Advanced Grignard-Based Synthesis of 2,5-Dihydroxy Terephthalic Acid for High-Performance Polymers
The chemical industry is constantly seeking more efficient pathways to synthesize critical monomers for high-performance materials, and patent CN102211996A presents a significant breakthrough in the production of 2,5-dihydroxy terephthalic acid (DHTA). This valuable compound serves as a pivotal intermediate for synthesizing organic luminescence polymers, advanced dyes, and high-strength fibers like poly-(2,5-dihydroxy-1,4-phenylene pyrido diimidazole) resin. Traditional methods often suffer from severe limitations regarding safety and operational complexity, but this novel approach leverages a Grignard reaction mechanism to achieve superior results. By shifting from high-pressure carboxylation to a controlled atmospheric process, the technology offers a robust solution for manufacturers aiming to optimize their supply chains for specialty chemicals. The method ensures stability in reaction conditions and delivers products with exceptional purity, addressing the critical needs of R&D directors who require consistent quality for downstream polymerization processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxy-benzoic acid compounds like DHTA has relied heavily on the Kolbe-Schmitt reaction, a process that necessitates the use of high-pressure carbon dioxide in neutral solvents. This requirement introduces substantial safety hazards due to the danger coefficient associated with high-pressure operations, alongside the need for expensive, specialized reactor equipment capable of withstanding such conditions. Furthermore, alternative methods described in prior art, such as those utilizing copper sources and coordination complexes, often demand excessively long reaction times exceeding 40 hours, which drastically reduces throughput and increases energy consumption. These conventional routes also frequently encounter issues with low transformation efficiency and complex separation procedures, leading to lower overall yields and higher production costs. The combination of harsh reaction conditions, prolonged processing times, and difficult purification steps creates a significant bottleneck for commercial scale-up of complex aromatic dicarboxylic acids.
The Novel Approach
In stark contrast, the methodology disclosed in CN102211996A utilizes a Grignard reaction mechanism starting from 1,4-dihalo-2,5-dialkoxy benzene to construct the carbon framework under mild conditions. This innovative route eliminates the need for high-pressure equipment by conducting the carbochain growth step at normal pressure, thereby drastically simplifying the engineering requirements and enhancing operational safety. The process is characterized by its short reaction period and ease of control, allowing for precise management of the exothermic Grignard formation and subsequent carboxylation steps. By employing anhydrous solvents and dry carbon dioxide sources, the method achieves high conversion rates and minimizes side reactions that typically plague older technologies. This shift not only improves the economic viability of DHTA manufacturing but also provides a more reliable pharmaceutical intermediate supplier pathway for clients demanding consistent batch-to-batch quality.
Mechanistic Insights into Grignard Carboxylation and Hydrolysis
The core of this synthesis lies in the precise formation of the Grignard reagent, where magnesium metal reacts with 1,4-dihalo-2,5-dialkoxy benzene in an anhydrous environment such as tetrahydrofuran (THF). This step is critical, as the presence of moisture would quench the organometallic species, so the process strictly maintains drying conditions and nitrogen protection to ensure maximum reagent stability. Once the Grignard reagent is formed, it acts as a potent nucleophile that attacks dry carbon dioxide, effectively inserting a carboxyl group into the aromatic ring structure to form the 2,5-dialkoxy terephthalic acid disalt. This carbochain increase is highly efficient and occurs rapidly under cold conditions, preventing thermal degradation of the sensitive intermediate. The subsequent acidification step converts the disalt into the free acid, setting the stage for the final deprotection phase.
Following the construction of the dicarboxylic acid backbone, the process moves to the hydrolysis stage where the alkoxy protecting groups are removed to reveal the target phenolic hydroxyl groups. This is achieved using strong acid systems, such as concentrated hydroiodic acid or boron tribromide solutions, which cleave the ether bonds under heated conditions. The choice of acid system is crucial for minimizing impurities; for instance, using a mixture of hydroiodic acid and glacial acetic acid allows for controlled cleavage without damaging the sensitive aromatic core. The resulting 2,5-dihydroxy terephthalic acid precipitates out of the solution, allowing for straightforward filtration and washing to remove residual salts and acid traces. This rigorous purification protocol ensures that the final product meets stringent purity specifications required for high-end electronic and polymer applications.
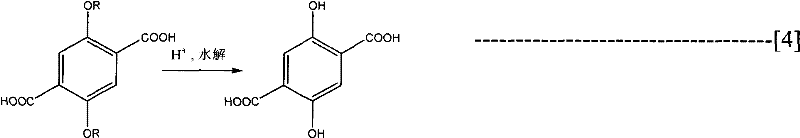
How to Synthesize 2,5-Dihydroxy Terephthalic Acid Efficiently
To implement this synthesis effectively, operators must adhere to strict anhydrous protocols during the initial Grignard formation to prevent reagent decomposition. The process begins with the activation of magnesium metal using initiators like iodine or 1,2-dibromoethane, followed by the controlled addition of the dihalo substrate in THF. Once the Grignard reagent is established, dry ice or gaseous CO2 is introduced to effect carboxylation, after which the mixture is acidified to isolate the dialkoxy intermediate. The final hydrolysis step requires careful temperature control to ensure complete deprotection while avoiding charring or degradation of the product.
- Prepare the Grignard reagent by reacting 1,4-dihalo-2,5-dialkoxy benzene with magnesium metal in an anhydrous solvent like THF under nitrogen protection.
- Introduce dry carbon dioxide gas or dry ice to the Grignard reagent at low temperatures to effect carbochain growth and form the dialkoxy terephthalic acid disalt.
- Acidify the reaction mixture to isolate the intermediate, then perform acid hydrolysis to remove alkoxy groups and obtain the final 2,5-dihydroxy terephthalic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Grignard-based methodology represents a strategic opportunity to optimize cost structures and mitigate operational risks. By eliminating the dependency on high-pressure reactors, capital expenditure for new production lines is significantly reduced, and existing standard glass-lined or stainless steel reactors can be utilized without modification. The shorter reaction cycles compared to copper-catalyzed alternatives mean that facility throughput can be increased substantially without expanding the physical footprint of the plant. Additionally, the use of readily available starting materials like dihalobenzenes and magnesium metal ensures a stable supply chain, reducing the vulnerability to raw material shortages that often affect specialty chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the reduction in reaction time lead to a drastic simplification of the production process, which inherently lowers both fixed and variable costs. Without the need for expensive pressure-rated vessels and the associated safety monitoring systems, the overhead for production is minimized, allowing for more competitive pricing strategies in the market. Furthermore, the high yields reported in the patent embodiments suggest that raw material utilization is highly efficient, reducing waste disposal costs and maximizing the output per batch. This economic efficiency is further enhanced by the use of common solvents like THF, which are easily recovered and recycled in standard distillation units.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as magnesium, carbon dioxide, and simple halo-aromatics ensures that the supply chain remains robust against market fluctuations. Unlike processes requiring exotic catalysts or specialized high-pressure gases, this method utilizes materials that are globally sourced and readily available in bulk quantities. The operational simplicity also means that production can be scaled up or down rapidly in response to market demand without the long lead times associated with commissioning complex high-pressure infrastructure. This flexibility is crucial for maintaining continuity of supply for downstream customers in the polymer and pharmaceutical sectors.
- Scalability and Environmental Compliance: Operating at normal pressure significantly reduces the environmental and safety risks associated with chemical manufacturing, simplifying the permitting process and ongoing regulatory compliance. The waste streams generated are primarily aqueous acid washes and spent solvents, which are well-understood and manageable through standard wastewater treatment and solvent recovery systems. The high purity of the crude product reduces the need for extensive chromatographic purification, thereby minimizing the volume of organic waste generated during the finishing stages. This alignment with green chemistry principles makes the process attractive for companies aiming to reduce their carbon footprint and improve their sustainability profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Grignard-based synthesis route for DHTA. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for large-scale production.
Q: What are the primary advantages of this Grignard method over the Kolbe-Schmitt reaction?
A: Unlike the Kolbe-Schmitt reaction which requires high-pressure carbon dioxide and poses significant safety risks, this Grignard-based method operates under normal atmospheric pressure, significantly reducing equipment costs and operational hazards while maintaining high yields.
Q: What purity levels can be achieved with this synthesis route?
A: The patent data indicates that through optimized separation and purification steps, including thorough washing and drying, product purity can consistently reach between 98% and 99%, making it suitable for high-performance polymer applications.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes standard reaction conditions such as normal pressure and common solvents like THF, avoiding the need for specialized high-pressure reactors, which facilitates easier commercial scale-up and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Dihydroxy Terephthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the development of next-generation polymers and electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global supply chains. We are committed to delivering 2,5-dihydroxy terephthalic acid with stringent purity specifications, supported by our rigorous QC labs that verify every batch against international standards. Our expertise in Grignard chemistry allows us to optimize this specific route for maximum efficiency and safety, providing our partners with a reliable source of this essential intermediate.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can benefit your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this atmospheric pressure process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of our high-purity materials into your manufacturing workflow.
