Revolutionizing Aromatic Nitration: Scalable Green Catalysis for High-Purity Intermediates
Revolutionizing Aromatic Nitration: Scalable Green Catalysis for High-Purity Intermediates
The chemical manufacturing landscape is undergoing a profound transformation driven by the urgent need for sustainable and efficient synthetic methodologies, particularly in the realm of electrophilic aromatic substitution. Patent CN102408339A introduces a groundbreaking approach to the clean nitration of aromatic hydrocarbons, utilizing a novel class of heteropolyacid pyridinium salt ionic liquids as highly effective catalysts. This technology represents a significant departure from the century-old reliance on corrosive nitric-sulfuric mixed acids, offering a pathway to high-purity nitro-aromatic intermediates with drastically reduced environmental footprints. By leveraging the unique superacidic properties of heteropolyanions combined with the tunable solubility of pyridinium cations, this method achieves exceptional catalytic activity under mild conditions. For R&D directors and process engineers, this patent provides a robust framework for developing cleaner, safer, and more economically viable production routes for critical pharmaceutical and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For over a hundred years, the industrial standard for aromatic nitration has relied heavily on the use of mixed acids, typically comprising concentrated nitric acid and concentrated sulfuric acid. While effective, this traditional methodology suffers from severe inherent drawbacks that pose significant challenges for modern chemical manufacturing and supply chain management. The process generates massive quantities of spent acid waste, which is highly corrosive and requires expensive, energy-intensive neutralization and disposal procedures. Furthermore, the harsh oxidative environment often leads to unwanted side reactions, such as oxidation of sensitive functional groups or polysubstitution, complicating the purification process and reducing overall yield. The equipment corrosion associated with mixed acids also necessitates the use of specialized, costly materials of construction, driving up capital expenditure and maintenance costs for production facilities.
The Novel Approach
In stark contrast to the legacy mixed acid processes, the technology disclosed in CN102408339A utilizes a task-specific ionic liquid that functions simultaneously as a solvent and a catalyst, fundamentally altering the reaction landscape. This novel system employs a pyridinium-based cation paired with a bulky heteropolyacid anion, creating a medium that is not only highly acidic but also immiscible with the organic product phase under specific conditions. This biphasic nature allows for facile product separation simply by decanting the upper organic layer, leaving the catalyst in the lower phase for immediate regeneration and reuse. The elimination of concentrated sulfuric acid removes the primary source of waste and corrosion, while the mild reaction conditions preserve the integrity of sensitive substrates. This approach aligns perfectly with the principles of green chemistry, offering a scalable solution that addresses both economic and environmental pain points in fine chemical synthesis.
Mechanistic Insights into Heteropolyacid Pyridinium Ionic Liquid Catalysis
The exceptional catalytic performance of this system stems from the synergistic interaction between the Brønsted acidic protons of the heteropolyacid anion and the structural stability of the pyridinium cation. Heteropolyacids, such as phosphomolybdic acid (H3PMo12O40) or phosphotungstic acid (H3PW12O40), are known for their superacidic strength, which far exceeds that of conventional mineral acids like sulfuric acid. In the ionic liquid form, these anions provide a high density of active protons necessary for generating the nitronium ion (NO2+) electrophile from nitric acid, yet they remain stable and do not decompose under the reaction conditions. The pyridinium cation, specifically functionalized with a propyl sulfonic acid group as shown in the structural diagram, further enhances the acidity and facilitates the solubilization of reactants. 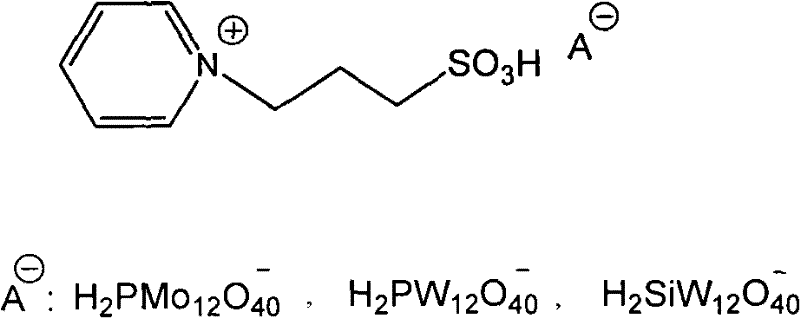
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods by minimizing oxidative degradation and polysubstitution. The controlled acidity of the ionic liquid prevents the excessive generation of reactive species that typically lead to tar formation or over-nitration. Moreover, the phase separation behavior ensures that the product is rapidly removed from the catalytic environment once formed, preventing secondary reactions that could compromise purity. The water tolerance of the heteropolyacid anion is another critical feature; unlike many Lewis acid catalysts that are deactivated by moisture, this system remains active even in the presence of water generated during the nitration reaction. This stability allows for the use of aqueous nitric acid solutions and simplifies the catalyst recycling process, as simple dehydration is sufficient to restore catalytic activity for subsequent batches.
How to Synthesize Nitro-Aromatics Efficiently
The implementation of this catalytic system in a laboratory or pilot plant setting is straightforward and relies on standard unit operations familiar to chemical engineers. The process involves mixing the aromatic substrate with aqueous nitric acid and a catalytic amount of the ionic liquid, followed by heating under atmospheric pressure. The reaction progress is monitored until completion, after which the mixture is allowed to cool and settle, resulting in a clear separation of the product phase from the catalyst phase. Detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures for various substrates like toluene and chlorobenzene, are outlined in the comprehensive guide below.
- Prepare the reaction mixture by combining the aromatic hydrocarbon substrate and nitric acid (65-70% concentration) with the heteropolyacid pyridinium ionic liquid catalyst (1-10 mol%).
- Heat the mixture under atmospheric pressure with stirring at temperatures between 40°C and 80°C for a duration of 1 to 10 hours depending on substrate reactivity.
- Allow the reaction system to stand for phase separation, remove the upper organic layer containing the nitro-product, and regenerate the lower catalyst layer by dehydration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this heteropolyacid ionic liquid technology translates into tangible strategic advantages regarding cost structure and operational reliability. The primary driver for cost reduction lies in the elimination of the massive waste streams associated with spent sulfuric acid, which traditionally accounts for a significant portion of downstream processing expenses. By removing the need for neutralization and complex waste treatment, manufacturers can achieve substantial savings in both raw material consumption and environmental compliance costs. Additionally, the recyclability of the catalyst means that the initial investment in the ionic liquid is amortized over numerous production cycles, further driving down the variable cost per kilogram of the final product.
- Cost Reduction in Manufacturing: The transition away from mixed acid nitration eliminates the requirement for expensive corrosion-resistant reactor linings and reduces the consumption of neutralizing agents like caustic soda. Since the catalyst can be recovered and reused multiple times without significant loss of activity, the overall material cost for the nitration step is drastically simplified. This efficiency gain allows for a more competitive pricing structure for high-purity nitro-intermediates, enhancing margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the heteropolyacid ionic liquid against water deactivation ensures consistent batch-to-batch performance, reducing the risk of production delays caused by catalyst failure or inconsistent quality. The simplicity of the workup procedure, which relies on gravity phase separation rather than complex extraction or distillation steps, shortens the overall cycle time for each batch. This operational efficiency contributes to reducing lead time for high-purity nitro compounds, enabling suppliers to respond more agilely to fluctuating market demands.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard stainless steel equipment, as the absence of concentrated sulfuric acid mitigates severe corrosion risks. The process inherently generates less hazardous waste, aligning with increasingly stringent global environmental regulations and reducing the regulatory burden on manufacturing sites. This sustainability profile makes the supply chain more resilient against future regulatory changes and enhances the brand value of the final chemical products in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of heteropolyacid ionic liquid catalysis in industrial nitration processes. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this green chemistry solution into existing production workflows.
Q: How does this ionic liquid catalyst improve upon traditional mixed acid nitration?
A: Unlike traditional nitric-sulfuric mixed acids which generate severe corrosion and hazardous waste, this heteropolyacid ionic liquid is water-stable, recyclable, and eliminates the need for concentrated sulfuric acid, drastically reducing environmental impact and post-treatment costs.
Q: Is the catalyst recoverable for industrial scale-up?
A: Yes, the patent demonstrates that after reaction, the system separates into two liquid phases. The lower layer contains the catalyst and water, which can be dehydrated and directly reused for subsequent batches without significant loss of activity.
Q: What types of aromatic compounds are suitable for this process?
A: The technology is versatile and has been successfully applied to various substrates including toluene, chlorobenzene, fluorobenzene, and p-chlorobenzotrifluoride, yielding high-purity mononitro products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heteropolyacid Ionic Liquid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies like the heteropolyacid pyridinium ionic liquid system in advancing the fine chemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust manufacturing processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of nitro-aromatic intermediate meets the exacting standards required by the global pharmaceutical and agrochemical sectors.
We invite you to collaborate with us to leverage this advanced nitration technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current production bottlenecks, demonstrating how this catalyst can optimize your operational expenditures. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you build a more sustainable and efficient supply chain.
