Advanced Synthesis of Monohalogenated Phenylboronic Acid for Global Pharmaceutical Manufacturing
The chemical landscape for critical coupling partners is evolving rapidly, driven by the relentless demand for higher purity and safer manufacturing processes in the pharmaceutical and electronic sectors. Patent CN111647011B introduces a groundbreaking preparation method for monohalogenated phenylboronic acid that fundamentally shifts the paradigm from hazardous cryogenic chemistry to a robust, catalytic Grignard exchange protocol. This technology addresses the longstanding bottleneck of producing high-purity intermediates required for Suzuki and Sonogashira couplings, which are indispensable in the synthesis of Active Pharmaceutical Ingredients (APIs) and Organic Light Emitting Diode (OLED) materials. By leveraging a unique catalyst system comprising a mixture of lithium salts and alkaline ionic liquids, the process achieves an exceptional HPLC content of more than 99.5% and a total yield exceeding 80%, setting a new benchmark for quality in fine chemical synthesis.
Monohalogenated phenylboronic acids serve as pivotal building blocks in modern medicinal chemistry, yet their synthesis has historically been plagued by selectivity issues and safety concerns. The traditional reliance on direct Grignard formation from dihalobenzenes often results in uncontrollable side reactions, where the high activity of certain halogens leads to self-coupling or the formation of difficult-to-remove impurities like phenyl diboronic acids. Furthermore, alternative lithiation methods utilizing butyl lithium necessitate extreme low-temperature conditions around -78°C, imposing severe energy costs and safety risks on manufacturing facilities. The innovation disclosed in this patent circumvents these limitations by introducing a controlled exchange mechanism that selectively targets one halogen atom while preserving the other, thereby ensuring the structural integrity required for downstream cross-coupling applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic routes for generating monohalogenated phenylboronic acids face significant hurdles that impact both economic viability and product quality. When attempting to prepare these compounds directly from dihalobenzenes using standard Grignard reagents, the similar reactivity of halogens such as chlorine and bromine often leads to a statistical mixture of products. This lack of chemoselectivity inevitably generates impurities, specifically other halogen-substituted phenylboronic acids and phenyl diboronic acids, which are notoriously difficult to separate and can poison downstream catalytic cycles. Additionally, the prior art reports yields generally less than 60% for dichlorobenzene or dibromobenzene substrates, with purities failing to reach the stringent 98% threshold required for high-value applications. The reliance on butyl lithium for lithiation further exacerbates these issues by introducing extreme operational hazards, requiring specialized cryogenic equipment and inert atmosphere handling that drastically increases capital expenditure and operational complexity.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this synthesis by employing a sophisticated catalyst system that modulates the reactivity of the Grignard exchange reaction. By utilizing a mixture of lithium salts, such as lithium chloride or lithium formate, combined with alkaline ionic liquids like 1-butyl-3-methylimidazole hydroxide or formate, the process achieves unprecedented control over the reaction pathway. This catalytic environment effectively promotes the exchange of the Grignard reagent with the dihalobenzene substrate at mild temperatures ranging from 0°C to 80°C, completely eliminating the need for dangerous cryogenic conditions. The result is a dramatic improvement in process metrics, with the total yield of the product consistently surpassing 80% and the content of critical impurities, such as monohalogenated phenylboronic acid of another halogen and phenyl diboronic acid, suppressed to levels below 0.003%. This level of purity ensures that the material meets the rigorous specifications of modern fine chemical synthesis without the need for extensive and yield-eroding purification steps.
Mechanistic Insights into Lithium Salt and Ionic Liquid Catalyzed Grignard Exchange
The core of this technological breakthrough lies in the synergistic interaction between the lithium salt and the alkaline ionic liquid, which creates a highly active yet selective catalytic species for the Grignard exchange. In this mechanism, the alkaline ionic liquid likely acts as a phase transfer catalyst or a ligand that stabilizes the magnesium species, facilitating the transmetallation process between the alkyl magnesium chloride and the dihalobenzene. The presence of the lithium salt further enhances the nucleophilicity of the Grignard reagent while simultaneously coordinating with the halogen leaving group, lowering the activation energy for the exchange reaction. This precise tuning of the reaction environment allows for the selective activation of the more reactive halogen (such as iodine or bromine) in the presence of a less reactive one (such as chlorine), or manages the reactivity of similar halogens to prevent double substitution. Consequently, the formation of the desired monohalophenyl magnesium chloride intermediate is favored kinetically and thermodynamically, preventing the runaway reactions that typically plague uncatalyzed systems.
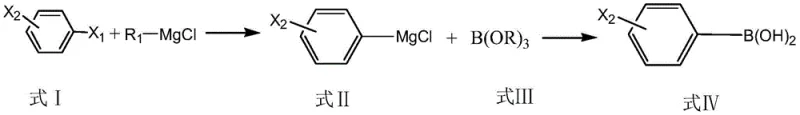
Impurity control is rigorously maintained through the specific choice of catalyst components and reaction conditions, which suppress the formation of bis-Grignard species that lead to diboronic acid byproducts. In conventional methods, the high reactivity of the Grignard reagent often attacks both halogen positions on the benzene ring, especially when using substrates like diiodobenzene or bromoiodobenzene, leading to significant contamination. However, the optimized catalyst system described in the patent moderates this reactivity, ensuring that once the first exchange occurs, the resulting monohalophenyl magnesium chloride is rapidly trapped by the borate ester in the subsequent step. The process involves reacting the intermediate with boric acid esters like trimethyl borate or triisopropyl borate at controlled temperatures, followed by a mild acidic hydrolysis. This sequence ensures that the final hydrolysis step yields the target monohalogenated phenylboronic acid with an HPLC content greater than 99.5%, effectively eliminating the spectral interference and catalytic poisoning associated with lower-grade intermediates.
How to Synthesize Monohalogenated Phenylboronic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing these valuable intermediates with high efficiency and safety. The process begins with the preparation of a standard alkyl magnesium chloride solution, followed by the critical addition of the lithium salt and alkaline ionic liquid catalyst mixture. This catalytic cocktail is then introduced to the dihalobenzene substrate under inert gas protection, where the Grignard exchange proceeds smoothly at temperatures between 0°C and 80°C, depending on the specific halogen combination. Following the exchange, the reaction mixture is treated with a borate ester to form the boronic acid ester intermediate, which is subsequently hydrolyzed under acidic conditions to release the final product. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and temperature profiles for each stage, are provided in the guide below to ensure reproducible results for process chemists.
- Prepare alkyl magnesium chloride (R1-MgCl) by reacting chloroalkane with metallic magnesium in an organic solvent at 0-80°C.
- Perform Grignard exchange reaction by adding a catalyst mixture of lithium salt and alkaline ionic liquid to the Grignard reagent, followed by dihalobenzene at 0-80°C.
- React the resulting monohalophenyl magnesium chloride with a borate ester (B(OR)3) at temperatures between -20°C and ambient conditions.
- Hydrolyze the intermediate ester under acidic conditions at -5 to 20°C to obtain the final high-purity monohalogenated phenylboronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple yield improvements. By replacing hazardous and expensive reagents like butyl lithium with common, commercially available lithium salts and recyclable ionic liquids, the process significantly reduces the raw material cost base and minimizes the dependency on specialized supply chains for dangerous chemicals. The elimination of cryogenic requirements translates directly into substantial capital expenditure savings, as facilities no longer need to invest in or maintain complex low-temperature reactors and cooling infrastructure. Furthermore, the mild reaction conditions enhance operational safety, reducing the risk of thermal runaways and exposure to pyrophoric reagents, which in turn lowers insurance premiums and regulatory compliance burdens associated with hazardous material handling.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the substitution of high-cost lithiation reagents with inexpensive Grignard exchange catalysts. The process utilizes readily available dihalobenzenes and standard alkyl chlorides, which are commodity chemicals with stable pricing and abundant global supply. By avoiding the use of butyl lithium, manufacturers eliminate the significant costs associated with its storage, handling, and disposal, while also removing the energy-intensive requirement for maintaining reactions at -78°C. The high selectivity of the reaction means that downstream purification costs are drastically reduced, as the crude product already meets high-purity specifications, minimizing solvent usage and waste generation during recrystallization.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method ensures consistent availability of critical intermediates, mitigating the risk of production stoppages due to reagent shortages. The raw materials, including various dihalobenzenes and magnesium metal, are produced on a massive industrial scale, ensuring a secure and diversified supply base that is less susceptible to market volatility. The simplicity of the operation, which does not require highly specialized operator skills or exotic equipment, allows for flexible manufacturing across multiple sites, enhancing the overall resilience of the supply network. This reliability is crucial for pharmaceutical companies that require uninterrupted access to high-quality intermediates to maintain their own production schedules for life-saving medications.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, with reaction conditions that are easily manageable in large-scale reactors without the engineering challenges posed by extreme temperatures. The use of green solvents and the reduction of hazardous waste streams align perfectly with modern environmental, social, and governance (ESG) goals, facilitating easier permitting and regulatory approval in strict jurisdictions. The high atom economy and reduced impurity profile mean that less waste is generated per kilogram of product, lowering the environmental footprint and the costs associated with waste treatment and disposal. This sustainability advantage positions the technology as a future-proof solution for the long-term manufacturing of complex pharmaceutical intermediates and electronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for evaluating the technology's fit for your specific application. Understanding these details is essential for R&D teams planning process transfers and procurement officers assessing vendor capabilities.
Q: How does this method improve purity compared to traditional Butyl Lithium routes?
A: Traditional Butyl Lithium routes require cryogenic temperatures (-78°C) and often struggle with selectivity, leading to impurities. This novel method uses a lithium salt and alkaline ionic liquid catalyst system that operates at mild temperatures (0-80°C), effectively suppressing side reactions and achieving HPLC purity greater than 99.5% with impurity levels below 0.003%.
Q: What are the specific advantages of using alkaline ionic liquids in this synthesis?
A: The use of alkaline ionic liquids, such as imidazole derivatives, combined with lithium salts acts as a highly efficient catalyst for the Grignard exchange. This eliminates the need for hazardous and expensive reagents like Butyl Lithium, reduces equipment requirements by avoiding ultra-low temperatures, and significantly enhances the conversion rate while minimizing the formation of diboronic acid impurities.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes common raw materials like dihalobenzenes and standard Grignard reagents, operates under safe and mild conditions, and achieves total yields exceeding 80%. The simplified operation and high safety profile make it ideal for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monohalogenated Phenylboronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the success of your drug development and manufacturing programs. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. Our commitment to quality is unwavering, with stringent purity specifications enforced through our rigorous QC labs, guaranteeing that every batch of monohalogenated phenylboronic acid meets the exacting standards required for Suzuki coupling and other sensitive transformations. We understand that consistency is key in pharmaceutical manufacturing, and our state-of-the-art facilities are equipped to handle the specific nuances of this catalytic Grignard exchange technology.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this process can optimize your overall manufacturing budget. Please contact our technical procurement team today to request specific COA data for our current inventory and to discuss route feasibility assessments for your target molecules. Let us be your partner in driving innovation and efficiency in your supply chain.
