Advanced One-Pot Synthesis of 17 Alpha-Hydroxyprogesterone for Commercial Scale Pharmaceutical Manufacturing
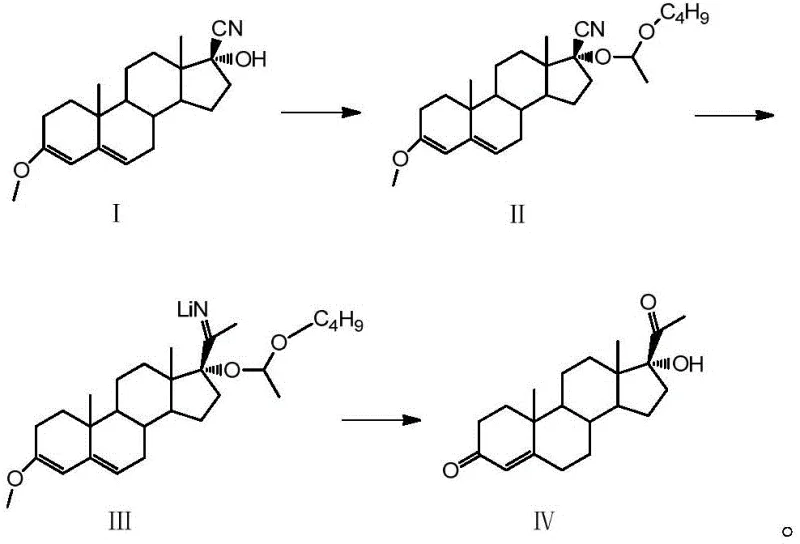
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical steroid intermediates, and the recent disclosure in patent CN112175034B offers a transformative approach to producing 17 alpha-hydroxyprogesterone. This vital compound serves as a cornerstone precursor for synthesizing a wide array of corticosteroids, including cortisone acetate, hydrocortisone, and dexamethasone, which are essential for treating inflammatory and autoimmune conditions. The disclosed methodology represents a significant leap forward in process chemistry, utilizing a streamlined one-pot technique that converts Compound I directly into the target 17 alpha-hydroxyprogesterone IV through sequential etherification, cyanomethylation, and hydrolysis reactions. By integrating these steps into a cohesive workflow, the process not only simplifies operational complexity but also dramatically enhances the overall molar yield to levels exceeding 94%, far surpassing the limitations of legacy technologies. For R&D directors and procurement strategists, this innovation signals a pivotal opportunity to optimize supply chains for high-purity pharmaceutical intermediates while mitigating the environmental and safety risks associated with traditional multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 17 alpha-hydroxyprogesterone has been plagued by inefficient and hazardous synthetic routes that struggle to meet modern standards for sustainability and cost-effectiveness. The earliest methods relied on saponin or diosgenin as raw materials, necessitating a treacherous sequence of oxygen bridging, Wolff oxidation, bromination, and debromination, which resulted in severe pollution, low yields, and significant safety hazards that have largely led to the elimination of this pathway. Even the more contemporary approach utilizing 4-androstenedione, derived from the microbial degradation of cholesterol or phytosterols, suffers from substantial drawbacks, particularly regarding yield and operational complexity. In these existing processes, the molar yield typically hovers around 82%, primarily due to uncontrolled side reactions during the critical 17-position hydroxyl etherification step where strong acids like p-toluenesulfonic acid are employed. Furthermore, the use of such aggressive catalysts often triggers violent polymerization of butyl vinyl ether, leading to deeply colored products that require extensive and costly refining procedures to remove polymeric impurities, thereby inflating production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel process described in the patent introduces a sophisticated one-pot strategy that fundamentally reengineers the reaction landscape to maximize efficiency and product quality. By substituting harsh catalysts with mild alternatives such as pyridine hydrochloride or trifluoroacetic acid, the new method effectively suppresses side reactions and prevents the polymerization of butyl vinyl ether, ensuring that the reaction mixture remains clean and the product retains a desirable color profile without the need for post-synthesis refining. This strategic shift allows for the direct isolation of qualified products with purity levels exceeding 99.5% and content greater than 99%, effectively bypassing the purification bottlenecks that hinder conventional manufacturing. Additionally, the clever selection of diethoxymethane as a universal solvent serves a dual purpose: it acts as the reaction medium for the initial etherification and simultaneously functions as the solvent for the methyllithium reagent in the subsequent cyanomethylation step, facilitating seamless transitions between reaction stages and enabling efficient solvent recycling. This holistic optimization results in a stable, safe, and highly selective process that is uniquely suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into One-Pot Sequential Synthesis
The core of this technological breakthrough lies in the precise control of reaction conditions and the synergistic interaction of reagents within the one-pot system, which collectively drive the high selectivity observed in the transformation of Compound I to Compound IV. The initial hydroxyl etherification is meticulously conducted under inert gas protection at temperatures between 40-60°C, where the mild acidity of the chosen catalyst activates the 17-position hydroxyl group for etherification with butyl vinyl ether without degrading the sensitive steroid backbone. Following this, the cyanomethylation step leverages the unique solubility properties of diethoxymethane to maintain the high activity of the methyllithium reagent, ensuring that the nucleophilic attack on the nitrile group proceeds with minimal interference from competing side reactions. The reaction temperature is tightly controlled between 10-20°C during this phase to further stabilize the reactive intermediates, while the subsequent workup involving ammonium chloride quenching and phase separation efficiently removes inorganic byproducts before the final hydrolysis. This careful orchestration of chemical events ensures that the intermediate Compound III is generated in situ with high fidelity, setting the stage for the final conversion to the target ketone.
Impurity control is another critical aspect of this mechanism, achieved primarily through the prevention of polymer formation and the strategic use of crystallization solvents to purge residual contaminants. In traditional routes, the polymerization of vinyl ethers creates high-molecular-weight impurities that are notoriously difficult to separate from the steroid product, often necessitating chromatographic purification or repeated recrystallizations that erode overall yield. The new method circumvents this issue entirely by maintaining a reaction environment where polymerization is thermodynamically unfavorable, thus preserving the optical clarity and chemical integrity of the crude product. Furthermore, the hydrolysis step utilizes solvents such as acetone, tetrahydrofuran, or isopropanol, which not only facilitate the cleavage of the ether protecting group but also act as anti-solvents during the final crystallization, effectively precipitating the pure 17 alpha-hydroxyprogesterone while leaving soluble impurities in the mother liquor. This dual functionality of the hydrolysis solvent system exemplifies the process's elegance, delivering a final product with a maximum single impurity content as low as 0.11% as confirmed by HPLC analysis.
How to Synthesize 17 Alpha-Hydroxyprogesterone Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield on an industrial scale. The process begins with the preparation of the first reaction solution containing Compound II via hydroxyl etherification, followed immediately by the addition of the methyllithium reagent for cyanomethylation without isolating the intermediate, which significantly reduces handling time and exposure to atmospheric moisture. The detailed standardized synthesis steps involve precise control of stoichiometric ratios, such as maintaining a mass ratio of Compound I to diethoxymethane between 1:4 and 1:10, and carefully managing the exothermic nature of the methyllithium addition to prevent thermal runaway. For a comprehensive guide on executing these steps with precision, please refer to the structured protocol below.
- Perform hydroxyl etherification of Compound I using diethoxymethane, butyl vinyl ether, and a mild catalyst like pyridine hydrochloride at 40-60°C.
- Conduct cyanomethylation by adding methyllithium reagent to the first reaction solution, followed by extraction and concentration to obtain Compound III.
- Execute hydrolysis using hydrochloric acid aqueous solution at 20-30°C, followed by pH adjustment and crystallization to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method translates into tangible strategic advantages that extend far beyond simple yield improvements, fundamentally altering the cost structure and reliability of the supply base. The elimination of refining steps due to the high purity of the crude product directly reduces the consumption of auxiliary materials, energy, and labor, leading to substantial cost savings in 17 alpha-hydroxyprogesterone manufacturing without compromising on quality specifications. Moreover, the stability of the reaction and the use of recyclable solvents like diethoxymethane minimize waste generation and simplify wastewater treatment protocols, aligning the production process with increasingly stringent environmental regulations and reducing the risk of regulatory shutdowns. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands with shorter lead times and greater consistency.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the drastic simplification of the downstream processing workflow which traditionally accounts for a significant portion of total production expenses. By preventing the formation of colored polymeric impurities, the method eliminates the need for expensive decolorization agents, activated carbon treatments, or complex chromatographic separations that are standard in older protocols. Additionally, the ability to recycle diethoxymethane across multiple reaction steps reduces the volume of fresh solvent required per batch, lowering raw material procurement costs and waste disposal fees. This lean manufacturing approach ensures that the cost of goods sold is significantly optimized, allowing for more competitive pricing strategies in the global marketplace for steroid intermediates.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this one-pot method offers superior predictability compared to fragile multi-step syntheses that are prone to failure at intermediate stages. The use of readily available starting materials like 4-androstenedione derivatives and common reagents such as butyl vinyl ether ensures that raw material sourcing is not a bottleneck, while the high selectivity of the reaction minimizes the risk of batch failures due to off-spec impurities. This reliability enables suppliers to commit to firmer delivery schedules and maintain consistent inventory levels, which is critical for pharmaceutical customers who operate on tight Just-In-Time manufacturing schedules and cannot afford disruptions in their API production lines.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, featuring stable reaction conditions that do not require extreme temperatures or pressures, making it safe to operate in large-scale reactors without specialized high-pressure equipment. The reduction in hazardous waste streams, particularly the avoidance of heavy metal catalysts or toxic brominating agents found in obsolete routes, simplifies environmental compliance and reduces the carbon footprint of the manufacturing facility. This alignment with green chemistry principles not only future-proofs the supply chain against tightening environmental legislation but also enhances the brand reputation of the manufacturer as a sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their supply networks. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for appreciating the full scope of the process improvements and how they translate into commercial value.
Q: What is the primary advantage of the new synthesis method over traditional routes?
A: The new method achieves a molar yield exceeding 94%, significantly higher than the traditional 82%, while eliminating the need for complex refining processes due to reduced side reactions and polymerization.
Q: Why are pyridine hydrochloride or trifluoroacetic acid preferred as catalysts?
A: These mild catalysts prevent the violent polymerization of butyl vinyl ether often caused by strong acids like p-toluenesulfonic acid, thereby solving product discoloration issues and improving selectivity.
Q: How does the solvent system contribute to process efficiency?
A: Using diethoxymethane as a dual-purpose solvent for both the etherification step and as a carrier for the methyllithium reagent simplifies solvent recovery and ensures high reactivity during cyanomethylation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17 Alpha-Hydroxyprogesterone Supplier
As a premier CDMO and fine chemical manufacturer, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this cutting-edge synthesis technology to deliver high-purity 17 alpha-hydroxyprogesterone that meets the rigorous demands of the global pharmaceutical industry. Our state-of-the-art facilities boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can seamlessly transition this innovative lab-scale protocol into full-scale manufacturing operations. With our stringent purity specifications and rigorous QC labs equipped with advanced analytical instrumentation, we guarantee that every batch of 17 alpha-hydroxyprogesterone delivered adheres to the highest international quality standards, providing our partners with absolute confidence in their raw material supply.
We invite forward-thinking pharmaceutical companies and chemical distributors to collaborate with us to unlock the full potential of this efficient manufacturing route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments to validate the integration of this material into your existing production workflows. Let us partner with you to drive down costs and enhance the reliability of your steroid intermediate supply chain today.
