Advanced Synthesis of Semaglutide Main Chain Fragments for Commercial Scale-Up
Introduction to Next-Generation GLP-1 Analog Manufacturing
The rapidly expanding market for glucagon-like peptide-1 (GLP-1) receptor agonists, driven by the clinical success of drugs like Semaglutide, has placed immense pressure on the supply chain for high-quality peptide intermediates. Patent CN115160430B introduces a pivotal advancement in the synthesis of the Semaglutide main chain fragment, specifically addressing the challenges associated with the alpha-amino isobutyric acid (Aib) modification at the 8th position. Unlike traditional biological fermentation which is limited to natural amino acids, or solid-phase synthesis which struggles with steric hindrance, this invention proposes a robust liquid-phase strategy. The core innovation lies in a highly selective alkaline hydrolysis step that converts a protected precursor (Compound F) into the target carboxylic acid fragment (Compound G) with exceptional purity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding this shift from complex multi-step protection strategies to streamlined hydrolysis is critical for securing long-term supply stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Semaglutide analogs has been plagued by inefficiencies inherent to both biological and solid-phase chemical methods. In solid-phase peptide synthesis (SPPS), the introduction of alpha-amino isobutyric acid (Aib) at the 8th position creates significant steric hindrance, drastically reducing coupling yields and complicating the purification of the growing peptide chain. Furthermore, existing liquid-phase alternatives often rely on multiple hydrogenolysis steps to remove protecting groups like Cbz. These hydrogenation processes are not only operationally hazardous on a large scale but also necessitate repeated pH adjustments in acidic environments post-reaction. Such harsh conditions increase the risk of unintended hydrolysis of sensitive tert-butyl and methyl esters, leading to a complex mixture of homologous impurities that are difficult and costly to separate. This results in lower overall material throughput and inconsistent batch-to-batch quality.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by optimizing the final deprotection and assembly stages. Instead of relying on aggressive acidic hydrolysis or multiple catalytic hydrogenations, the process utilizes a controlled alkaline hydrolysis of Compound F. By employing specific bases such as Barium Hydroxide Octahydrate or Lithium Hydroxide in a solvent system comprising methanol or tetrahydrofuran, the reaction selectively cleaves the methyl ester to form the free carboxylic acid while preserving the acid-labile tert-butyl esters. This selectivity is achieved under mild conditions (0-40°C) over a defined period (6-10 hours). This approach effectively eliminates the need for repeated pH cycling and minimizes the formation of degradation byproducts. The result is a synthesis pathway that is not only chemically cleaner but also operationally simpler, offering a distinct advantage for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Selective Alkaline Hydrolysis
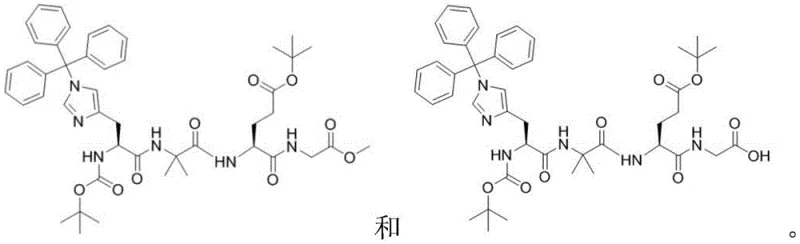
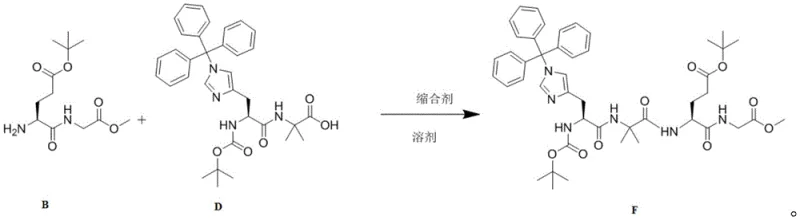
The chemical elegance of this process lies in the differential reactivity of ester groups under specific basic conditions. In the precursor Compound F, there are multiple ester functionalities: a methyl ester at the C-terminus and tert-butyl esters on the glutamic acid side chain and the histidine nitrogen protector. Standard strong base hydrolysis would typically saponify all ester groups indiscriminately. However, the patent specifies the use of Barium Hydroxide Octahydrate or Lithium Hydroxide at controlled molar ratios (1:0.5 to 1:1 relative to the substrate). The bulky nature of the tert-butyl group provides steric protection against the nucleophilic attack of the hydroxide ion, whereas the less hindered methyl ester is more accessible. Furthermore, the choice of solvent, such as a mixture of THF and water or pure methanol, modulates the solvation of the hydroxide ion, fine-tuning its nucleophilicity. This precise control ensures that the hydrolysis stops at the mono-acid stage (Compound G) without stripping the essential Boc or OtBu protecting groups required for subsequent peptide couplings.
Impurity control is further enhanced by the physical properties of the intermediates. The reaction design facilitates the removal of unreacted starting materials and side products through straightforward crystallization rather than preparative HPLC. For instance, after the condensation of Compound B and Compound D to form Compound F, the addition of water induces crystallization of the product, leaving soluble impurities in the mother liquor. This reliance on phase separation and crystallization rather than chromatography is a hallmark of a process designed for industrial viability. It ensures that the final high-purity pharmaceutical intermediate meets stringent specifications with minimal solvent waste and processing time, directly addressing the purity concerns of regulatory bodies.
How to Synthesize Semaglutide Main Chain Fragment Efficiently
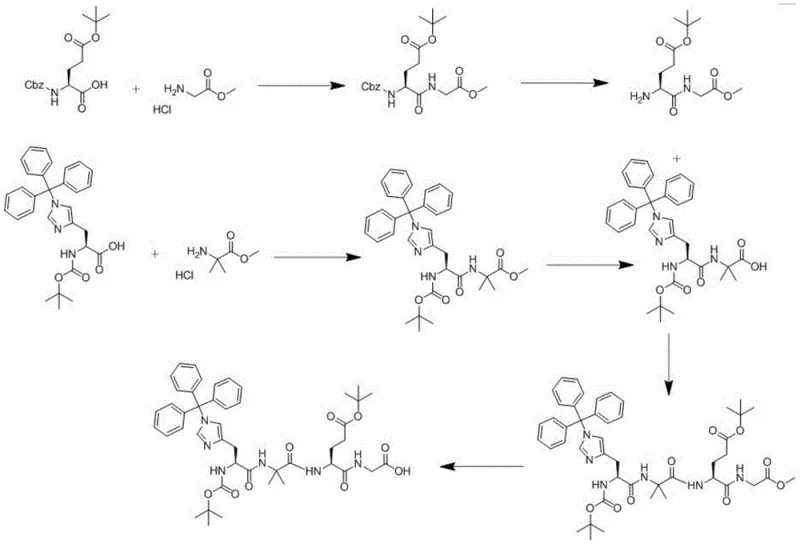
The synthesis protocol outlined in the patent provides a clear roadmap for producing the target backbone segment with high reproducibility. The process begins with the preparation of key building blocks, such as the glutamic acid derivative (Compound B) and the histidine-Aib dipeptide (Compound D), which are then coupled using standard peptide coupling reagents like HBTU and HOBt. The critical final step involves the selective hydrolysis described previously. This route is designed to be telescoped where possible, reducing isolation steps and maximizing yield. For process chemists looking to implement this technology, the detailed reaction conditions regarding temperature, stoichiometry, and workup procedures are essential for replicating the high purity reported in the examples. The standardized synthesis steps below summarize the critical operational parameters required for successful execution.
- Condense Compound B (deprotected glutamic acid derivative) with Compound D (histidine-Aib fragment) using HBTU/HOBt coupling agents to form Compound F.
- Perform selective alkaline hydrolysis on Compound F using Barium Hydroxide Octahydrate or Lithium Hydroxide in methanol/THF at 0-40°C.
- Adjust pH to 4-5, extract with ethyl acetate, and purify the final Compound G via crystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route represents a significant opportunity to optimize the cost structure and reliability of the API supply chain. Traditional methods involving multiple hydrogenolysis steps require expensive palladium catalysts and specialized high-pressure equipment, which introduce bottlenecks and safety risks. By replacing these with a simple alkaline hydrolysis, the process drastically simplifies the equipment requirements and reduces the dependency on precious metal catalysts. This simplification translates directly into lower capital expenditure (CAPEX) for manufacturing facilities and reduced operating expenses (OPEX) due to lower catalyst consumption and waste disposal costs. Furthermore, the robustness of the crystallization-based purification ensures consistent quality, reducing the risk of batch failures that can disrupt supply continuity.
- Cost Reduction in Manufacturing: The elimination of multiple hydrogenolysis operations removes the need for expensive palladium-on-carbon catalysts and the associated filtration and recovery infrastructure. Additionally, the selective hydrolysis prevents the loss of valuable protecting groups, thereby improving the overall atom economy and reducing the amount of raw materials needed to produce a kilogram of the final intermediate. The process relies on commodity chemicals like lithium hydroxide and barium hydroxide, which are significantly cheaper and more readily available than specialized coupling reagents or catalysts used in alternative routes.
- Enhanced Supply Chain Reliability: By reducing the number of discrete reaction steps and avoiding complex chromatographic purifications, the lead time for production is significantly shortened. The use of stable, non-hazardous reagents for the key hydrolysis step mitigates the risk of supply disruptions caused by the scarcity of specialized catalysts. This robustness ensures that manufacturers can maintain consistent production schedules, meeting the demanding delivery timelines required by global pharmaceutical clients without compromising on quality or safety standards.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids unit operations that are difficult to enlarge, such as high-pressure hydrogenation or preparative HPLC. The reliance on crystallization for purification generates less solvent waste compared to chromatography, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. This makes the technology not only economically attractive but also compliant with increasingly stringent environmental regulations governing pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology resolves historical pain points in peptide intermediate manufacturing. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is liquid-phase synthesis preferred for the Aib-modified fragment of Semaglutide?
A: Solid-phase synthesis often suffers from reduced yields due to the large steric hindrance of alpha-amino isobutyric acid (Aib) at the 8th position. Liquid-phase synthesis allows for better control over this specific coupling, ensuring higher conversion rates and purity before final assembly.
Q: How does the novel hydrolysis method prevent side reactions?
A: By utilizing specific bases like Barium Hydroxide Octahydrate at controlled temperatures (0-40°C), the method selectively hydrolyzes the methyl ester without significantly affecting the tert-butyl ester groups, thereby minimizing homologous impurities and protecting group loss.
Q: What are the scalability advantages of this process?
A: The process replaces complex chromatographic purifications with robust crystallization steps and reduces the number of hydrogenolysis operations. This simplification makes the route highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semaglutide Intermediate Supplier
As the demand for GLP-1 analogs continues to surge, partnering with a manufacturer who possesses deep technical expertise in peptide chemistry is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthesis technologies like the one described in CN115160430B to deliver superior intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Semaglutide main chain fragment meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain for next-generation diabetes and obesity treatments. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative manufacturing capabilities can drive value and efficiency for your organization.
