Advanced Liquid-Phase Synthesis of Fmoc-L-Orn(Boc)-Gly-OH for Commercial Atosiban Production
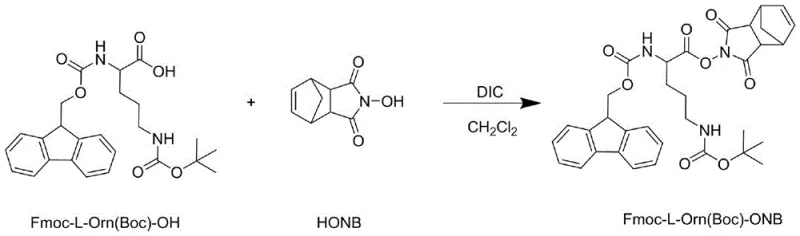
The pharmaceutical industry's relentless pursuit of cost-effective and scalable synthetic routes for complex peptide therapeutics has found a significant breakthrough in patent CN116410260A. This intellectual property details a novel preparation method for Fmoc-L-Orn(Boc)-Gly-OH, a critical dipeptide fragment used in the synthesis of Atosiban, a potent tocolytic agent widely utilized for managing preterm labor. Unlike traditional methodologies that rely heavily on resource-intensive solid-phase peptide synthesis (SPPS), this invention leverages an optimized liquid-phase peptide synthesis (LPPS) strategy. By utilizing N-hydroxy-5-norbornene-2,3-dicarboximide (HONB) as a superior activating agent in conjunction with N,N'-diisopropylcarbodiimide (DIC), the process achieves exceptional purity levels exceeding 99.7% without the necessity for column chromatography. For global procurement leaders and R&D directors, this technology represents a paradigm shift towards more sustainable and economically viable manufacturing of high-purity pharmaceutical intermediates, directly addressing the challenges of cost and scalability in the production of oxytocin receptor antagonists.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Atosiban short peptide fragments has been dominated by solid-phase peptide synthesis (SPPS) techniques. While SPPS offers advantages in terms of automation and speed for small-scale library generation, it presents severe economic and logistical bottlenecks when applied to the commercial manufacturing of specific dipeptide building blocks like Fmoc-L-Orn(Boc)-Gly-OH. The conventional SPPS route necessitates the initial loading of Fmoc-Gly-OH onto expensive polymeric resins, followed by iterative deprotection and coupling cycles. This approach inherently requires a vast excess of protected amino acids and coupling reagents to drive reactions to completion on the solid support, leading to substantial raw material wastage. Furthermore, the final cleavage step to release the peptide from the resin consumes additional hazardous reagents and generates significant volumes of difficult-to-treat waste. The cumulative cost of the resin, the excessive reagent consumption, and the complex downstream processing renders the traditional SPPS route prohibitively expensive for large-scale production of this specific intermediate, creating a pressing need for a more efficient alternative.
The Novel Approach
The methodology disclosed in CN116410260A fundamentally re-engineers the synthesis pathway by transitioning to a solution-phase condensation strategy that circumvents the inherent inefficiencies of solid supports. This novel approach utilizes a stoichiometric ratio of reactants close to 1:1, drastically reducing the consumption of valuable protected amino acids compared to the multi-fold excesses required in SPPS. The core innovation lies in the generation of a highly reactive active ester intermediate, specifically Fmoc-L-Orn(Boc)-ONB, which exhibits superior stability and reactivity profiles compared to traditional succinimide esters. By conducting the coupling reaction in a carefully engineered ternary solvent system comprising tetrahydrofuran (THF), ethanol, and water, the process ensures excellent solubility for both reactants while maintaining the integrity of the sensitive peptide bond. This liquid-phase method not only simplifies the operational workflow by eliminating resin handling and cleavage steps but also facilitates straightforward purification through crystallization, making it ideally suited for the commercial scale-up of complex peptide fragments required for Atosiban manufacturing.
Mechanistic Insights into HONB-Mediated Active Ester Coupling
The chemical elegance of this synthesis lies in the precise activation of the carboxyl group of Fmoc-L-Orn(Boc)-OH. The reaction initiates with the formation of an O-acylisourea intermediate upon treatment with DIC, which is subsequently converted into the stable active ester Fmoc-L-Orn(Boc)-ONB through nucleophilic attack by HONB. The choice of HONB over common activators like HOSu is critical; experimental data within the patent indicates that the norbornene-based active ester provides higher reaction yields and significantly lower impurity profiles. The steric and electronic properties of the HONB moiety appear to facilitate a more efficient aminolysis during the subsequent coupling step, minimizing racemization risks and side reactions. This activation step is conducted in dichloromethane at temperatures below 0°C to suppress potential side reactions, ensuring the generation of a high-quality intermediate ready for immediate use in the next stage.
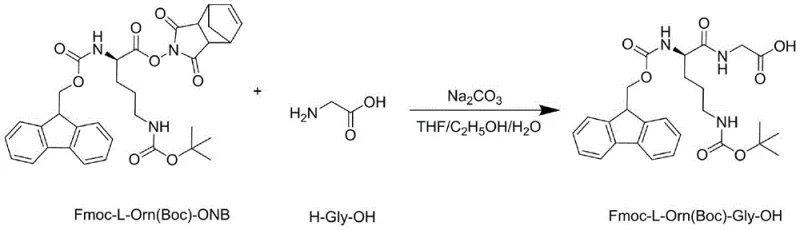
Following activation, the coupling with glycine proceeds via a nucleophilic attack of the glycine amine on the carbonyl carbon of the active ester. A crucial mechanistic feature of this process is the solvent engineering employed to manage the phase behavior of the reactants. Glycine is dissolved in an alkaline aqueous solution (pH 8-9) to form the soluble glycinate salt, while the hydrophobic active ester is dissolved in THF. The addition of ethanol acts as a co-solvent that bridges the polarity gap between the aqueous and organic phases. This miscibility enhancement ensures intimate molecular contact between the nucleophile and the electrophile, accelerating the reaction kinetics at mild temperatures of 20-25°C. Moreover, ethanol plays a protective role by suppressing the hydrolysis of the active ester in the alkaline medium, a common degradation pathway that leads to the regeneration of the starting material Fmoc-L-Orn(Boc)-OH. This suppression of hydrolysis is key to achieving the reported high purity of >99.7% and yields around 85-88%.
How to Synthesize Fmoc-L-Orn(Boc)-Gly-OH Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this dipeptide intermediate with pharmaceutical-grade quality. The process is designed to be operationally simple, avoiding the need for specialized equipment associated with solid-phase synthesizers or complex chromatographic purification systems. By adhering to the specific molar ratios and solvent compositions identified in the optimization studies, manufacturers can consistently achieve high purity suitable for direct use in subsequent peptide elongation steps. The detailed standardized synthesis steps for implementing this efficient liquid-phase route are provided in the guide below.
- Activate Fmoc-L-Orn(Boc)-OH using DIC and HONB in dichloromethane at below 0°C to form the active ester intermediate.
- Prepare an alkaline aqueous solution of glycine and add organic co-solvents (THF/Ethanol) to create a homogeneous reaction phase.
- Couple the active ester with the glycine solution at 20-25°C, maintaining pH 8-9, followed by extraction and crystallization to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this liquid-phase synthesis technology offers transformative advantages in terms of cost structure and supply reliability. The elimination of solid-phase resin, which is a significant cost driver in traditional peptide synthesis, immediately lowers the bill of materials. Furthermore, the ability to use near-stoichiometric amounts of expensive protected amino acids like Fmoc-L-Orn(Boc)-OH reduces raw material inventory costs and minimizes waste disposal expenses associated with excess reagents. The simplified downstream processing, which relies on extraction and crystallization rather than column chromatography, translates to faster batch cycle times and reduced solvent consumption. These factors collectively contribute to a substantially more competitive cost position for the final API, enabling better margin management in the highly price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The transition from solid-phase to liquid-phase synthesis fundamentally alters the cost equation by removing the requirement for expensive polymeric resins and the large excesses of amino acids typically needed to drive solid-phase reactions to completion. By utilizing a stoichiometric approach with high-efficiency coupling agents like HONB, the process maximizes atom economy and minimizes the waste of high-value starting materials. Additionally, the avoidance of column chromatography for purification significantly reduces the consumption of silica gel and elution solvents, leading to lower operational expenditures and waste treatment costs. This streamlined approach ensures that the production of Fmoc-L-Orn(Boc)-Gly-OH is economically viable even at large scales, providing a clear pathway for cost reduction in atosiban manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain security by reducing dependency on specialized solid-phase reagents and resins that may have limited suppliers or long lead times. The use of commodity chemicals such as THF, ethanol, and dichloromethane ensures that solvent supply remains stable and cost-effective. Furthermore, the demonstrated scalability of the process, with successful runs from 0.1 mol up to 1.0 mol showing consistent yields and purity, indicates a low risk of failure during technology transfer to commercial plants. This reliability allows for more accurate production planning and reduces the risk of stockouts for critical peptide intermediates, ensuring continuous availability for downstream API production.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers significant benefits by generating less hazardous waste compared to traditional solid-phase methods. The reduction in solvent volume and the elimination of resin waste simplify the effluent treatment process, aligning with increasingly stringent environmental regulations. The process operates under mild conditions (20-25°C) and uses standard reaction vessels, making it easily scalable from pilot plant to multi-ton commercial production without the need for complex engineering modifications. This ease of scale-up, combined with the high purity achieved without extensive purification, supports the commercial scale-up of complex peptide fragments while maintaining a smaller environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Fmoc-L-Orn(Boc)-Gly-OH based on the patented technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is liquid-phase synthesis preferred over solid-phase for this dipeptide fragment?
A: Liquid-phase synthesis eliminates the need for expensive peptide resins and large excesses of amino acids required in solid-phase methods. It allows for near-stoichiometric reactant ratios (close to 1:1), significantly reducing raw material costs and solvent waste while simplifying post-treatment by avoiding resin cleavage steps.
Q: What is the role of ethanol in the coupling reaction solvent system?
A: Ethanol serves a dual critical function in the THF/Ethanol/Water system. Firstly, it enhances the solubility and contact between the organic-soluble active ester and the water-soluble glycine salt, accelerating the reaction rate. Secondly, it effectively inhibits the hydrolysis of the active ester intermediate in the alkaline aqueous environment, thereby minimizing the formation of the Fmoc-L-Orn(Boc)-OH impurity and boosting overall purity.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the patent demonstrates successful scalability from 0.1 mol to 1.0 mol scales (Examples 2-4) with consistent high yields (85-88%) and purity (>99.7%). The process avoids column chromatography, relying instead on crystallization and extraction, which are standard unit operations easily adaptable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fmoc-L-Orn(Boc)-Gly-OH Supplier
The technological advancements detailed in patent CN116410260A underscore the immense potential of optimized liquid-phase peptide synthesis in modern pharmaceutical manufacturing. As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to leverage such innovative routes for the benefit of our global clients. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into reliable industrial supply. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of Fmoc-L-Orn(Boc)-Gly-OH meets the exacting standards required for Atosiban synthesis.
We invite you to collaborate with us to optimize your supply chain for peptide intermediates. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance your project's success.
