. It uses common organic solvents like acetone and THF, and standard reagents like NBS and hydrogen fluoride. The intermediates can be purified by general chemical methods, and the route allows for the doubling of production capacity for both hexamethylprednisolone and fluorometholone series products from a shared intermediate, optimizing resource utilization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorometholone Supplier
The technological advancements detailed in patent CN101397325A underscore the potential for more efficient and sustainable production of critical steroid APIs. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of fluorometholone or its derivatives complies with the highest international pharmacopoeia standards. We are committed to leveraging such innovative synthetic routes to deliver superior value to our global partners.
We invite pharmaceutical companies and contract manufacturers to explore the possibilities of collaborating with us on this advanced platform. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can support your supply chain goals for high-purity fluorometholone and related hormonal intermediates.
